A Case Report of Cardiac Papillary Fibroelastoma; Is It as Benign as it Seems?
A Case Report of Cardiac Papillary Fibroelastoma; Is It as Benign as it Seems?
Vraynko E¹*. Zafirovska P². Andova V¹. Vavlukis M.¹ Georgievska Ismail Lj. ¹
1. University Clinic of Cardiology, Skopje, R. N. Macedonia.
2. Zan Mitrev Clinic, Skopje, R. N. Macedonia.
*Correspondence to: Vraynko E, University Clinic of Cardiology, Skopje, R. N. Macedonia.
Copyright
© 2024 Vraynko E. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 04 July 2024
Published: 18 July 2024
Abstract
Cardiac papillary fibroelastoma (PFE) is one of the most common types of benign cardiac tumors that consists of multiple papillary fronds arising from a central stalk. The prevalence is 48-61% of all cardiac tumors. Even though most patients with PFE are asymptomatic, they may present with dyspnea, chest pain, clinical heart failure symptoms, thromboembolic complications, or even sudden cardiac death. Herein, we present the case of a 44-year-old female patient with post-toxic cardiomyopathy and intracardiac masses with a past medical history of non-Hodgkin lymphoma treated with chemotherapy and mediastinal radiation therapy.
The patient complained of fatigue and dyspnea. On transthoracic echocardiography (TTE), a highly mobile small mass attached to the anterolateral left ventricular wall was detected. Cardiac magnetic resonance revealed a left ventricle with characteristics of post-toxic cardiomyopathy, severely reduced global contractility with an EF of 34%, and a cardiac mobile mass (11x7mm) attached to a small stalk to the mid-segment of the anterior wall of the left ventricle, with characteristics consistent with papillary fibroelastoma and a differential diagnosis for myxoma or metastasis. A control PET/CT scan was also performed, and no increased metabolic activity of the intracardiac masses was found, ruling out cardiac metastasis. The cardio-surgery team recommended conservative treatment because of the high operative risk, considering the multiple comorbidities of the patient. After 15 months of follow-up, the patient was in a good clinical condition, and the control TTE examination didn’t show any changes in the dimensions or structure of the intracardiac masses.
Papillary fibroelastomas are rare, benign cardiac tumors with an unknown etiology. The development of imaging methods enables the adequate and early diagnosis of cardiac tumors. Even though surgical resection is considered the gold standard for the treatment of PFE, an individualized approach is essential for the decision on the optimal management and prevention of complications.
Key words: cardiac papillary fibroelastoma, cardiac benign tumors, intracardiac masses.
A Case Report of Cardiac Papillary Fibroelastoma; Is It as Benign as it Seems?
Introduction
Primary cardiac tumors are rare conditions, where the signs and symptoms depend on the side of the cardiac involvement. (1) According to a comprehensive review, by Gowda, the prevalence is approximately 0.02% of autopsie results. (2) Generally, 75% of the cardiac tumors are benign. Cardiac papillary fibroelastoma (PFE) is the most common type of cardiac tumor with a prevalence of 48-61% of all cardiac tumors. It is a benign tumor that consists of multiple papillary fronds arising from a central stalk. (3) Histopathologically, these tumors are avascular, with a layer of endocardial cells surrounding the inner matrix, which is a hyaline stroma with different amounts of elastic fibers, fibroblasts and smooth muscle cells. According to a retrospective follow-up study of patients with papillary fibroelastoma, PFEs are slowly-growing tumors with a rate of 0.5-0.9 ml/y. The most common site of occurrence are heart valves, even though they can be found on any endocardial surface. It has been suggested that cardiac papillary fibroelastoma is not a real tumor but lesion which can be reactivated by a mechanical trauma, radiotherapy or inflammatory processes. (4,5)
