Pedal Intraosseous Epidermoid Inclusion Cysts, a Case Report and Literature Review.
Pedal Intraosseous Epidermoid Inclusion Cysts, a Case Report and Literature Review.
Mr Anurag Rana,1 Mr Tariq Adam Kwaees,*2, Mr Arun Daniel Joseph3, Dr Lucia Sepesiova4, Mr Samer Morgan5
1. Mr Anurag Rana, MS (Orth); Speciality Doctor in Trauma and Orthopaedic surgery.
2. Mr Tariq Adam Kwaees, MD, MSc, MRCS, FRCS (Tr & Orth); Consultant Trauma and Orthopaedic Surgeon.
3. Mr Arun Daniel Joseph, MBBS, MS (Orth), DNB (Orth), MNAMS, MRCS(Ed). Speciality Doctor in Trauma and Orthopaedic Surgery.
4. Dr Lucia Sepesiova, MD (MUDr). Foundation Year Two Doctor in Trauma and Orthopaedic Surgery.
5. Mr Samer Morgan, MBBch, MRCS, MSc, FRCSEd (Tr & Orth). Consultant Trauma and Orthopaedic Surgeon.
*Correspondence to: Mr Tariq Adam Kwaees, St Helens and Knowsley Hospitals NHS Trust, Whiston Hospital, Prescot, United Kingdom, L35 5DR.
Copyright
© 2025 Mr Tariq Adam Kwaees is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 29 Jan 2025
Published: 06 Feb 2025
DOI: https://doi.org/10.5281/zenodo.14822856
Abstract:
Intraosseous epidermoid inclusion cysts (IO-EIC) rarely affect the foot and may be difficult to diagnose in the setting of concomitant infection. We present the first recorded case involving a metatarsal and an “inside out” mode of tissue violation, along with a review of the literature. A search of the PubMed, Embase and Google Scholar databases was undertaken. Only complete or near complete cases were reviewed. Twelve other cases of pedal IO-EIC were found. The majority of cases involved a phalanx of the hallux, were in the older age group and all had a delay in diagnosis necessitating the need for amputation in some. In the setting of infection, antibiotics were only effective when coupled with operative intervention. Careful clinical examination is critical for timely diagnosis of IO-EIC of the foot. Antibiotics for an infected or ruptured cyst are ineffective in isolation. Early surgical excision is recommended as any delay results in significant tissue loss and amputation.
Pedal Intraosseous Epidermoid Inclusion Cysts, a Case Report and Literature Review.
Introduction
An epidermal inclusion cyst (EIC) is a benign soft tissue aberration rarely affecting bones [1]. Intraosseous EIC (IO-EIC) predominantly involves the phalanges and skull with the exact pathophysiology of such cysts remaining an enigma [2-5]. Pathogenesis theory varies from epidermal cell migration through a violated bony cortex and subsequent seeding into bone, to gradual erosion of bone by an adjacent primary soft tissue cyst [6-8]. Trauma is thought to be the most common initiating factor while congenital aberration is considered to be a less important etiological determinant [2]. Furthermore, IO-EIC poses a diagnostic conundrum as it is clinically and radiologically indistinguishable from infection and other benign and malignant tumors. Histopathology is the only definitive diagnostic modality, meaning management is often delayed [4,6,7]. We present the first case of a metatarsal IO-EIC misdiagnosed as osteomyelitis along with a literature review of all pedal IO-EIC cases.
Case Presentation
A 47-year-old female presented with a five months history of a chronic wound on the lateral aspect of her right fifth (little) toe. This began as a blister which had been drained by her general practitioner (GP), after which she received a course of flucloxacillin, temporarily alleviating her symptoms. After five weeks the lesion evolved into a nonhealing ulcer and osteomyelitis was suspected, for which she was referred to secondary care.
The patient’s past medical history included injectable substance abuse and hepatitis C for which she had received treatment. On presentation, a 10 mm in diameter ulcer was noted over the lateral aspect of the fifth toe. There was a surrounding well demarcated cellulitis but little discharge. The ulcer did not penetrate the dermis, there were no systemic features of infection, pedal neurovascular examination was normal, as were inflammatory markers and vitals. Radiographs taken at presentation demonstrated bony destruction with swelling and some gas in the soft tissues immediately adjacent to the ulcer [figure 1]. She was started on another course of flucloxacillin and an outpatient MRI scan was organized. The wound improved with the antibiotics however, her toe remained swollen and painful.
