Adjunctive Viral Load Assessments of Torque Teno Virus may Enhance Clinical Interpretation of Plasma Donor-derived Cell-free DNA after Lung Transplantation
Adjunctive Viral Load Assessments of Torque Teno Virus may Enhance Clinical Interpretation of Plasma Donor-derived Cell-free DNA after Lung Transplantation
Justin P. Rosenheck1, DO; Anastasia Butskova2, MS, Sangeeta M. Bhorade2, MD; Brian C. Keller3 MD, PhD; David J. Ross2, MD
1.The Ohio State University, Division of Pulmonary & Critical Care Medicine, 241 W 11th Ave Suite 5000, Columbus, OH 43201, USA.
2. Natera, Inc., Austin, TX , 78753, USA.
3.Massachusetts General Hospital, Harvard School of Medicine, Division of Pulmonary & Critical Care Medicine, 55 Fruit Street, Boston, MA, 02114, USA.
*Correspondence to: David J Ross, MD. Senior Medical Director, Lung Transplantation / Molecular Diagnostics, Natera, Inc., 13011A McCallen Pass, Austin, TX , U.S. 78753.
Copyright
© 2024 David J Ross. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 26 Sep 2024
Published: 04 Oct 2024
DOI: https://doi.org/10.5281/zenodo.1388899
Abstract:
Torque Teno Virus (TTV) viral load (VL) is affected by the net state of immunosuppression after lung transplantation (LT). We speculated TTV VL assessment could complement surveillance with donor-derived cell-free DNA (dd-cfDNA), a validated biomarker for acute rejection (AR). 132 plasma samples were analyzed for both TTV VL and dd-cfDNA%. Median dd-cfDNA% was elevated in biopsy proven AR, chronic lung allograft dysfunction, and infection cohorts (p<0.01, 0.03, 0.10, respectively) compared to stable. TTV loads were significantly lower in patients with higher dd-cfDNA (>1% vs. <1%) in both the <3-month (p=0.013) and >3-month periods (p=0.028). TTV loads were significantly lower in the AR cohort compared with non-AR and stable cohorts in the <3- month timeframe. This data suggests that TTV VL assessment, as an adjunct to routine %dd-cfDNA surveillance after LT, may have utility for distinction of AR, and potentially infection, from stable patients during the initial 3-month period post-LT. If substantiated in multi-center clinical trials, TTV VL may have clinical value as a proxy for net state of immunosuppression and allow for a personalized medicine approach to immunosuppressive drug adaption.
Key Words:Torque Teno virus, Anello Virus, cell-free DNA, lung transplantation, acute rejection.
Adjunctive Viral Load Assessments of Torque Teno Virus may Enhance Clinical Interpretation of Plasma Donor-derived Cell-free DNA after Lung Transplantation
Abbreviations:
ALAD: Acute Lung Allograft Dysfunction
AR: Acute Rejection
BAL: Bronchoalveolar Lavage
BOS: Bronchiolitis Obliterans Syndrome
CLAD: Chronic Lung Allograft Dysfunction
CNI: Calcineurin Inhibitor
COPD: Chronic Obstructive Pulmonary Disease
dd-cfDNA: donor-derived cell-free DNA
ILD: Interstitial Lung Disease
INFXN: Allograft Infection
IS: Immunosuppression
LT: Lung Transplant
NPV: Negative Predictive Value
NRAD: Neutrophilic-Responsive Allograft Dysfunction
PAH: Pulmonary Arterial Hypertension
PGD: Primary Graft Dysfunction
TBBx: Transbronchial Biopsy
TTV: Torque Teno Virus
Introduction
Lung transplant (LT) recipients are closely surveilled to detect, prevent, and mitigate the spectrum of etiologies of “allograft injury”. Transplant providers struggle to maintain adequate immunosuppression (IS) to avert acute rejection (AR) while balancing against the pitfalls of infection and cancer that can arise from a weakened immune system. “Standard of practice” (SOP) currently includes monitoring of calcineurin inhibitor (CNI) drug levels and assessment of patient clinical parameters to drive adjustments to a multi-drug IS regimen. This practice is imprecise since IS is effectuated by a protocolized regimen, and pharmacokinetics and pharmacogenetics are heterogeneously manifested across a population.[1] To this end, attempts at leveraging in vitro assays of immune function for titration of IS have met with varying levels of success and minimal clinical adoption.[2, 3]
