Multidisciplinary Approach for Extreme Oncoplastic Surgery for Non Palpable and Extensive Breast Tumours using Bracketed Localization Technique
Multidisciplinary Approach for Extreme Oncoplastic Surgery for Non Palpable and Extensive Breast Tumours using Bracketed Localization Technique
Geeta Shettyab*, Hira Naqvia, Ishita Laroyiaa , Simerjit Raia, Melissa Tana, Humaira Khana.
a. City Hospital Breast Unit. Sandwell & West Birmingham Hospitals NHS Trust, Dudley Road, B18 0QH.
b. Kasturba Medical College Mangalore, Manipal Academy of Higher Education, India.
*Correspondence to: Geeta Shetty, City Hospital Breast Unit. Sandwell & West Birmingham Hospitals NHS Trust, Dudley Road, B18 0QH
Copyright.
© 2025 Geeta Shetty This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 06 February 2025
Published: 12 March 2025
Abstract
Oncoplastic breast surgery has become popular in recent years for high tumour to breast ratio, with recent studies showing equivalent survival, lower morbidity and better cosmetic outcome. Accurate preoperative localization of lesions is imperative, especially for multifocal, large non-palpable and pre-invasive lesions to obtain adequate oncological and cosmetic results. Image guidance assures the precision required for this purpose and needs close collaboration between the radiology team and surgeons. Our study analyses the feasibility of bracketed localisations for multifocal and large non-palpable lesions in extreme oncoplastic breast surgery.
Key words: Localisation, Breast, Oncoplastic breast surgery, CWPF, Wires, Tag.
Multidisciplinary Approach for Extreme Oncoplastic Surgery for Non Palpable and Extensive Breast Tumours using Bracketed Localization Technique
Introduction
Oncoplastic breast surgery pushed the boundaries of conventional breast surgery which allows large area of pre-invasive and multifocal disease resection, whilst maintaining a good aesthetic outcome and patient satisfaction, thus improving the quality of life.(1,2,3,5,6)
Several reports have shown lower re-excision and mastectomy rates with no significant delay in initiation of adjuvant treatment because of immediate post-operative complications. (2,3,5,7) Additionally, on intermediate follow-up (up to 4.5 years) local recurrence rates were comparable with other studies. (55).
To contemplate oncoplastic breast surgical procedures a multidisciplinary team approach is imperative involving surgeons, radiologists and breast care nurses. According to several studies, the local recurrence after breast conserving surgery is directly correlated to surgical margin status (8,4,9,10). Hence, radiologist input is crucial for image guided preoperative localisation, especially for non-palpable T2/3, multifocal and pre-invasive cancerous lesions to improve both oncological and cosmetic outcomes(11,12,13). Surgeons need guidance to envisage three dimensional pictures while operating and this can be only possible by careful planning.
Conventionally, wire localisation(WL) has been used as a method for preoperative localisation of nonpalpable breast lesions since 1970s (14), with clear margins reported in a range of 70.8%–87.4% of cases(15). Often it is inserted via the shortest route and not been affected by incision, however in certain oncoplastic procedures such as chest wall perforator flap (CWPF) the focus should be avoiding the damage to the perforators.
Pre operative localization techinques have evolved with several non wire techniques which include Radio guided Occult Localization, Radioactive seeds, Magnetic seeds, Radar reflectors, Radiofrequency tags. There is no significant statistical difference in the surgical outcomes of these devices. The technique selected should be the most accurate to localise the lesion or marker clip, thus facilitating the resection of tumour with adequate free margin, and possibly causing minimal to no discomfort to the patient.