Most of the patients with PFE are generally asymptomatic. The signs and symptoms vary from dyspnea, chest pain, clinical heart failure symptoms to thromboembolic complications, even sudden cardiac death. In some patients, cerebrovascular complications like TIA or stroke may be the first sign of suspicion of PFE. (3) According to a retrospective study (4), 33 out of 67 patients (pts) with PFE presented an embolic event, with 27 of them, having neurological complications (TIA in 9 pts, stroke in 22, both in 4 pts). It was shown that the growth rate was not associated with a higher embolic potential. (4)
It has been reported that, PFEs are the most underdiagnosed benign cardiac tumors. Transthoracic echocardiography is the initial diagnostic modality to evaluate papillary fibroelastoma. However, despite the use of TTE, in most of the cases PFEs are not diagnosed due to their minimal effect on hemodynamics and clinical manifestations. Considering the strong association of PFEs with radiation and surgical trauma, the patients with primary cardiovascular diseases should undergo regular echocardiography check-ups, aiming toward early diagnosis of the PFEs and other cardiac tumors. (6,7) Even though, TTE plays an important role in the diagnostic approach of the PFEs, the cardiac magnetic resonance (CMR) remains the gold standard, due to the possibility for non-invasive tissue characterization. (5,7)
Herein, we present a case of a 44-year-old female patient with a past medical history of non-Hodgkin lymphoma and treated with chemotherapy and mediastinal radiotherapy, who after 18 years in a routine cardiology control presented with intracardial masses that with non-invasive diagnostic modalities evaluation were consistent with cardiac papillary fibroelastoma.
Case Report
A 44-year-old female patient presented for the regular cardiol?gy examination. She complained of fatigue and dyspnea. She had a past medical history of large cell non-Hodgkin lymphoma diagnosed in 2003, and treated with chemotherapy and mediastinal radiation therapy during 2003-2004 with complete remission of the disease.
Ten years later the patient was diagnosed with heart failure and treated conservatively with ACE-inhibitors, beta blockers, loop diuretics and mineralocorticoids.
On a routine TTE follow-up, a highly mobile small mass attached to the anterolateral left ventricular wall was detected (Image.1 -2). Thrombus was the initial suspicion and the patient was placed on oral anticoagulants and referred for further evaluation with cardiac magnetic resonance.
Image 1. 4Ch TTE of mass in LV and apex of RV
Image 2. Mass attached to anterolateral left ventricular wall
Cardiac magnetic resonance revealed left ventricle with characteristics of post-toxic cardiomyopathy and severely reduced global contractility with EF 34% (Image. 3- midmyocardial late gadolinium enhancement-LGE). Two chamber cine best visualized a cardiac mobile mass (11x7mm) attached to a small stalk to mid segment of the anterior wall of the left ventricle. The mass showed isointense signal on T1 (Image. 4), high intensity signal on T2 (Image. 5) and homogeneous late gadolinium enhancement (Image. 6). Characteristics were consistent with papillary fibroelastoma with differential diagnosis for myxoma or metastasis.
Image 3. CMR- midmyocardial LGE in IVS
Image 4. CMR- isointense signal on T1
Image 5. CMR- high intensity signal on T2
Image 6. CMR- homogeneous LGE of the intracardiac mass
Two years ago, a PET/CT scan was performed because of intensive fatigue, cough and suspicion of recurrence of non-Hodgkin lymphoma. There were no signs of recurrence of the disease on the PET /CT scan, however, in the lung parenchyma there were regions of patchy reticular changes alongside the airways, a diffuse non-homogeneous activity of FDG was also observed, which was typical for an inflammatory process, and diffusely increased metabolic activity in the wall of the right ventricle was present. In the meantime, she had her lung examined with findings for obstructive-restrictive type of respiratory insufficiency and bronchodilator therapy was initiated. She was a non-smoker, and had no family history for cardiovascular diseases.
A control PET/CT scan was also performed, in order to rule out a malignant process in the heart and the results were similar to the previous finding, no increased metabolic activity of the intracardiac masses was found, ruling out cardiac metastasis.