Figure 1: Radiographs
Selection of radiographs demonstrating gradual and complete destruction of the proximal phalanx and fifth metatarsal head. Initial presentation (a), 15 months later (b), 26 months later (c), 30 months after initial presentation (d).
Due to poor patient compliance, further radiographs and the MRI were performed 15 months after the first presentation [figure 1 - 2]. These showed complete bony destruction of proximal phalanx and metatarsal head with a 2 x 1cm collection in the lateral soft tissue [figure 2]. During this period, her symptoms had continued to fluctuate with the use of multiple antibiotics, including flucloxacillin, ciprofloxacin, metronidazole, co-trimoxazole and clarithromycin. The end result was the development of a discharging sinus, persistent swelling, localised erythema and pain. A month later a contrast MRI was performed to further delineate the lesion, which demonstrated no collection and a sinus tract in keeping with chronic infection [figure 3]. At three years following initial presentation, the symptoms worsened and the patient consented to surgical treatment consisting of amputation of the fifth toe at the level of the distal meta-diaphysis of the fifth metatarsal.
Figure 2: Initial MRI
Select T1 and T2 images from non-contrast MRI at 15 months demonstrating complete bony destruction of most of the proximal phalanx and metatarsal head. Axial (A,B), Sagittal (C,D), Coronal (E,F). Note sinus tract on image F.
Figure 3: Follow up MRI
Select T1 and T2 images from contrast MRI performed one month later demonstrating worsening destruction of 5th metatarsal. Axial (A,B,C), Sagittal (D,E), Coronal (F,G).
Detailed intraoperative examination of the fourth web space demonstrated a small tuft of hair which was assumed to be a normal variation. Thereafter, copious amounts of hair was found around and integrated within the pericapsular tissue of the metatarsophalangeal joint, including ligaments and adjacent tendons [Figure 4 - 5]. This hair traversed the skin of the webspace and created the tuft of innocuous looking hair within the fourth web. On opening the capsule, a large amount of pus was evacuated, and the hairs were found to be emerging from the head of the fifth metatarsal indicating an “inside-out” violation of the skin. An amputation at the level planned was undertaken, followed by a thorough debridement [figure 6]. Myodesis was undertaken and the wound was closed primarily. Results of microbial and histological analysis demonstrated a benign, ruptured squamous epithelium lined cyst within the metatarsal head, with an adjacent abscess extending into the bone. Fibrosis, hemosiderin deposition and foreign body giant cell reaction to hair fragments in the surrounding tissue were noted. Staphylococcus aureus, Coagulase-negative Staphylococcus, and mixed anaerobes were cultured, for which the patient received five days of ciprofloxacin and metronidazole. The patient was discharged at six weeks postoperatively without complications.
Figure 4: Gross Findings
Surgical field following the first stage of debridement.
Figure 5: Detailed view
Magnified view of the affected area following disarticulation and hemi-section of metatarsal head. Copious amounts of hair (H) was discovered within the metatarsal (M, lower arrow) and integrated within the periarticular connective tissue (upper arrow).
Figure 6: View at closure.
Surgical field prior to final wash and closure, demonstrating removal of all infected and nonviable material.
