An Alveolar Capillary Dysplasia with Misaligned Pulmonary Veins (ACDMPV) case series
An Alveolar Capillary Dysplasia with Misaligned Pulmonary Veins (ACDMPV) case series
Dr. Anusha *
*Correspondence to: Dr. Anusha CM, Consultant Pulmonologist, Consultant Manipal Hospital, Bangalore, India.
Copyright
© 2024 Dr. Anusha. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 21 Aug 2024
Published: 01 Sep 2024
Abstract
Alveolar capillary dysplasia with malpositioned pulmonary veins (ACD/MPV) is a rare and often fatal condition associated with the development of pulmonary veins. Since its initial description, significant research has led to a better understanding of the molecular mechanisms underlying ACD/MPV, and genetic studies have identified its association with genomic alterations at the FOXF1 locus. This has increased physician knowledge, and more than 200 cases have been reported to date, including genotyping of patients in the most recent report. Overall, this facilitates patient stratification and provides new insights into the study of pathogenesis. Here, we provide an overview of ACD/MPV management, including guidance for clinicians, and review ongoing research into the molecular mechanisms involved in severe pneumonia.
Keywords: pulmonary development, pulmonary vascular biology, neonatal lung disease, bronchopulmonary dysplasia, transcription factors
An Alveolar Capillary Dysplasia with Misaligned Pulmonary Veins (ACDMPV) case series
Clinical Presentation
Symptoms of alveolar capillary dysplasia with malpositioned pulmonary veins (ACD/MPV) are characterized by severe pulmonary edema (PH) and dyspnea and cyanosis caused by inadequate oxygenation. Birth weight and Apgar score are normal 4.5, and most patients develop symptoms within 24 hours of birth. -14 These malformations primarily affect the gastrointestinal tract but also affect the cardiovascular and genitourinary systems. Despite receiving supportive care, including mechanical ventilation (ECMO), most patients die of hypoxic respiratory failure within days or weeks of presentation. Atypical, mild cases of MPV presenting after 24 hours of life or in patients who survive the neonatal period have recently been described and will be discussed in the histology section.
More than 200 cases of ACD/MPV have been reported since it was first described in 1981. As a suggestion, there were two cases of ACD/MPV among 226 infants diagnosed over a 10-year period in Manitoba, Canada. It is unclear whether there were other cases of ACD/MPV among the other babies who died, as confirmation would require a medical examination or blood test. According to research, the probability is around 1/100,000. In the Netherlands, if calculated on the basis of all cases of ACD/MPV diagnosed since 1993 at the Radboudumc Amalia Children's Hospital in Nijmegen or at the Sofia Children's Hospital in Rotterdam ( 20 ), the incidence over time is approximately 1/200,000 births, this time (4.3 million 26). However, in the Netherlands, ACD/MPV can also be underdiagnosed and sometimes misdiagnosed as idiopathic PD due to lack of detection.
Diagnosis
The current gold standard for definitive diagnosis of ACD/MPV is lung histology. In most studies, the majority of patients were diagnosed posthumously. The median time to presentation for lung biopsy was 6.5 days. ACD/MPV was detected in 32% of infants who underwent open lung biopsy during ECMO therapy at the Sofia Children's Hospital in Rotterdam or the Radboudumc Amalia Children's Hospital in Nijmegen. All of these patients discontinued treatment because they did not benefit. Lung biopsy is recommended before starting ECMO therapy or within the first week of ECMO therapy if it is started, to avoid unnecessary and expensive ECMO therapy. Unequal distribution of involvement In some patients, it is recommended to obtain more than one lung sample to avoid false-positive results. 10,18,19,21,29 Chest X-rays do not show specific features for ACD/MPV but are unlikely to reveal abnormalities or pneumothorax caused by mechanical PA. 19,29 Computed tomography (CT) scans may show bilateral ground-glass infiltrates with septal line thickening,12,21 and CT angiography is not available because of limitations in evaluating most peripheral capillaries in the lungs. Photoacoustic imaging, however, overcomes the limitations of CT angiography by allowing high-resolution molecular imaging in different tissues. So far, photoacoustic imaging has only been used in biomedical research, but human studies have been very successful, and in the future it will provide an excellent tool for diagnosing ACD/MPV. There are no symptoms associated with ACD/MPV. However, if ACD/MPV is suspected to be widespread, such as in early-onset family members, genetic testing for FOXF1 locus deletions and mutations may aid early diagnosis. (MRI) and ultrasound-measured volumes (FLV) and reduced MRI signal can be used to assess fetal lung hypoplasia, which is associated with various lung developmental disorders, including ACD/MPV. Prognosis Antenatal counseling is sufficient, but postnatal care should be tailored to the individual patient. Histological examination should be performed to confirm the diagnosis.