Second, smaller mass was observed by TTE that was also previously there but due to the location in the apex of the right ventricle was not detected earlier. The patient was under optimal medical treatment for systolic heart failure including sacubitril/valsartan and empagliflozin which were initiated, as well as anticoagulation therapy according to the recommendations for prevention of thromboembolic events from intracardiac masses. The cardio-surgery team was consulted for surgical resection of the intracardiac masses, who recommended conservative treatment because of a high operative risk at that moment, considering the multiple comorbidities of the patient, including the heart failure and respiratory insufficiency.
After 15 months of follow-up, the TTE examinations didn’t show any changes in the dimensions or structure of the intracardiac masses, excluding metastatic disease and myxoma as differentials, and proving papillary fibroelastomas as the diagnosis. A control PET/CT scan showed no changes from the previous. Also, the patient didn’t report any cardiovascular or neurological complications during the follow-up period.
Discussion
Cardiac papillary fibroelastomas are primary benign cardiac tumors which are considered to be the second most common type of benign cardiac tumor, following myxomas. (8) However, according to a review study of patients at the Mayo Clinic, PFE was documented to be even more common than cardiac myxoma, with an incidence of 0.009% of patients who underwent echocardiographic examination. (9,10)
Generally, PFE originates from the endocardium of the valvular structures with the aortic and mitral valve (29% and 25% respectively) been reported to be the most common locations. (11) In 16% of the cases, a non-valvular origin of the PFE was observed. In rare cases, a multifocal tepete (carpet like) PFE in the left-ventricular cavity has been reported. (11) In our case, PFEs were in multifocal locations including the anterolateral wall of the left ventricular cavity and apex of the right ventricle. In a literature review, the multifocal location is rare and is reported in 2-13% of the cases. (12) Whereas, cardiac papillary fibroelastomas can arise from a variety of cardiac structures, they must be considered in the differential diagnosis of the intracardiac masses. (9)
The etiopathology of papillary fibroelastoma is unclear. There are several hypotheses postulated on the pathophysiology of PFEs. One of the mechanisms suggests that the PFE is acquired and begins as a microthrombi, which primarily joins in a lesion of endothelium of a cardiac structure and overtime these microthrombi evolve to PFEs. Among other possible causes of PFE are iatrogenic (radiation therapy, surgery), inflammation due to unusual response of endocardium to traumas, infections or hamartomas. (11,13) In this case, even though the definite cause of PFE is not well known, radiation and chemotherapy are considered as possible circumstances for the occurrence of PFE in our patient.
The presentation of PFE varies in a wide distribution of age. (14) The patient can be asymptomatic, and PFEs’ are often diagnosed incidentally. According to a retrospective-prospective study on 162 patients for clinical and echocardiographic features of PFE, it was diagnosed incidentally in asymptomatic patients with another underlying cardiovascular disease. In our case, the patient had multiple comorbidities, including heart failure, pulmonary fibrosis, hypothyroidism and a history of non-Hodgkin lymphoma, and presented with the symptoms of dyspnea and fatigue which can be the manifestation of any of the underlying diseases and is not specific for cardiac PFE.
The most characteristic feature of this benign tumor is the potential of embolization resulting in life-threatening cardiac and neurological complications, including, TIA, stroke, blindness, myocardial infarction as well as sudden cardiac death. The embolization may occur as a result of detachment of a fragment of PFE or either of a thrombus previously formed on the surface of the PFE. In symptomatic patients the most common complications are cerebrovascular events. A retrospective review by Tamin et al. (10) reported that the rate of cerebrovascular events at 10 years was 2.4 times higher in matched patients who were treated with surgery and 3.4 times higher in patients who were treated medically. They also reported that the five years mortality is twice higher in PFE patients as compared to age and sex-matched controls. On the other hand, in a meta-analysis of 725 PFE cases of Gowda et al. (2), it was revealed that tumor mobility is the only independent predictor of mortality and embolic complications in patients with cardiac PFE.