Torque Teno Virus (TTV) has gained recent attention as a potential biomarker of the “net state of IS”. TTV is a ubiquitous, nonpathogenic DNA virus detected in over 90% of immunocompromised patients. TTV viral load (VL) correlates with the robustness of the adaptive and innate immunologic response, [4] and inverse correlations between VL and risks of acute and chronic allograft rejection have been observed. [4-6] Furthermore, changes in TTV over time have been observed within the lung allograft perioperatively, and may be modulated in primary graft dysfunction (PGD). [7] TTV levels ultimately achieve “steady state” only after several months post-LT due to the prolonged effects of induction and maintenance immunosuppression. [4]
Donor-derived cell-free DNA (dd-cfDNA) is a clinically validated non-invasive plasma biomarker for detecting acute rejection or infection, termed acute lung allograft dysfunction (ALAD). [8, 9] Limitations of dd-cfDNA relate to its lack of specificity in differentiating various types of allograft injury, encompassing infectious or immunologic insults.
We hypothesized that TTV VL may be associated with rejection risk, and that quantitative assessment of TTV VL as an adjunct to routine plasma dd-cfDNA surveillance could enhance the ability of dd-cfDNA to reliably detect lung allograft injury.
Methods
This case-control study was approved by the Ohio State University Institutional Review Board (OSU #2020H0445). Informed patient consent was obtained for participation. Plasma samples were obtained from the Ohio State University Transplant Biorepository at the time of SOP flexible bronchoscopy with bronchoalveolar lavage (BAL) and transbronchial biopsy (TBBx). Cohort selection and adjudication into clinical cohorts was described previously.[8] In short, blinded investigators adjudicated samples to pre-specified clinical-pathologic cohorts based on review of spirometry, chest imaging, BAL microbiology, and TBBx histopathologic results. Isolated ISHLT Grade B histology was considered a nonspecific finding unless present along with perivascular mononuclear infiltration (i.e. Grade A1-4) and excluded from further analysis. [10] BAL microbiology analyses included SOP special stains, bacterial and fungal cultures, and multiplex PCR (BioFire 2.1 Respiratory Panel™, Qiagen, Inc). Deidentified plasma samples were analyzed using the Prospera™ Lung test (Natera, Inc., San Carlos, CA) as described previously. [8]
Metagenomic TTV and NGS Quantitation of “Viral Load”
DNA was extracted from plasma with EZ1 Virus Mini Kit v2 (Qiagen, Ltd.). Positive and negative controls were added to all samples before extraction. DNA sequencing libraries were prepared according to the manufacturer's protocols, using an investigational preparation kit and protocol (Cantata Bio, LLC). Final sequencing library preparations were quantified, and quality checked and sequenced on the NextSeq 500 sequencer (Illumina, Inc.). Demultiplexing was performed using bcl2fastq 2.20 with default parameters and no lane splitting. The resulting FASTQ files were uploaded and analyzed using an investigational cloud-based software (Cantata Bio, LLC). TTV quantification (Log10 Score) was normalized for read counts across libraries and based on the spiked-in internal controls, correcting for differences in genome lengths, host background, and technical bias.
Clinical Protocol
TTV assessment was not obtained prior to initiation of induction or maintenance IS post-LT. All patients received a 2-dose regimen of basiliximab and high dosage methylprednisolone as induction, while maintenance IS included a corticosteroid, tacrolimus, and mycophenolate mofetil. Patient demographics and cohort assignments are presented in Table 1.
