Pulmonary Arterial Hypertension Complicated with COVID-19
Pulmonary Arterial Hypertension Complicated with COVID-19
Attapon Cheepsattayakorn1,2,3,4*, Ruangrong Cheepsattayakorn5, Porntep Siriwanarangsun2
1. Faculty of Medicine Vajira Hospital, Navamindradhiraj University, Bangkok, Thailand.
2. Faculty of Medicine, Western University, Pathumtani Province, Thailand.
3. 10th Zonal Tuberculosis and Chest Disease Center, Chiang Mai, Thailand.
4. Department of Disease Control, Ministry of Public Health, Thailand.
5. Department of Pathology, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand.
Correspondence to: Attapon Cheepsattayakorn, 10th Zonal Tuberculosis and Chest Disease Center, 143 Sridornchai Road Changklan Muang Chiang Mai 50100 Thailand.
Copyright
© 2024 Attapon Cheepsattayakorn. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 01 Nov 2024
Published: 01 Dec 2024
Pulmonary Arterial Hypertension Complicated with COVID-19
Identification of the respiratory and cardiovascular systems, particularly, pulmonary arterial hypertension (PAH), acute cardiac injury, and venous thromboembolism (VTE) can be identified in long-term effects of COVID-19, but the virus also affects the neurological system, bones, endocrine glands, include acute respiratory distress syndrome (ARDS) [1, 2].
With a novel, and severe novel coronavirus infections, PAH has been reported to complicate the course of illness for 13.4% and 21% of patients, respectively. PAH is a serious complication of new coronavirus infection, increasing the likelihood of requiring mechanical ventilation, extracorporeal membrane oxygenation (ECMO), intensive care unit (ICU) care and even death. Enhancing the long-term prognosis of patients and minimize the hospitalization rate and death due to such complications therefore, by early detecting high pulmonary artery pressure in patients with SARS-CoV-2 [3-6]. As revealed by previous studies and reasons described by the processes of immunological dysfunction, endothelial dysfunction, vascular leakage, and thrombotic microangiopathy that are comparable to those that cause pulmonary vascular disease, such as PAH may be responsible for the effects of SARS-CoV-2 on pulmonary hemodynamics. On the other hand, reports of the study mechanism's depth and specificity are uncommon [3-7].
A recent study in 2024 conducted by Hou et al using a functional enrichment analysis on the GEO database to identify common differentially expressed genes (C-DEGs) across the PAH and COVID-19 datasets. The results of a screening of the key genes using three machine algorithms: LASSO, RF, and SVM-RFE-based were confirmed by the validation queue. The role of prioritized core genes was examined by using the gene set enrichment analysis (GSEA). The regulatory networks including these DEGs, including TF-gene connections and TF-microRNA co-regulation were next mapped out. Molecular docking simulations, drug-protein interaction networks, and molecular dynamics simulations were employed to screen for possible therapeutic medicines. The study findings are expected to offer a novel approach to elucidating the genetic connection between the aforementioned disorders (Figures 1-5) [8].
Additionally, CCL20 and SELE were found to be indicators of PAH and COVID-19 co-pathogenesis by various bioinformatics analyze and machine learning algorithms. Adaptive immune response, leukocyte, lymphocyte mediated immune responses, and proinflammatory response mediated by cytokines like IL-12, TNF-α were functionally enriched in these two hub genes. These two hub genes were selected for nomogram construction and their diagnostic value evaluated by machine learning. The nomogram was found to have high diagnostic value. Dendritic cells had the strongest connection with CCL20 and SELE, followed by activated CD4 T cells, active dendritic cells, natural killer T cells, neutrophils, and plasmacytoid dendritic cells. Using only 2 reference genes, they were able to isolate 12 shared TFs and 25 shared TF-miRNAs.by FFL tool, This FFL among CCL20, miR-1256 and PPARG may be a novel regulatory module in PAH complicated with COVID-19. It was hypothesized that AFLATOXIN B1, 1-NITROPYRENR, and FENRETINIDE would be useful in treating PAH complicated with COVID-19. Further molecular dynamics and molecular docking simulations demonstrated that 1-nitropyrene had the most stable binding with CCL20 and SELE (Figures 1-5) [8].