The diagnostic tool of choice for the initial assessment of cardiac PFE is 2D transthoracic echocardiography (TTE). It has been reported that the 2D TTE has a sensitivity of 88.9% and a specificity of 87.8% for detection of PFE greater than 2mm. (15) The studies show that transesophageal echocardiography (TEE) has higher sensitivity for diagnosis of PFEs in comparison to 2D TTE. (14) Tamin et al. (10) showed that 51% of patients with PFE were diagnosed with 2D TTE in comparison to 84% of patients diagnosed with TEE. (9) According to a retrospective-prospective study of clinical and echocadiographic features of PFE, there are typical echocariographic characteristics of PFE, which include: a round, oval or irregular shape of the tumor with well demarcated borders and homogenous texture, generally small (<20mm). Half of diagnosed PFE in this study had small stalks, which were mobile and distributed as uni- or multifocal lesions most often localised on valvular surfaces. (14) Despite these echocardiographic characteristics, many PFEs are not detected during echocardiographic examinations. Magnetic resonance imaging (MRI), multislice spiral computed tomography (MSCT) and positron emission tomography-computed tomography scan (PET/CT scan) are used for better delineation of these lesions and differential diagnosis of intracardiac masses. (11, 16) Considering the high temporal resolution, non-invasive tissue characterization and unresticted field of view, the cardiac MRI plays an imporatnt role in the diagnosis of cardiac PFE. (18) In the case series of Jorge et al. (19), which included two cases with intracardial methastases and two cases with benign cardiac tumors, cardiac MRI was decesive in diagnosis and provided a highly accurate histological diagnostic approach without use of an invasive procedure.
Even though the optimal medical management of the PFE is not yet defined, surgical resection is considered as the golden standard for its treatment. The surgical treatment is performed by the resection of the roof of the PFE and full thickness of the underlying endothelium, followed by pericardial patch or primary closure of the defect. (1,17,20) The recurrence of PFE after the surgical resection was reported to be 0.04% of all PFE cases. (1,10) In the study of Sun et al. (14), there was no case of a recent lesion in a total of 64 patients who underwent an echocardiographic examination after an average of 630 days follow-up from the surgical treatment. According to this study, the decision of surgical treatment must be based on the size, mobility, location and potential or strength of association of the tumor with the symptoms. Thus, the recommendations for surgery from this study include the following: 1.The excision of an isolated right sided PFE is indicated for large and mobile tumors, tumors resulting in hemodynamically significant embolization, and the presence of patent foramen ovale with a sizeable right-to-left shunt, 2. Left-sided, small and non-mobile PFE in asymptomatic patients, 3. In young, or patients with other cardiovascular disease, with large (>10mm), especially mobile PFE, with low surgical risk and high risk of embolization. (14,21, 22) In our case, the surgical resection of the PFEs was denied by the cardiosurgery team considering the high operative risk of the patient as a result of multiple comorbidities including respiratory insufficiency.
In terms of conservative treatment, there is a lack of recomendation for optimal anticoagulation and/or antiplatelet treatment. In two retrospective studies of Gowda et al.(2) and Tamin et al. (10) separately, various regimes for medical treatment were defined including the anticoagulation therapy with warfarin or heparin, antiplatelet therapy as dual antiplatelet therapy, or monotherapy with acetylsalicylic acid or clopidogrel. There hasn’t been a larger study describing the use of direct oral anticoagulation drugs in patients with PFE. (1,2,10, 23) In the study, Tamin et al. (10) reported neurological complications in 121 patients who were on medical treatment with anticoagulant or antiplatelet drugs (22% were on warfarin, 47% on acetylsalicylic acid, 1% on clopidogrel and 2 % on dual antiplatelet therapy). The individual cost-benefit effect is essential for the decision about the medical treatment of patients with PFE, considering the bleeding risk and risk of thromboembolic events. (1)
Conclusion
Papillary fibroelastomas are rare, benign cardiac tumors with an unknown etiology. The development of the imaging methods enables adequate and early diagnosis of cardiac tumors. The TTE is considered to be the diagnostic tool for initial assessment of these patients but CMR is the gold standard for further evaluation and non-invasive tissue characterization. More clinical trials are needed, in order to define adequate management and prevention of thromboembolic events in patients with PFE. Even though surgical resection is considered as the gold standard for the treatment of PFE, an individualized approach is essential for the decision on the optimal management and prevention of complications.