Table 1: Sample Clinical-Pathologic Cohort Assignments, Lung Transplant Patient Demographics
|
Cohort Assignments |
Total Acute Rejection Allograft Infection CLAD Stable Excluded |
132 samples (76 patients) 22 (ACR=18, AMR=4) 12 14 69 16 (“nonspecific histology”) |
|
Sex |
M F |
45 (59%) 31 (41%) |
|
Age |
61.0 years (53.5-67.0) |
|
|
Type of LT Procedure |
Double Single |
62 (82%) 14 (18%) |
|
Native Lung Diseases |
ILD COPD PAH BOS SARS-CoV2 Bronchiectasis |
35 (46%) 25 (33%) 3 (4%) 10 (13%) 1 (1%) 2 (3%) |
ILD: Interstitial Lung Disease, COPD: Chronic Obstructive Pulmonary Disease, PAH: Pulmonary Arterial Hypertension, BOS: Bronchiolitis Obliterans Syndrome
Statistical Calculations
P values were calculated using the Mann-Whitney test, with adjustments made for patients who were included in the dataset multiple times. This adjustment involved a procedure of iteratively and randomly selecting one sample per patient from all patients and performing the Mann- Whitney test for each iteration. The reported P values represent the median P values obtained from these iterations.
Results
132 plasma samples from 76 patients with detectable TTV levels were analyzed for dd-cfDNA%. Compared to a stable cohort, median dd-cfDNA% levels were significantly elevated in acute rejection (cellular or antibody mediated) and CLAD cohorts (p<0.01 and 0.03, respectively) and elevated in the allograft infection cohort (p= 0.10). TTV VL increased with “time from transplant”, peaking at approximately 3 months as depicted in Figure 1. When the clinical-pathologic cohorts were further classified (Figure 2a) by “High Risk for Rejection or Injury” (dd-cfDNA>1.0%), TTV VL was significantly lower in samples with high vs. low dd-cfDNA (>1% vs. <1%), in the <3 month (p=0.013) and > 3-month epoch (p=0.028) post-LT. TTV VL was significantly lower in the AR cohort compared with the combined non-AR cohort (i.e., infection, CLAD and stable) and stable cohort alone, but only in the <3-month timeframe. TTV levels did not discriminate AR from the combined non-AR or Stable cohorts beyond 3 months post-LT (Figure 2b). Intriguing but limited by inadequate sample size was an observed trend for higher TTV VL in the infection cohort compared to AR in the <3-month timeframe.
Figure 1: Individual patient Torque Teno Virus (TTV) Viral Load (Log10) on Y-axis versus Days after Lung Transplantation on X-axis, across all Diagnostic Cohorts-acute rejection (AR) encompassing cellular and antibody-mediated rejection, chronic lung allograft dysfunction or neutrophilic- responsive allograft dysfunction (CLAD/NRAD), allograft infection (INFXN), and stable. The blue curve represents the locally weighted smoothing line (LOWESS) with 95% confidence shading.
Figure 2a: Association of Torque Teno Virus (TTV) Viral Loads (Log10) on Y-axis with dd-cfDNA% classifier as “High Risk” (>1% dd-cfDNA) versus “Low Risk” (<1% dd-cfDNA) for the following cohorts: acute rejection (AR) (encompassing cellular and antibody-mediated rejection), chronic lung allograft dysfunction or neutrophilic-responsive allograft dysfunction (CLAD/NRAD), allograft infection (INFXN), and stable.
Figure 2b: Clinical-Pathologic Cohorts analyzed by Acute Rejection (combined cellular and antibody-mediated) versus the combined non-acute rejection categories (CLAD/NRAD, INFXN and STABLE). Data is presented for periods < 3-months and >3-months post- lung transplant.