In conclusion, by understanding the comorbidity of PAH and COVID-19 may be assisted by these angiogenesis and biomarkers connection between PAH and COVID-19.
Figure 1 Demonstrating identification and functional enrichment analysis of common DEGs. (A) Venn diagram revealing 47 Up-regulated common DEGs in PAH and COVID-19. (B) Venn diagram revealing 17 Down-regulated common DEGs in PAH and COVID-19. (C) KEGG pathway analysis was performed on common DEGs. (D) GO-BP terms of common genes. (E) GO-CC terms of common genes. (F) GO-MF terms of common genes. DEG Differentially expressed genes. COVID coronavirus disease. PAH pulmonary hypertension. KEGG Kyoto Encyclopedia of Genes and Genomes. GO-BP, GO-CC, GO-MF Gene Ontology terms in biological process, cellular component, and molecular function [8].
Figure 2 Demonstrating the selection of candidate diagnostic biomarkers of COVID-19 progression with machine learning approaches. (A,B) LASSO regression analysis was applied to screen diagnostic biomarkers. (C) The diagnostic error relating to control,COVID-19 and total groups was visualized from the random forest. (D) The column showing 30 DEGs ranked based on the importance score calculated from the random forest. (E,F) The number of DEGs with the lowest error and highest accuracy after 100 folds were considered the most suitable candidates via the SVM-RFE algorithm. (G) The intersection of 3 machine learning algorithms was obtained with a Venn diagram tool. LASSO least absolute shrinkage and selection operator, SVM-RFE support vector machine recursive feature elimination, DEGs Differentially expressed genes [8].
Figure 3 Demonstrating the association between the hub genes and immune infiltration. (A) In GSE147507cohort, CCL20, SELE were revealed to positively correlate with most cell types. Except for CD56dim natural killer cells, CD56bright natural killer cells, memory B cells, and Type 2 T helper cells. (B) In GSE113439 cohort, CCL20, SELE were shown to positively correlate with many cell types. Including: type 1 T helper cell, regulatory T cell, plasmacytoid dendritic cell, neutrophil, natural killer T cell, mast cell, central memory CD4 T cell, activated dendritic cell, and activated CD4 T cell. Red: positive correlation; Blue: negative correlation. (C) According to a Pearson correlation study, plasmacytoid dendritic cells had the highest level of association with CCL20,while Activated dendritic cells had the highest level of linkage with SELE [8].
Figure 4 Demonstrating the network for TF-gene and TF-miRNA interaction with Common DEGs. (A) They predicted 25 miRNAs and 11 TF-genes interacting with SELE and CCL20 through the Network analyst website. Red nodes: hub genes; blue nodes: miRNA. Green nodes:TF-genes. (B) They showed that among CCL20, miR-1256 and PPARG may be a novel regulatory module in PAH complicated with COVID-19 through the FFL tool [8].
Figure 5 Demonstrating 3D and 2D molecular docking patterns for (A) FENRETINIDE, (B) 1-NITROPYRENE, (C) AFLATOXIN B1 with CCL20 respectively. (D) FENRETINIDE, (E) 1-NITROPYRENE, (F) AFLATOXIN B1 with SELE, respectively [8].
References
1. The Lancet. Understanding long COVID: A modern medical challenge. Lancet. 398, 725.
DOI : https://doi.org/10.1016/S0140-6736(21)01900-0 (2021).
2. Zeng, H. et al. A new coronavirus estimation global score for predicting mortality during hospitalization in patients with COVID-19. Cardiol. Discov. 2, 69–76.
DOI : https://doi.org/10.1097/CD9.0000000000000052 (2022).
3. Kobusiak-Prokopowicz, M. et al. Cardiovascular, pulmonary, and neuropsychiatric short- and long-term complications of COVID-19. Cells. 11, 3882.