Reference
1. Yiu AC, Hussain A, Okonkwo UA, O'Shea JP. A Case of Cardiac Papillary Fibroelastoma - An Increasingly Described Cardiac Tumor with Fatal Consequences. Hawaii J Health Soc Welf. 2021 Sep;80(9):207-211. PMID: 34522888; PMCID: PMC8433577.
2. Gowda RM. Khan IA. Nair CK. Et al. Cardiac papillary fibroelastoma: a comprehensive analysis of 725 cases. Am Heart J. 2003;146(3):404–410. doi: 10.1016 /S0002-8703(03)00249-7.
3. Kurmann R. El-Am E. Ahmad A. et al. Cardiac Masses Discovered by Echocardiogram; What to Do Next? Structural Heart, 2023, 100154. doi.org/10.1016/j.shj.2022. 100154.
4. Kurmann RD, El-Am EA, Sorour AA, et al. Papillary Fibroelastoma Growth: A Retrospective Follow-Up Study of Patients With Pathology-Proven Papillary Fibroelastoma. J Am Coll Cardiol. 2021;77(16):2154-2155. doi: 10.1016/j.jacc.2021.02.027.
5. Cristina B. Souhayla S. Jose S. et al. Left ventricular papillary fibroelastoma. Portugese Journal of Cardiothoracis and Vascular Surgery. 2023;30(1):49-52. doi:https://doi.org /10.48729/pjctvs.332
6. Edwards FH. Hale D. Cohen A. et al. Primary cardiac valve tumors. Ann Thorac Surg. 1991; 52: 1127–1131.
7. Elbardissi AW, Dearani JA, Daly RC, Mullany CJ, Orszulak TA, Puga FJ, Schaff HV. Survival after resection of primary cardiac tumors: a 48-year experience. Circulation. 2008; 30;118(14 Suppl):S7-15. doi: 10.1161/CIRCULATIONAHA.107.783126.
8. Reynen K. Frequency of primary tumors of the heart. Am J Cardiol. 1996; 77 (1):107. doi: 10.1016/S0002-9149(97)89149-7.
9. Sabet A. Haghighiabyaneh M. Tazelaar H et al. The Clinical Dilemma of Cardiac Fibroelastic Papilloma. Structural heart. 2018; 2(4):274-280. doi:https://doi.org /10.1080 /24748706. 2018.1475782
10. Tamin SS. Maleszewski JJ. Scott CG. et al. Prognostic and bioepidemiologic implications of papillary fibroelastomas. J Am Coll Cardiol. 2015;65(22):2420–2429. doi: 10.1016/j.jacc.2015.03.569.
11. Neerod J. Michael K. Donogh M. et al. Papillary fibroelastoma of the aortic valve - a case report and literature review. J Cardiothorac Surg. 2010; 5:84. doi: 10.1186/1749-8090-5-84
12. Vittala SS. Click RL. Challa S. et al. Multiple papillary fibroelastomas. Circulation. 2012; 126: 242-243. doi: 10.1161/CIRCULATIONAHA.111.049569
13. Bicer M. Cikirikcioglu M. Pektok E. et al. Papillary fibroelastoma of the left atrial wall: a case report. Journal of Cardiothoracic Surgery. 2009;4:28. doi: 10.1186/1749-8090-4-28.
14. Jing S. Craig A. Xing Y. et al. Clinical and Echocardiographic Characteristics of Papillary Fibroelastomas-a Retrospective and Prospective Study in 162 Patients. Circulation. 2001; 103:2687–2693. doi: https://doi.org/10.1161/01.CIR.103.22.2687
15. Arvind D. Lawrence L. Papillary Fibroelastoma. National Library of Medicine, August 8, 2022. Accessed: https://www.ncbi.nlm.nih.gov/books/NBK549829/
16. Parthenakis F. Nyktari E. Patrianakos A. et al. Asymptomatic papillary fibroelastoma of the aortic valve in a young woman-a case report. Cardiovascular Ultrasound . 2009;7:43. doi: 10.1186/1476-7120-7-43.