On full evaluation, the lungs of ACD/MPV cases appear very large and a threefold increase in weight is expected due to increased tissue volume. It has arrived. In a normal lung, alveolar capillaries lie close to the alveolar epithelium lined with flattened type 1 pneumocytes to ensure adequate air exchange. In ACD/MPV, lung tissue is characterized by widespread thickening of alveolar septa and marked reduction in alveolar capillaries. In addition, sparse alveolar capillaries were located away from the alveolar epithelium, which consisted mainly of proliferating cuboidal type 2 pneumocytes (Figure 1a). Small blood vessels thicken due to hypertrophy of muscle fibers in the middle layer (Figure 1). 1b). In addition, the peripheral blood vessels in the bronchovascular system of most patients with ACD/MPV are adjacent to the pulmonary vessels outside the interlobular septum, most in the septum. This phenomenon is called “pulmonary vein misalignment” (Figure 1c).
Figure 1
Lung histology of a 2-week-old infant with ACD/MPV. (a) Immunostaining for CD31 (brown color) highlighting a reduced number of alveolar capillary endothelial cells located away (arrows) from the inner side of the alveoli (dashed lines) in ACD/MPV compared to control lung. (b) Illustration of a hypertrophic arterial wall (*) in hematoxylin and eosin stained lung tissue from an ACD/MPV case compared to control. (c) Illustration of the misaligned pulmonary veins (V) adjacent to the bronchiole (B) and thickened pulmonary arteriole (A) in trichrome stained ACD/MPV lung tissue. Magnifications: (a) 200×; (b) 50×; (c) 100×.
For example, the mild phenotype, including late presentation and long survival in patients with atypical ACD/MPV, is associated with the extent of lung involvement. has been described, 18,22 and a recent study has shown that all patients with atypical ACD/MPV have different, inconsistent histological findings. The disease remains difficult to diagnose due to incomplete medical data, different associations and, especially, different treatment modalities, 5 and the number of organisms is not equal. This is particularly important if there are no other problems related to poor lifestyle and the outcome depends solely on the severity of lung disease. Intubation and mechanical ventilation should be performed immediately upon presentation in most cases because of the very low oxygen saturation. Rarely, oxygen supplementation alone is sufficient to temporarily stabilize the patient's condition before ventilation is required. Injectable vasodilators such as milrinone, Cialis or epoprostenol. This may be satisfactory in the short term, but data suggest that multiple injections are necessary and that eventually severe hypoxemia may lead to death. At the time of writing, a patient with atypical ACD/MPV was surviving at 38 months of age on oral vasodilators and supplemental oxygen. If it is not clear, a threatening situation will arise. However, the majority of ACD/MPV patients cannot be weaned off ECMO, which is itself invasive, carries the risk of complications, and is very expensive. Perform an open lung biopsy before starting ECMO therapy, or at least before deciding to “second” ECMO therapy. Lung transplantation for patients with ACD/MPV. St. Louis Children's Hospital Indications. However, lung transplantation after ECMO treatment is a treatment option in patients selected for ACD/MPV. To be clear, many theories have been proposed with many aspects and conditions. ACD/MPV is thought to result from early disruption of the embryonic lung. By the end of the embryonic stage of lung development (five weeks of pregnancy), both lung buds are surrounded by a network of blood vessels connected to the organs. During the pseudoglandular stage (5-16 weeks of gestation), this network expands through angiogenesis when two buds branch into interstitial tissue. During the canalicular and saccular phases (weeks 16-26 and 26-38 of pregnancy, respectively), the network continues to expand, increasing capillary density in the lungs. Early embryonic and pseudoglandular stages are already affected. The lungs of ACD/MPV have thickened diaphragms and simple acini with a reduced number of type 1 pneumocytes, indicating that the saccular level, where alveolar sacs often form separate septa and are lined by type 1 pneumocytes, is still affected. ,46 ,47
The etiology of PH in ACD/MPV is still a matter of debate. Some believe that hypoxemia in infants may cause hypertrophic changes in the blood vessels, causing PH. Hypoxemia occurs when there is insufficient oxygen. In addition, this hypothesis is not consistent with the poor outcome in ACD/MPV patients, even when hypoxemia is reduced by mechanical ventilation or ECMO therapy. Another more widely accepted fact is that PH is the result of a reduction in the alveolar capillary bed. question.