Discussion
EIC are lesions consisting of true cysts, within a confined space, lined with mature stratified keratinized squamous epithelium resembling that of the epidermis [3]. These cysts can be further subclassified depending on their contents and histological makeup. Dermoid cysts such as in our case, are most common around the cranial vault and contain true skin organelles including hair and cutaneous glands in addition to keratin debris [9]. Contained cysts may remain asymptomatic until rupture, which leads to seeding of adjacent tissues with its contents and a resultant inflammatory reaction. Chronic granulomatous change in response to the keratin “foreign body” is common on histology [10]. Clinically EIC results in swelling, pain and erythema, features of inflammatory change common to a variety of conditions leading to delays in diagnosis [8-19],[Table 1]. Histological analysis is the only method of definitive diagnosis and should be performed by an experienced histopathologist given the wide differentials. Complete surgical excision remains the gold standard for symptomatic cysts, but this is frequently delayed with the diagnosis. The use of antibiotics is a continuous issue. Historically, some authors isolated skin commensals in inflamed cysts, indicating the need for antibiotic use; however, later studies found microbiological colonization was similar in inflamed and non-inflamed cases and recommended the avoidance of antimicrobials [11, 12]. The use of anti-inflammatory medication may also be helpful for symptom management. C5, an anaphylatoxin produced by both uninflamed cysts and normal skin commensals, is the initiator of inflammation in EIC. Its identification encouraged the use of anti-inflammatory medication and intra-cystic steroid injections but results are varied [10].
|
Condition |
History & Clinical features |
Radiographs |
Higher imaging |
Histology |
Treatment |
|
EIC-IO [8] |
Localised inflammatory changes (swelling, erythema, pain) at the site. |
Diaphyseal / meta-diaphyseal, well circumscribed, lytic, sclerotic margin +/- soft tissue swelling. |
CT: well defined radiolucent lesion +/- soft tissue swelling MRI: similar features as in CT finding. |
Macro: true cystic mass with osseous material. Micro: benign; stratified squamous epithelium + cutaneous debris +/- skin appendages |
Curettage +/- bone grafting. |
|
Glomus tumour [15] |
Subungual swelling and bluish discolouration, nail plate deformity. Classic triad of pain, tenderness and temperature sensitivity. |
Osseous erosion, cortical thinning. |
CT: Pressure erosions of the bone. MRI: low T1 and high T2 signal. T1(Gd) shows contrast enhancement indicating high vascularity USG: subuncal hypoechoic nodular lesion, hypervascularity may be seen in Doppler |
Macro: Small red/blue firm nodule Micro: benign; small round cells, microvasculature, pseudo-glands in myxoid stroma. Variable smooth muscle and nerve ends penetration. |
Marginal excision and reconstruction. |
|
Osteoid osteoma [16] |
Variable symptomology depending on location. Appendicular lesions may be painless or exhibit swelling, erythema and deformity. |
May appear normal or may show solid periosteal reaction with cortical thickening. In advanced cases, vivid radiolucent nidus, sclerotic margin and reactive adjacent bone are seen However, a dense sclerosis may obscure the nidus |
CT: radiolucent nidus, sclerotic rim. Modality of choice. MRI: often nonspecific and unable to identify the nidus. May be misinterpreted for aggressive pathology. |
Macro: brownish red mottled gritty lesion distinct from the surrounding bone. Micro: woven bone bordered by osteoblastic rim. Large and vacuous Osteoblasts. Variable penetration of nerve endings. Mature sclerotic bone surrounds osteoblastic rim. |
Surgery is the final resort after High Intensity Focussed Ultrasound (HIFU) and/or radiotherapy and includes curettage to remove entire nidus. |
|
Osteomyelitis [17] |
Pain, erythema, swelling, instability or pseudoarthrosis, discharging sinus. |
Soft tissue swelling, osteopenia, bony destruction, sequestrum, involucrum and periosteal reaction |
CT: Superior to MRI and plain films to detect bony erosions, sequestrum or involucrum. MRI: most sensitive and specific and is able to identify soft tissue and joint complications. Penumbra sign (dark central abscess with bright inner wall and dark sclerotic outer rim) in subacute cases. |
Macro: variable; from near normality to bone destruction and purulence. Micro: Bony spicules with live osteocytes surrounded by inflammatory cells, areas of bone necrosis and reactive new bone formation. |
Antimicrobial treatment followed by debridement +/-reconstruction if unresponsive. |
|
Enchondroma [18] |
Pain. Bony tenderness secondary to pressure effects, pathological fractures. Predilection to involve proximal phalanx. |
Intramedullary (diaphyseal/ metaphyseal-diaphyseal regions), well-defined, expansive radiolytic lesion with stippled or punctate calcifications. (Hyaline cartilage) |
CT: can detect subtle matrix calcification MRI: Shows signal features typical of benign cartilaginous tumour. |
Macro: bluish hyaline cartilage in lobules of a few millimetres/centimetres size Micro: small chondrocytes in lacunae. No features of atypia/ malignancy |
Observation is the mainstay of treatment. Enchondromas with recurrent symptoms are treated surgically with curettage +/- bone grafting. |
|
ABC [19] |
Pain. Pathological fracture, swelling, soft tissue changes. |
Osteolytic expansile lesion |
CT: can demonstrate sharply defined expansile osteolytic lesions with thin sclerotic margins. MRI: Cyst formation with characteristic fluid-fluid levels |
Macro: Irregular, soft multiloculated, hemorrhagic lesion with thin shell of sclerotic bone. Variable fluid components. Micro: Cavernous blood-filled spaces without endothelial lining. Strands of bone may be seen in the fibrous septa. Benign giant cells. |
Sequential intralesional sclerotherapy. High rates of recurrence with intralesional curettage alone (50%) Sclerotherapy can be used as an adjunct to resection to minimise recurrence |
Table 1: Differential Diagnosis
Higher imaging list is not exhausted and may include nuclear medicine scans review of which is beyond the scope of this article. High-intensity focused ultrasound (HIFU).