17. Gopaldas RR. Atluri PV. Blaustein AS. et al. Papillary fibroelastoma of the aortic valve: operative approaches upon incidental discovery. Tex Heart Inst J. 2009;36((2)):160–163. PMID: 19436815
18. Edward H. Muhammad Sh. Arul G. et al. MRI assessment of cardiac tumors: part 1, multiparametric imaging protocols and spectrum of appearances of histologically benign lesions. Quant Imaging Med Surg. 2014; 4(6):478–488. doi:10.3978/j.issn. 2223-4292.2014.11.23
19. Jorge M. Mauro A. Manuel G. et al. Magnetic Resonance Imaging in the Study of Cardiac Masses: A Case Series. Medicina (Kaunas) 2023;59(4):705. doi:10.3390/ medicina59040705.
20. Ngaage DL. Mullany CJ. Daly RC. et al. Surgical treatment of cardiac papillary fibroelastoma: a single center experience with eighty-eight patients. Ann Thorac Surg. 2005;80((5)):1712–1718. doi: 10.1016/j.athoracsur.2005.04.030.
21. Zhang, M., Liu, X., Song, Z. et al. Cardiac papillary fibroelastoma: a retrospect of four cases. J Cardiothorac Surg 8, 65 (2013). https://doi.org/10.1186/1749-8090-8-65
22. Srikanth Y. Bella M. Pratik M. et al. Cardiac papillary fibroelastoma: The need for a timely diagnosis. World J Clin Cases 2017; 5(1): 9-13. doi: 10.12998/wjcc. v5.i1.9
23. Anand S, Sydow N, Janardhanan R. Papillary fibroelastoma diagnosed through multimodality cardiac imaging: a rare tumour in an uncommon location with review of literature. BMJ Case Rep. 2017; 2017:bcr2017219327. doi: 10.1136/bcr-2017-219327.
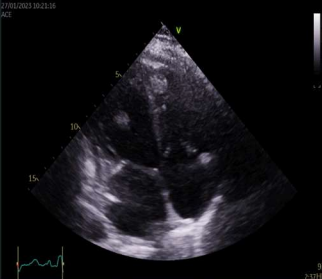
Figure 1
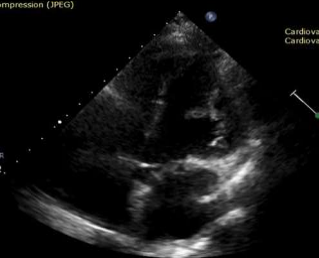
Figure 2
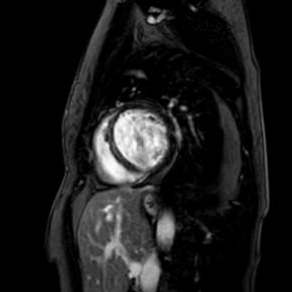
Figure 3
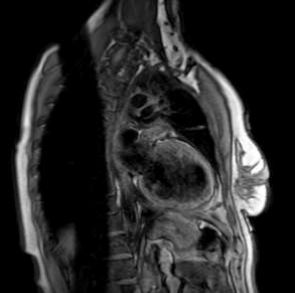
Figure 4
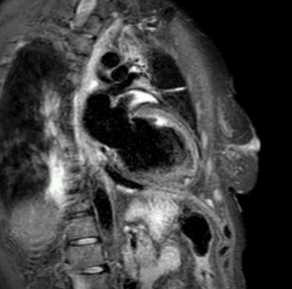
Figure 5
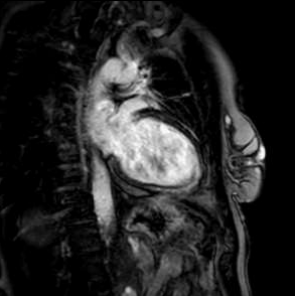
Figure 6