The origin of the "misplaced pulmonary vessels" found in peripheral ACD/MPV lung tissue and whether they are truly misplaced is unknown. Given the presence of normal valves, the abnormal vessels most likely arise from bronchial arteries, which usually do not connect to larger bronchopulmonary branches. The "pulmonary vein malposition" arises from the bronchial artery and acts as a shunt vessel between the bronchi and pulmonary veins. Shaw-Smith proposed a link between ACD/MPV and haploinsufficiency of the Forkhead Box F1 (FOXF1) gene. Pathogenic variants, including single-digit variations (CNVs), point mutations, and rearrangements, are defined by region involving the FOXF1 gene or its regulatory region. 16,20 Most variants are registered in the Leiden Open Variation Database (LOVD).
Conclusion
ACD/MPV is a rare, almost always fatal lung disease. The patient had severe hypoxemia and pulmonary PH that progressed over time. Even if the doctor knows about ACD/MPV, it can be confused with idiopathic PH because of the similar symptoms. This delays the diagnosis and causes unnecessary pain and high medical costs for the patients. In case of a hopeful interruption of treatment, histological examination should be performed immediately. In addition, ideally an open lung biopsy is performed before starting ECMO or joint surgery. A small number of patients with atypical ACD/MPV may be considered for lung transplantation, but survival rates remain disappointing. ACD/MPV is associated with haploinsufficiency of FOXF1, a TF regulated by SHH that plays an important role during lung development. Despite phenotypic differences, prenatal or postnatal blood tests can facilitate early diagnosis and allow adequate feedback for assessment and decision making.
References
1. Eulmesekian P, Cutz E, Parvez B, et al. Alveolar capillary dysplasia: a six-year single center experience. J Perinat Med 2005; 33: 347–352. [PubMed] [Google Scholar]
2. Stankiewicz P, Sen P, Bhatt SS, et al. Genomic and genic deletions of the FOX gene cluster on 16q24.1 and inactivating mutations of FOXF1 cause alveolar capillary dysplasia and other malformations. Am J Hum Genet 2009; 84: 780–791. [PMC free article] [PubMed] [Google Scholar]
3. Bishop NB, Stankiewicz P, Steinhorn RH. Alveolar capillary dysplasia. Am J Respir Crit Care Med 2011; 184: 172–179. [PMC free article] [PubMed] [Google Scholar]
4. Al-Hathlol K, Phillips S, Seshia MK, et al. Alveolar capillary dysplasia. Report of a case of prolonged life without extracorporeal membrane oxygenation (ECMO) and review of the literature. Early Hum Dev 2000; 57: 85–94. [PubMed] [Google Scholar]
5. Sen P, Thakur N, Stockton DW, et al. Expanding the phenotype of alveolar capillary dysplasia (ACD). J Pediatr 2004; 145: 646–651. [PubMed] [Google Scholar]
6. Al-Hathlol K, Idiong N, Hussain A, et al. A study of breathing pattern and ventilation in newborn infants and adult subjects. Acta Paediatr 2000; 89: 1420–1425. [PubMed] [Google Scholar]
7. Michalsky MP, Arca MJ, Groenman F, et al. Alveolar capillary dysplasia: a logical approach to a fatal disease. J Pediatr Surg 2005; 40: 1100–1105. [PubMed] [Google Scholar]
8. Ma Y, Jang MA, Yoo HS, et al. A novel de novo pathogenic variant in FOXF1 in a newborn with alveolar capillary dysplasia with misalignment of pulmonary veins. Yonsei Med J 2017; 58: 672–675. [PMC free article] [PubMed] [Google Scholar]
9. Arreo Del Val V, Avila-Alvarez A, Schteffer LR, et al. Alveolar capillary dysplasia with misalignment of the pulmonary veins associated with aortic coarctation and intestinal malrotation. J Perinatol 2014; 34: 795–797. [PubMed] [Google Scholar]