An IO-EIC is an uncommon occurrence. Symptomatic involvement of the hand and foot can often be confused with other benign and malignant lesions [table 1]. IO-EIC typically affects the middle to older age group, with a preponderance to the phalanges of the hands [1]. Additionally, a history of trauma or surgery usually precedes the appearance of symptoms further clouding diagnosis [1,2]. Radiologically, an IO-EIC appears as an expanding, unilocular lytic lesion with a narrow zone of transition and sclerotic margins with or without an associated soft tissue swelling; features common to all localized chronic infective, inflammatory or benign neoplasia [1,3,7,8]. Higher imaging such as computed tomography (CT) or MRI may also be limited in the ability to identify an IO-EIC, in particular ruptured or infected case [12]. Unruptured cysts usually contain fluid and develop a sclerotic margin, mimicking simple cysts or geodes [12]. Histologically, IO-EIC contains a thick caseous, keratinous material with or without purulence depending on cyst integrity [9,13]. Micro-histology will resemble that of an extraosseous EIC, namely mature epithelium with or without skin adnexa, chronic inflammatory response and giant cells if ruptured [2]. Furthermore, multiple histological samples are required for diagnosis as the characteristic squamous epithelium may be patchy in distribution [14].
Our case is the first to report an IO-EIC in a metatarsal and a suspected “inside out” mode of cyst rupture with violation of the skin. The hair from the cyst penetrated not only the deep prearticular tissues, but the skin away from the suspected ulcer, creating a secondary nidus for infection. Our patient had no prior history of trauma or surgery of the affected toe. She was an intravenous drug user but had never injected at or adjacent to the foot. This history of substance abuse, her ten-pack years of smoking and the above clinical features pointed towards an “outside-in” loss of skin integrity and infection as the primary diagnosis, which initially favored non-surgical treatment of a suspected superficial infection. Her normal inflammatory markers at presentation and lack of pyrexia were attributed to the excessive use of antibiotics and an EIC was not suspected until dysplastic hair was found intraoperatively. Initial radiographs were also misleading in that the changes were more pronounced at the proximal phalanx [figure 1]. These included a well demarcated zone of destruction and sclerosis, likely resulting from the extraosseous pressure of a ruptured metatarsal cyst, a tracking infection and/or an abscess.
There are only 12 other cases of pedal IO-EIC reported in the literature [table 2]. Of these nine involved a phalanx of the hallux, two involved a lesser ray and one the calcaneum. Most of these cases involved trauma, five of which were iatrogenic following surgery to the affected digit. Patients with chronic conditions, in particular a smoking history and vascular disease, required multiple debridements, prolonged antibiotics and later amputations. Although delayed in most cases, surgery which included excision, excision and grafting or amputation, proved effective in all 12 cases, with satisfactory wound healing and recovery. The majority of the published cases did not undergo microbiological testing of the sample, however of the six cases that did, half grew a wide mix of organisms [5, 8, 12, 20-24], [table 1 and 2]. Chronic cases were associated with multi-microbial infection.