DOI : https://doi.org/10.3390/cells11233882 (2022).
4. Castiglione, L. & Droppa, M. Pulmonary hypertension and COVID-19. Hamostaseologie. 42, 230–238.
DOI : https://doi.org/10.1055/a-1661-0240 (2022).
5. Pagnesi, M. et al. Pulmonary hypertension and right ventricular involvement in hospitalised patients with COVID-19. Heart. 106, 1324–1331.
DOI : https://doi.org/10.1136/heartjnl-2020-317355 (2022).
6. Wesley Milks, M. et al. Risk assessment in patients with pulmonary arterial hypertension in the era of COVID 19 pandemic and the telehealth revolution: State of the art review. J. Heart Lung Transplant. 40(3), 172–182.
DOI : https://doi.org/10.1016/j.healun.2020.12.005 (2021).
7. Farha, S. & Heresi, G. A. COVID-19 and pulmonary arterial hypertension: Early data and many questions. Ann. Am. Thorac. Soc. 17, 1528–1530.
DOI : https://doi.org/10.1513/AnnalsATS.202008-1014ED (2020).
8. Hou, Q., Jiang, J., Na, K. et al. Potential therapeutic targets for COVID-19 complicated with pulmonary hypertension: a bioinformatics and early validation study. Sci Rep 14, 9294 (2024).
DOI : https://doi.org/10.1038/s41598-024-60113-7.
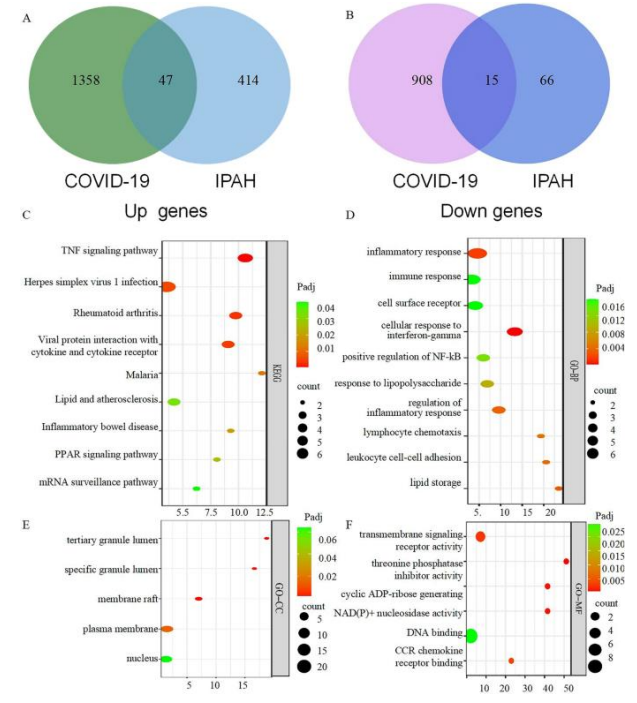
Figure 1
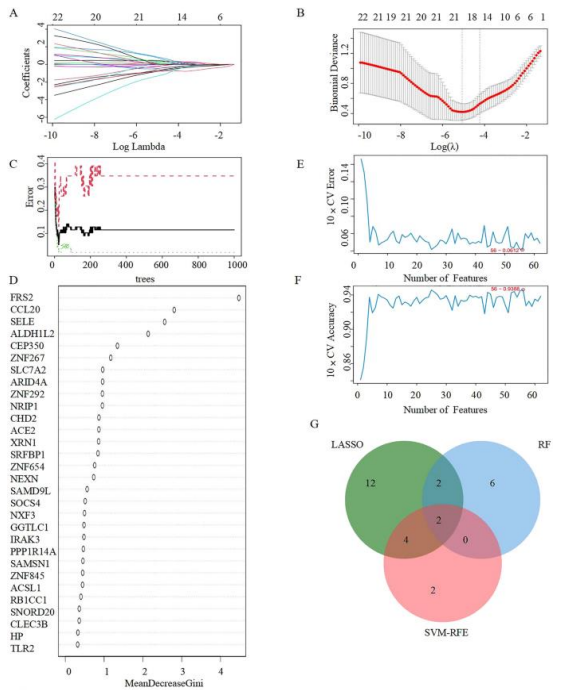
Figure 2
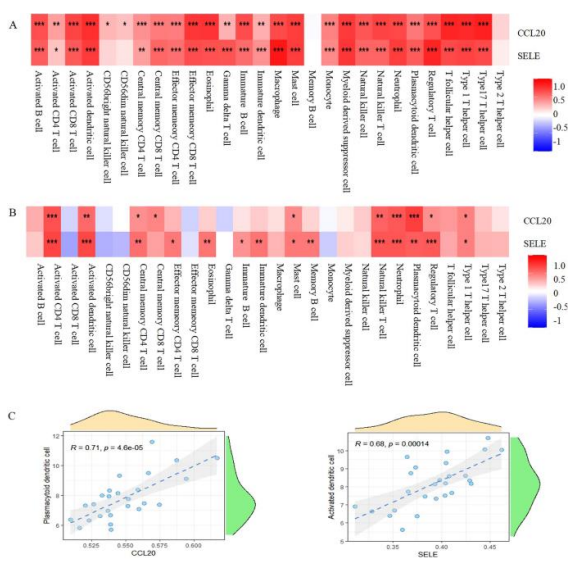
Figure 3
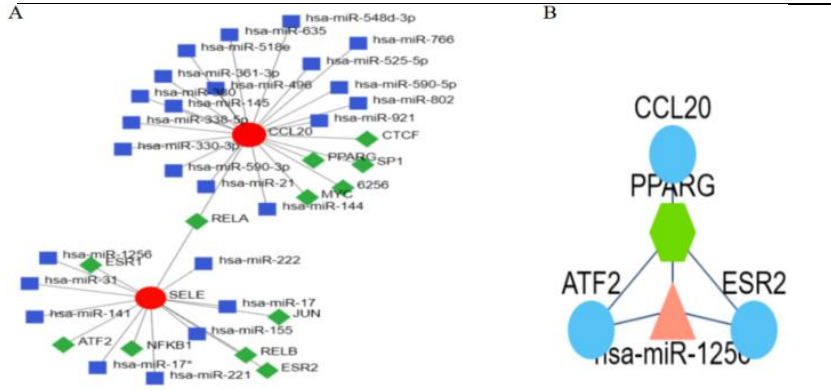
Figure 4
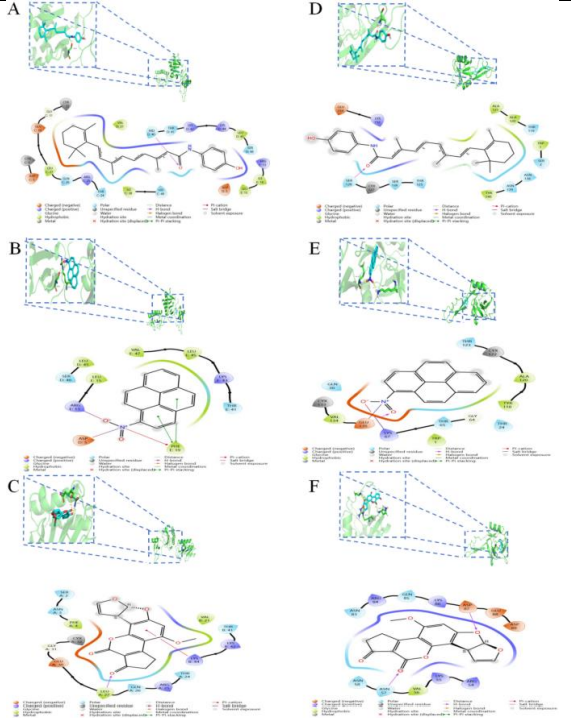
Figure 5