Discussion
dd-cfDNA is a clinically validated biomarker with high negative predictive value (NPV) for acute rejection increasingly being adopted widely in the U.S. for surveillance of lung allograft status. However, since dd-cfDNA% reflects increased cellular apoptosis and turnover, the test cannot accurately discriminate amongst diverse etiologies of organ injury. In contrast, TTV viral load does not inform of the allograft status per se, rather it serves as a proxy for the net state of IS. Elevation in TTV viral load (VL) has been associated with excess IS and thus increased likelihood of infection. TTV can function as a potential differentiator for injury type in patients with elevated dd-cfDNA. [4, 5] Moreover, the Vienna lung transplant group reported an increase in steady-state TTV VL after the initial 3 months, predictive of infectious complications.[11] This same group was also the first to describe, in a case-control analysis, an association between lower TTV VL and chronic lung allograft dysfunction (CLAD).[12] Recently, TTV VL had been implemented as an indicator of net state of IS, during analysis of efficacy of SARS-CoV-2 mRNA1273 vaccination for lung transplant recipients. A lower TTV VL was associated with higher anti-spike specific humoral response and frequency of protective immunologic status after vaccination. Further, TTV VL was confirmed as “time-dependent” with lower TTV VL in association with a longer time after transplantation and ensuant the reduction in immunosuppressive drug treatment. [13]
To our knowledge, this is the first study to demonstrate an inverse association between dd-cfDNA% and plasma TTV VL. This intriguing finding is suggestive of a potential causal relationship between reduced net state of immunosuppression and molecular cellular injury involving the allograft, in either the presence or absence of biopsy-proven AR. Further, since biopsy with routine histopathology can be insensitive to cellular or antibody-mediated rejection [14][15], future clinical trials may develop algorithms for treatment when dd-cfDNA% is elevated in association with a low TTV VL in attempt to preserve organ health.[14] The NIH (GRAfT consortium) recently analyzed long-term clinical outcomes, and “extreme molecular injury” (EMI) as defined by a significant elevation in dd-cfDNA (>5.0%) after lung transplantation. Regardless of whether EMI resulted from confirmed AR, infection events or was of unknown etiology (i.e. “primary”), the hazard ratio was approximately 2.78 for later development of severe CLAD or mortality.[16]. From our analysis, TTV VL can potentially complement plasma %dd-cfDNA assessment to distinguish AR from stable and infection cohorts during the initial 3-month period post-LT.
Limitations of this study include its small size, cross-sectional design, and monocentric approach. Nevertheless, future multicenter clinical studies with longitudinal assessments of the impact of quantitative TTV VL would appear warranted. To this end, and underpinning the potential for serious adverse sequelae of excess IS, the VIGILung study (ClinTrials.gov identifier: NCT04198506) is a European two-center randomized-controlled study currently in progress which aims to assess safety and preliminary efficacy of IS as guided by TTV VL assessments in concert with conventional IS drug monitoring and dosage adaption. The study is anticipated to conclude at the end of year 2025 with a primary endpoint of estimated and cystatin glomerular filtration rate (GFR) and numerous secondary endpoints which examine acute allograft rejection and infection rates, and also measures of cellular and humoral immunologic function.[17] The promise of TTV VL to allow for a personalized medicine approach to LT patient care may yet be realized, with data from appropriately designed and powered clinical trials poised to give clinicians the ability to optimally balance the risk of allograft rejection and the counterpoised risks of infection and cancer.
Authors’ disclosures and acknowledgments:
AB, SB, and DJR are employees of Natera, Inc with the option to own stock. JPR has received research funding from Natera, Inc. BCK has received research funding from Natera, Inc. and CareDX, is a consultant and member of Scientific Advisory boards of CareDx and Zambon, and has received speaker fees from WebMD.
References
[1] H. Abboudi and I. A. Macphee, "Individualized immunosuppression in transplant patients: potential role of pharmacogenetics," Pharmgenomics Pers Med, vol. 5, pp. 63-72, 2012, doi:
10.2147/PGPM.S21743.
[2] M. Mian et al., "Evaluation of a Novel Global Immunity Assay to Predict Infection in Organ Transplant Recipients," (in eng), Clin Infect Dis, vol. 66, no. 9, pp. 1392-1397, Apr 17 2018, doi: 10.1093/cid/cix1008.