|
Study |
Age/Sex |
Site |
Co morbidities |
TTIS |
Histology |
Microbiology |
Treatment |
|
Wang et al. 2003 [7] |
54 M |
Hallux - distal phalanx |
NA |
NA |
Squamous epithelium lined cyst, keratin debris |
No cultures |
Incision and curettage. |
|
Connolly et al. 2010 [6] |
78 M |
Hallux - distal phalanx |
Smoker, PVD, AE |
5 months |
Squamous epithelium lined cyst, keratin debris |
No growth |
Five episodes of debridement followed by emergency partial amputation at 10 months. |
|
Sugimoto et al. 2021 [4] |
82 F |
Hallux - distal phalanx |
SLE, SLE nephritis (on prednisolone) |
6 months |
Squamous epithelium lined cyst, keratin debris |
Finegoldia magna, Corynebacterium striatum, Anaerobic gram-positive cocci |
Incision and drainage + six months of antibiotics followed by partial amputation. |
|
Chung et al. 2016 [12] |
71 F |
Hallux - distal phalanx |
Takayasu arteritis |
10 days |
Squamous epithelium lined cyst, keratin debris |
Coagulase negative staphylococcus |
Radical (mass and nail plate removed) debridement. |
|
Patel et al. 2006 [8] |
48 F |
Fourth ray - middle phalanx |
Fourth ray hammer toe correction |
NA |
Squamous epithelium lined cyst, keratin debris |
No cultures |
Incision and curettage. |
|
Richardson et al. 2017 [5] |
63 F |
Fifth ray - proximal phalanx |
Fifth ray hammer toe correction |
8 months |
Squamous epithelium lined cyst, bony erosion, inflammation |
No growth |
Amputation of the little toe. |
|
Berghs et al. 1998 [20] |
61 F |
Hallux - distal phalanx |
In growing toe nail surgery |
NA |
Squamous epithelium lined cyst |
No cultures |
Excision and auto-graft bone packing of defect |
|
Berghs et al. 1998 [20] |
33 F |
Hallux - distal phalanx |
In growing toe nail surgery |
NA |
Squamous epithelium lined cyst, keratin debris |
Not growth |
Excision and auto-graft bone packing of defect |
|
Posthuma et al. 2018 [21] |
49 M |
Calcaneum |
Surgical fixation of calcaneum |
4 months |
Squamous epithelium lined cyst, keratin debris, giant cells |
No cultures |
Excision |
|
Kumar et al. 2017 [22] |
54 M |
Hallux - distal phalanx |
Avulsion injury to hallux |
2 months |
Squamous epithelium lined cyst |
No cultures |
Curettage and antibiotics. |
|
Tamburrelli 1984 [23] |
56 M |
Hallux - distal phalanx |
NA |
<24 hours |
Stratum corneum* lined cyst, keratin debris, necrotic cells |
No cultures |
Partial amputation. |
|
Roth 1964 [24] |
41 M |
Hallux - distal phalanx |
Hallux crush injury |
6 months |
Squamous epithelium lined cyst |
No cultures |
Excision |
|
Present Case |
47 F |
Hallux- Metatarsal head |
Smoker, IDU |
3 years |
Ruptured squamous epithelium lined cyst, mature skin organelles, keratin debris, inflammatory cells. |
Staphylococcus aureus, Coagulase negative Staphylococcus, mixed anaerobes |
Amputation and antibiotics |
Table 2: Other cases of IO-EIC.
Studies reporting IO-EIC in the foot. Time to initial surgery (TTIS), Peripheral vascular disease (PVD), alcohol excess (AE), systemic lupus erythematosus (SLE), illicit drug use (IDU). *Stratum corneum is the most superficial later of the epidermis.
Conclusions
Pedal IO-EIC is extremely rare and diagnosis is university delayed due to its mimicking of other conditions. Antibiotics are seldom helpful in isolation, particularly in patients with a history of smoking or vascular disease, and surgical excision is the gold standard. We recommend careful clinical examination for subtle skin changes around the foot and early surgical intervention in the setting of fluctuating responses to repeated courses of antibiotics. A delay in treatment results in significant tissue loss and amputation.