10. Miranda J, Rocha G, Soares H, et al. Alveolar capillary dysplasia with misalignment of pulmonary veins (ACD/MPV): a case series. Case Rep Crit Care 2013; 2013: 327250. [PMC free article] [PubMed] [Google Scholar]
11. Rabah R, Poulik JM. Congenital alveolar capillary dysplasia with misalignment of pulmonary veins associated with hypoplastic left heart syndrome. Pediatr Dev Pathol 2001; 4: 167–174. [PubMed] [Google Scholar]
12. Szafranski P, Dharmadhikari AV, Wambach JA, et al. Two deletions overlapping a distant FOXF1 enhancer unravel the role of lncRNA LINC01081 in etiology of alveolar capillary dysplasia with misalignment of pulmonary veins. Am J Med Genet A 2014; 164A: 2013–2019. [PMC free article] [PubMed] [Google Scholar]
13. Garola RE, Thibeault DW. Alveolar capillary dysplasia, with and without misalignment of pulmonary veins: an association of congenital anomalies. Am J Perinatol 1998; 15: 103–107. [PubMed] [Google Scholar]
14. Gerrits LC, De Mol AC, Bulten J, et al. Omphalocele and alveolar capillary dysplasia: a new association. Pediatr Crit Care Med 2010; 11: e36–37. [PubMed] [Google Scholar]
15. Antao B, Samuel M, Kiely E, et al. Congenital alveolar capillary dysplasia and associated gastrointestinal anomalies. Fetal Pediatr Pathol 2006; 25: 137–145. [PubMed] [Google Scholar]
16. Prothro SL, Plosa E, Markham M, et al. Prenatal diagnosis of alveolar capillary dysplasia with misalignment of pulmonary veins. J Pediatr 2016; 170: 317–318. [PMC free article] [PubMed] [Google Scholar]
17. Abdallah HI, Karmazin N, Marks LA. Late presentation of misalignment of lung vessels with alveolar capillary dysplasia. Crit Care Med 1993; 21: 628–630. [PubMed] [Google Scholar]
18. Ahmed S, Ackerman V, Faught P, et al. Profound hypoxemia and pulmonary hypertension in a 7-month-old infant: late presentation of alveolar capillary dysplasia. Pediatr Crit Care Med 2008; 9: e43–46. [PubMed] [Google Scholar]
19. Boggs S, Harris MC, Hoffman DJ, et al. Misalignment of pulmonary veins with alveolar capillary dysplasia: affected siblings and variable phenotypic expression. J Pediatr 1994; 124: 125–128. [PubMed] [Google Scholar]
20. Goel D, Oei JL, Lui K, et al. Antenatal gastrointestinal anomalies in neonates subsequently found to have alveolar capillary dysplasia. Clin Case Rep 2017; 5: 559–566. [PMC free article] [PubMed] [Google Scholar]
21. Ito Y, Akimoto T, Cho K, et al. A late presenter and long-term survivor of alveolar capillary dysplasia with misalignment of the pulmonary veins. Eur J Pediatr 2015; 174: 1123–1126. [PubMed] [Google Scholar]
22. Kodama Y, Tao K, Ishida F, et al. Long survival of congenital alveolar capillary dysplasia patient with NO inhalation and epoprostenol: effect of sildenafil, beraprost and bosentan. Pediatr Int 2012; 54: 923–926. [PubMed] [Google Scholar]
23. Shankar V, Haque A, Johnson J, Pietsch J. Late presentation of alveolar capillary dysplasia in an infant. Pediatr Crit Care Med 2006; 7: 177–179. [PubMed] [Google Scholar]
24. Oldenburg J, Van Der Pal HJ, Schrevel LS, et al. Misalignment of lung vessels and alveolar capillary dysplasia. Histopathology 1995; 27: 192–194. [PubMed] [Google Scholar]
25. Janney CG, Askin FB, Kuhn C., 3rd Congenital alveolar capillary dysplasia–an unusual cause of respiratory distress in the newborn. Am J Clin Pathol 1981; 76: 722–727. [Google Scholar]
26. Central Bureau for Statistics. (2018, June 5). Statistics Netherlands: Birth; key figures. Retrieved from http://statline.cbs.nl/Statweb/publication/?DM=SLEN&PA=37422eng&D1=0,3-27,32-39,48-51,53-54&D2=0,5,10,15,20,25,30,35,43-66&LA=EN&HDR=T&STB=G1&VW=T.