[3] M. Ravaioli et al., "Immunosuppression Modifications Based on an Immune Response Assay: Results of a Randomized, Controlled Trial," (in eng), Transplantation, vol. 99, no. 8, pp. 1625-32, Aug 2015, doi: 10.1097/tp.0000000000000650.
[4] B. C. Frye et al., "Kinetics of Torque Teno Virus-DNA Plasma Load Predict Rejection in Lung Transplant Recipients," (in eng), Transplantation, vol. 103, no. 4, pp. 815-822, Apr 2019, doi: 10.1097/tp.0000000000002436.
[5] P. Jaksch et al., "Torque Teno Virus as a Novel Biomarker Targeting the Efficacy of Immunosuppression After Lung Transplantation," (in eng), J Infect Dis, vol. 218, no. 12, pp. 1922-1928, Nov 5 2018, doi: 10.1093/infdis/jiy452.
[6] I. Görzer, P. Jaksch, R. Strassl, W. Klepetko, and E. Puchhammer-Stöckl, "Association between plasma Torque teno virus level and chronic lung allograft dysfunction after lung transplantation," The Journal of Heart and Lung Transplantation, vol. 36, no. 3, pp. 366-368, 2017, doi: 10.1016/j.healun.2016.10.011.
[7] A. A. Abbas et al., "The Perioperative Lung Transplant Virome: Torque Teno Viruses Are Elevated in Donor Lungs and Show Divergent Dynamics in Primary Graft Dysfunction," (in eng), Am J Transplant, vol. 17, no. 5, pp. 1313-1324, May 2017, doi: 10.1111/ajt.14076.
[8] J. P. Rosenheck et al., "Clinical Validation of a Plasma Donor-derived Cell-free DNA Assay to Detect Allograft Rejection and Injury in Lung Transplant," Transplantation Direct, vol. 8, no. 4, p. e1317, 2022, doi: 10.1097/txd.0000000000001317.
[9] M. Keller et al., "Donor-derived cell-free DNA as a composite marker of acute lung allograft dysfunction in clinical care," J Heart Lung Transplant, vol. 41, no. 4, pp. 458-466, Apr 2022, doi: 10.1016/j.healun.2021.12.009.
[10] S. Stewart et al., "Revision of the 1996 working formulation for the standardization of nomenclature in the diagnosis of lung rejection," J Heart Lung Transplant, vol. 26, no. 12, pp. 1229-42, Dec 2007, doi: 10.1016/j.healun.2007.10.017.
[11] P. Jaksch et al., "Torque teno virus as a novel biomarker targeting the efficacy of immunosuppression after lung transplantation," The Journal of infectious diseases, vol. 218, no. 12, pp. 1922-1928, 2018.
[12] I. Görzer, P. Jaksch, R. Strassl, W. Klepetko, and E. Puchhammer-Stöckl, "Association between plasma Torque teno virus level and chronic lung allograft dysfunction after lung transplantation," (in
eng), J Heart Lung Transplant, vol. 36, no. 3, pp. 366-368, Mar 2017, doi: 10.1016/j.healun.2016.10.011.
[13] R. A. S. Hoek et al., "High torque tenovirus (TTV) load before first vaccine dose is associated with poor serological response to COVID-19 vaccination in lung transplant recipients," The Journal of Heart and Lung Transplantation, vol. 41, no. 6, pp. 765-772, 2022, doi: 10.1016/j.healun.2022.03.006.
[14] S. M. Arcasoy et al., "Pathologic Interpretation of Transbronchial Biopsy for Acute Rejection of Lung Allograft Is Highly Variable," American Journal of Transplantation, vol. 11, no. 2, pp. 320-328, 2011, doi: https://doi.org/10.1111/j.1600-6143.2010.03382.x.
[15] S. M. Bhorade et al., "Interobserver variability in grading transbronchial lung biopsy specimens after lung transplantation," Chest, vol. 143, no. 6, pp. 1717-1724, Jun 2013, doi: 10.1378/chest.12-2107.