References
1. Memon F: Intraosseous Epidermoid Inclusion Cyst of Distal Phalanx: A Rare Entity. J Clin DIAGNOSTIC Res. Published online. 2016, 10.7860/JCDR/2016/16539.7072
2. Sasaki H, Nagano S, Shimada H, et al.: Intraosseous epidermoid cyst of the distal phalanx reconstructed with synthetic bone graft. J Orthop Surg. 2017, 25:230949901668409. 10.1177/2309499016684096
3. Simon K, Leithner A, Bodo K, Windhager R: Intraosseous epidermoid cysts of the hand skeleton: a series of eight patients. J Hand Surg (European Vol. 2011, 36:376-378. 10.1177/1753193411401987
4. Sugimoto H, Yasue K, Hara M, et al.: Intraosseous epidermal cyst of the great toe that was difficult to distinguish from chronic osteomyelitis: A case report and literature review. Clin Case Reports. 2021, 9:1890-1895. 10.1002/ccr3.3814
5. Richardson MP, Foster JR, Logan DB: Intraosseous Epidermal Inclusion Cyst of the Proximal Phalanx of the Fifth Toe and Review of the Literature: A Case Study. Foot Ankle Spec. 2017, 10:470-472. 10.1177/1938640017690859
6. Connolly JE, Ratcliffe NR: Intraosseous Epidermoid Inclusion Cyst Presenting as a Paronychia of the Hallux. J Am Podiatr Med Assoc. 2010, 100:133-137. 10.7547/1000133
7. Wang BY, Eisler J, Springfield D, Klein MJ: Intraosseous Epidermoid Inclusion Cyst in a Great Toe. Arch Pathol Lab Med. 2003, 127:298-300. 10.5858/2003-127-e298-IEICIA
8. Patel K, Bhuiya T, Chen S, Kenan S, Kahn L: Epidermal inclusion cyst of phalanx: a case report and review of the literature. Skeletal Radiol. 2006, 35:861-863. 10.1007/s00256-005-0058-0