27. Inwald D, Brown K, Gensini F, et al. Open lung biopsy in neonatal and paediatric patients referred for extracorporeal membrane oxygenation (ECMO). Thorax 2004; 59: 328–333. [PMC free article] [PubMed] [Google Scholar]
28. Houmes RJ, Ten Kate CA, Wildschut ED, et al. Risk and relevance of open lung biopsy in pediatric ECMO patients: the Dutch experience. J Pediatr Surg 2017; 52: 405–409. [PubMed] [Google Scholar]
29. Ng PC, Lewindon PJ, Siu YK, et al. Congenital misalignment of pulmonary vessels: an unusual syndrome associated with PPHN. Acta Paediatr 1995; 84: 349–353. [PubMed] [Google Scholar]
30. Zackrisson S, van de Ven S, Gambhir SS. Light in and sound out: emerging translational strategies for photoacoustic imaging. Cancer Res 2014; 74: 979–1004. [PMC free article] [PubMed] [Google Scholar]
31. Parris T, Nik AM, Kotecha S, et al. Inversion upstream of FOXF1 in a case of lethal alveolar capillary dysplasia with misalignment of pulmonary veins. Am J Med Genet A 2013; 161A: 764–770. [PubMed] [Google Scholar]
32. Zirpoli S, Munari AM, Rustico M, et al. Fetal-MRI prenatal diagnosis of severe bilateral lung hypoplasia: alveolar capillary dysplasia case report. J Prenat Med 2016; 10: 15–19. [PMC free article] [PubMed] [Google Scholar]
33. Cullinane C, Cox PN, Silver MM. Persistent pulmonary hypertension of the newborn due to alveolar capillary dysplasia. Pediatr Pathol 1992; 12: 499–514. [PubMed] [Google Scholar]
34. Sirkin W, O’Hare BP, Cox PN, et al. Alveolar capillary dysplasia: lung biopsy diagnosis, nitric oxide responsiveness, and bronchial generation count. Pediatr Pathol Lab Med 1997; 17: 125–132. [PubMed] [Google Scholar]
35. Haraida S, Lochbuhler H, Heger A, et al. Congenital alveolar capillary dysplasia: rare cause of persistent pulmonary hypertension. Pediatr Pathol Lab Med 1997; 17: 959–975. [PubMed] [Google Scholar]
36. Towe CT, White FV, Grady RM, et al. Infants with atypical presentations of alveolar capillary dysplasia with misalignment of the pulmonary veins who underwent bilateral lung transplantation. J Pediatr 2018; 194: 158–164. [PMC free article] [PubMed] [Google Scholar]
37. Castilla-Fernandez Y, Copons-Fernandez C, Jordan-Lucas R, et al. Alveolar capillary dysplasia with misalignment of pulmonary [corrected] veins: concordance between pathological and molecular diagnosis. J Perinatol 2013; 33: 401–403. [PubMed] [Google Scholar]
38. Steinhorn RH, Cox PN, Fineman JR, et al. Inhaled nitric oxide enhances oxygenation but not survival in infants with alveolar capillary dysplasia. J Pediatr 1997; 130: 417–422. [PubMed] [Google Scholar]
39. Eldridge WB, Zhang Q, Faro A, et al. Outcomes of lung transplantation for infants and children with genetic disorders of surfactant metabolism. J Pediatr 2017; 184: 157–164. [PMC free article] [PubMed] [Google Scholar]
40. Boston US, Fehr J, Gazit AZ, et al. Paracorporeal lung assist device: an innovative surgical strategy for bridging to lung transplant in an infant with severe pulmonary hypertension caused by alveolar capillary dysplasia. J Thorac Cardiovasc Surg 2013; 146: e42–43. [PubMed] [Google Scholar]
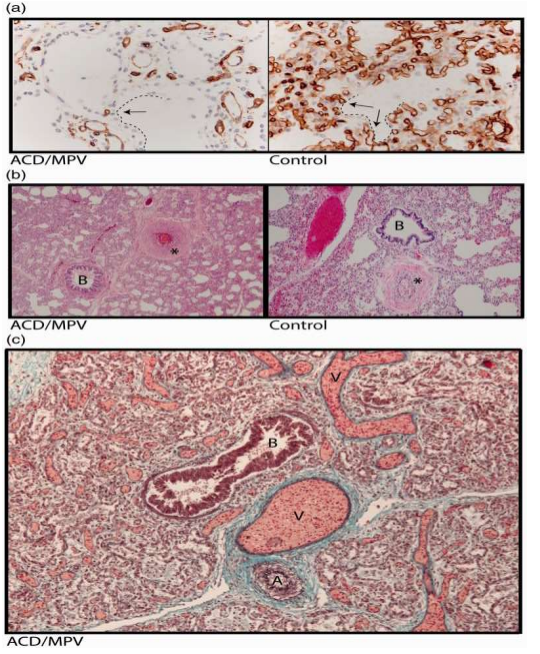
Figure 1