[16] M. B. Keller et al., "Extreme elevations of donor-derived cell-free DNA increases the risk of chronic lung allograft dysfunction and death, even without clinical manifestations of disease," The Journal of Heart and Lung Transplantation, 2024/05/03/ 2024, doi: https://doi.org/10.1016/j.healun.2024.04.064.
[17] J. Gottlieb et al., "Viral load-guided immunosuppression after lung transplantation (VIGILung)— study protocol for a randomized controlled trial," Trials, vol. 22, pp. 1-10, 2021.
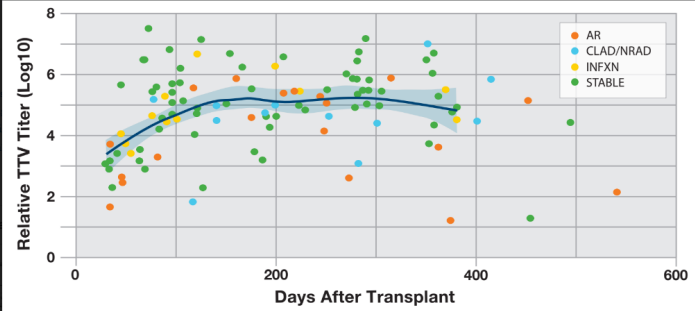
Figure 1
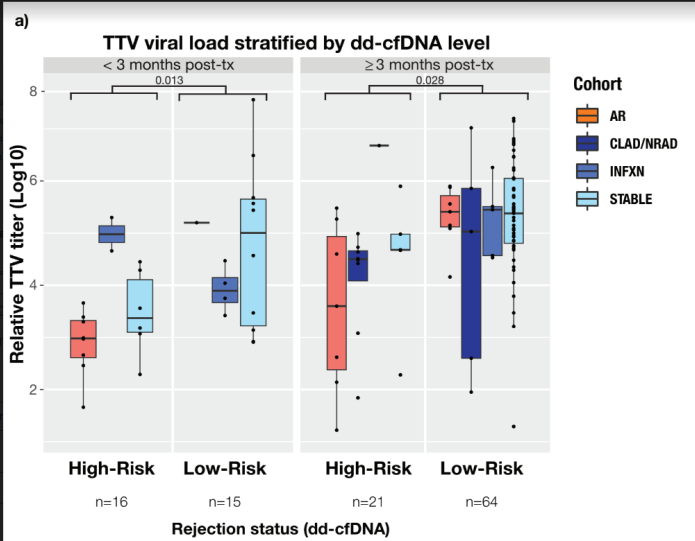
Figure 2
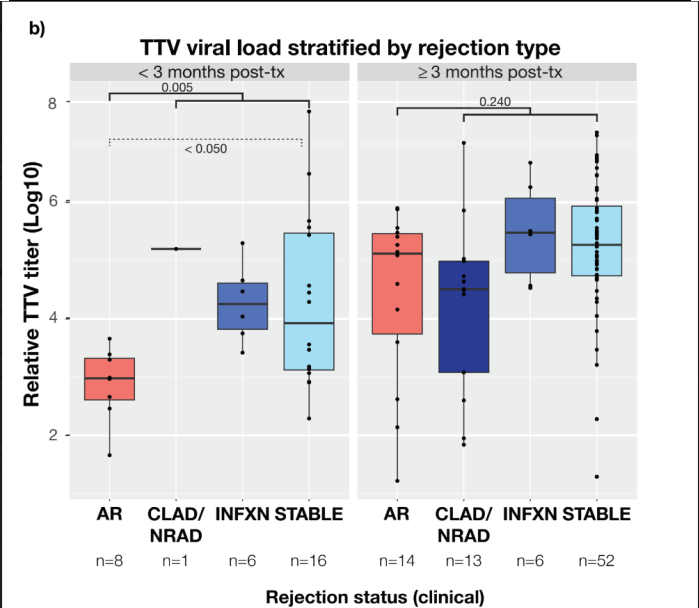
Figure 3