9. Feliciano DR, Reich DR, Freedman DP, Kanter DA. INTRAOSSEOUS DERMOID CYST OF THE MANDIBLE: A CASE REPORT AND REVIEW OF LITERATURE: Oral Surg Oral Med Oral Pathol Oral Radiol. 2019, 128:52. 10.1016/j.oooo.2019.02.114
10. Terui T, Rokugo M, Kato T, Tagami H: Analysis of the proinflammatory property of epidermal cyst contents: chemotactic C5a anaphylatoxin generation. Arch Dermatol Res. 1989, 281:31-34. 10.1007/BF00424269
11. Poonawalla T, Uchida T, Diven DG: Survey of Antibiotic Prescription Use for Inflamed Epidermal Inclusion Cysts. J Cutan Med Surg. 2006, 10:79-84. 10.2310/7750.2006.00022
12. Moser T, Ehlinger M, Chelli Bouaziz M, Fethi Ladeb M, Durckel J, Dosch J-C: Pitfalls in osteoarticular imaging: How to distinguish bone infection from tumour?. Diagn Interv Imaging. 2012, 93:351-359. 10.1016/j.diii.2012.01.021
13. Handa U, Chhabra S, Mohan H: Epidermal inclusion cyst: Cytomorphological features and differential diagnosis. Diagn Cytopathol. 2008, 36:861-863. 10.1002/dc.20923
14. Hoessly M, Lagier R: Anatomico-radiological study of intraosseous epidermoid cysts. RöFo - Fortschritte auf dem Gebiet der Röntgenstrahlen und der Bildgeb Verfahren. 1982, 137:48-54. 10.1055/s-2008-1056157
15. Bao-guo S, Yun-tao W, Jia-zhen L: Glomus tumours of the hand and foot. Int Orthop. 1997, 20:339-341. 10.1007/s002640050092
16. Smolle MA, Gilg MM, Machacek F, et al.: Osteoid osteoma of the foot. Wien Klin Wochenschr. Published online November. 4:2021. 10.1007/s00508-021-01966-0
17. Hogan A, Heppert VG, Suda AJ: Osteomyelitis. Arch Orthop Trauma Surg. 2013, 133:1183-1196. 10.1007/s00402-013-1785-7
18. Chung MK, Park MS, Kim YS, Lee T, Lee KM, Cho BC: An Intraosseous Epidermoid Cyst That Originated from the Nail Bed of Great Toe with Concurrent Joint Infection: A Case Report. J Korean Foot Ankle Soc. 2016, 20:50. 10.14193/jkfas.2016.20.1.50
19. Deventer N, Toporowski G, Gosheger G, et al.: Aneurysmal bone cyst of the foot: A series of 10 cases. Foot Ankle Surg. Published online March. 2021, 10.1016/j.fas.2021.03.002
20. Berghs, B. and Feyen, J., 1998: Intraosseous epidermal inclusion cyst following surgery for ingrowing toenail. 8:183-185. 10.1016/S0958-2592(98)90047-5
21. Posthuma JJ, de Ruiter KJ, de Jong VM, Schepers T: Traumatic epidermal inclusion cyst after minimally invasive surgery of a displaced intra-articular calcaneal fracture: A case report. J Foot Ankle Surg. 2018, 57:1253-1255. 10.1053/j.jfas.2018.03.042
22. Kumar, U. and Lamba, S., 2017: Intraosseous epidermal inclusion cyst of the great toe: Masquerading as bone tumour. Journal of Clinical and Diagnostic Research, 11, pp.EJ01-EJ02.
23. Tamburrelli, F. (1984: A case of epidermoid cysts of the distal phalanx of the big toe. Archivio di Putti per le Chirurgia degli Organi del Movimento. 34:417-422.
24. Roth SI: Squamous cysts involving the skull and distal phalanges. J Bone Joint Surg. 1964, 46:1442-50.
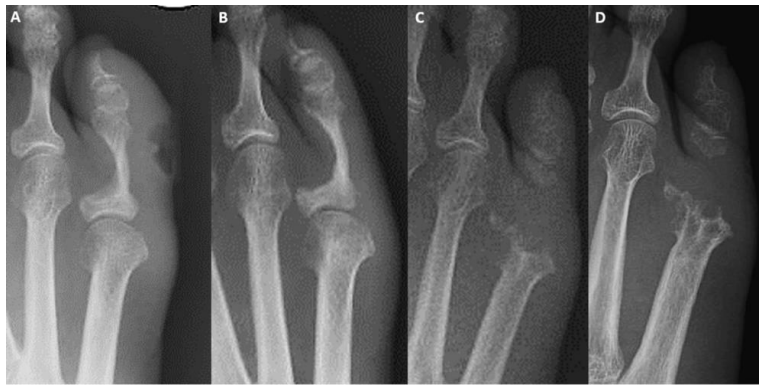
Figure 1
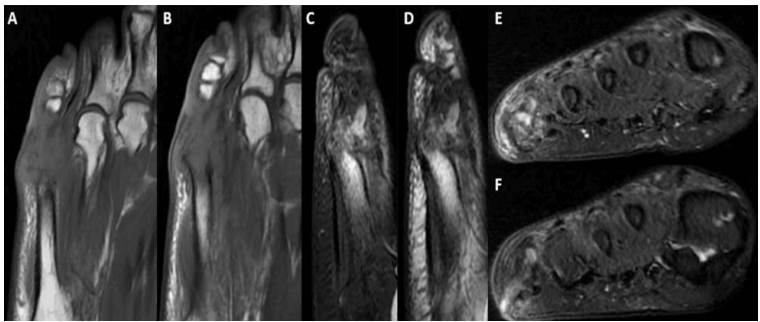
Figure 2
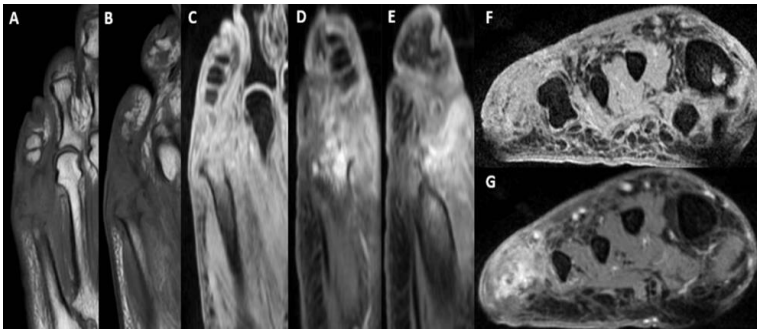
Figure 3
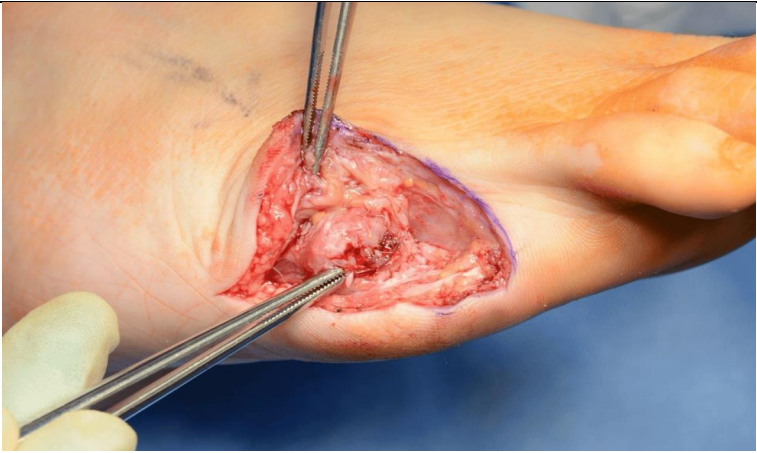
Figure 4
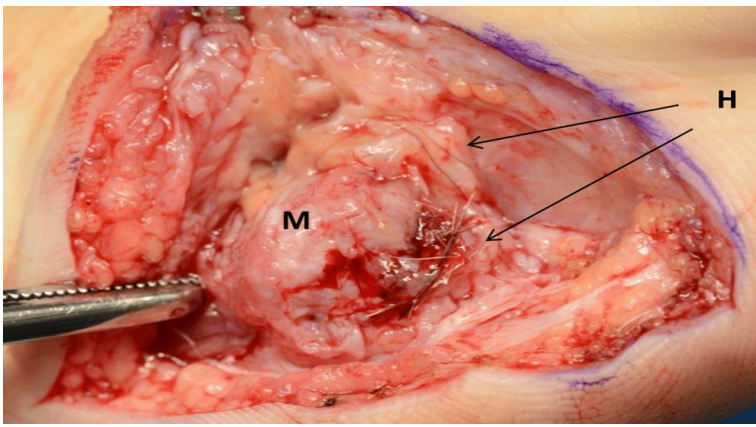
Figure 5
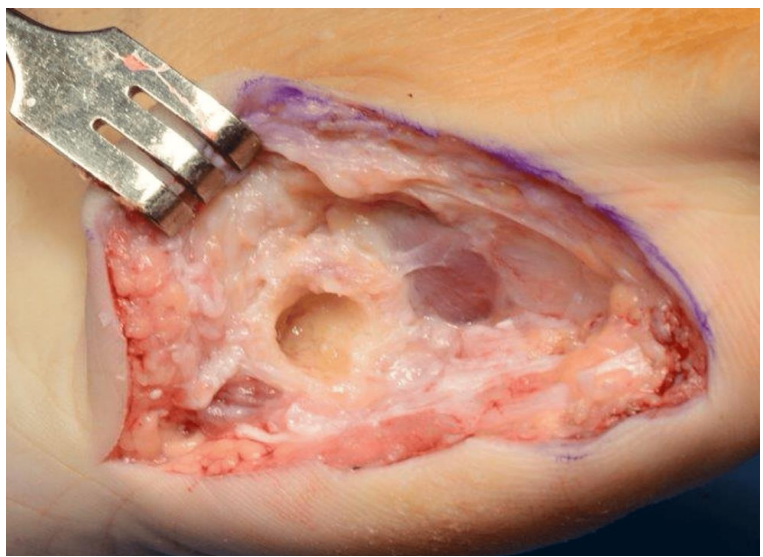
Figure 6