In our department we use LOCalizer system by HOLOGIC Inc for wireless localizations . This technique uses a radiofrequency tag (RFID) and a hand-held reader for localization of non-palpable lesions during surgery. Each tag has its own unique serial number for identification. The localizer can be placed up to 30 days preoperatively and of late we have also used them in patients undergoing neo adjuvant chemotherapy. Similar to wires, for lesions more than 3 cm, two or more localisers can be placed to mark the extreme ends of the lesion. During surgery, the surgeon activates the reflector with the hand piece and follows the signal to guide the excision. The audible and numerical signals change with increasing proximity to the lesion. Once the tissue is removed, the reader console can be used to confirm that all tags have been removed from the tissue cavity and serial numbers are recorded.
Our primary aim was to study the feasibility of multiple localisation techniques for extreme oncoplastic breast surgery with high tumour to breast ratio for non-palpable lesions of more than 3cm. Our secondary aim was to evaluate the re-excision rates.
Methods
This is a single institutional study involving retrospective analysis from a prospectively maintained database with tumour size of more than 3 cm. The recruitment period is from September 2017 to February 2022.
The inclusion criteria are patients undergoing wide local excisions followed by either replacement or displacement oncoplastic procedures, with tumour size equal to or more than 3 cm requiring either bracketed wires or more than one tag for localisation. The exclusion criteria being, multiple localisations for excision biopsies.
The patient demographics, radiological localisation methods, histology, status of surgical margin, re-excisions, weight of resected specimen and complications were retrieved from the electronic record. Localisation accuracy rate was defined as wire or radiofrequency tag deployed within 10 mm of target lesion. The retrieval rate of wire or tag were also recorded.
Two methods of localisation were used for the duration of study; wire localization ( Hawkins™ II BLN) from 2017 September to 2019 December and RFID from January 2020 to February 2022.
Pre-operative patient pathway:
Following diagnosis of non-invasive and invasive malignancy of breast, all patients were discussed in the multidisciplinary team (MDT) meeting. The potential patients with high tumour to breast ratio for oncoplastic procedures were identified in the MDT. The detailed discussion of imaging was carried out between radiologists and surgeons before consultation with patients. This included location and extent of disease and distance from the nipple. This is was recorded as 3D extent of lesion from superior to inferior, medial to lateral and anterior to posterior. This discussion allowed the surgeon to envision and understand the 3D dimension in relation to breast and also to calculate the tumour to breast ratio. This step was imperative because this allowed the surgeon to offer different options of breast operative procedures to patients.
For oncoplastic breast conserving surgery, surgeons assessed the patients for its suitability, and parameters considered were brassier & cup size, ptosis and frame of the patient. For replacement procedures such as chest wall perforator flap, surgeons also assessed the adequacy of tissue on lateral side of chest wall and upper abdomen with a pinch test.
General factors like co-morbidities and BMI were also considered before contemplating any operative procedures. Patients were given all possible options for breast surgery considered as oncologically safe procedures. If the patient consented to go ahead with oncoplastic breast conserving surgery, further in-depth discussion regarding the risks, operative time, recovery period and likely long term implications including symmetrisation surgery for displacement procedure were explained. Pre-operative and post-operative medical photography were prerequisite in all the cases and were part of the medical records.
Tumour localisation
Once a patient was consented for oncoplastic breast conserving surgery, a meeting with radiology collegues was arranged to discuss the image localisation technique and 3D lesion size. The antero posterior extension was considered less important, because conventionally breast tissue is dissected from mastectomy plain anteriorly to chest wall posteriorly. Calcification or distortion in the vicinity of the nipple was vital to know to make decision regarding nipple sparing. The lesion to nipple distance of more than 2 cm was considered feasible to preserve the nipple. Consideration was made to include satellite lesions, distortions, density and calcifications. Other factors to consider were the type of surgical procedures such as chest wall perforator flap, location and number of clip/s, targets for insertion of wires or tag, method of localization e.g; stereotactic or US guidance, skin marks and specific information on measurements required from post wire or tag insertion mammogram. All this information was recorded on the proforma and filed in a designated folder. Annotations were also saved on PACS as key images to help the radiologist performing the procedure.
Image guided localization was performed with either mammographic or sonographic guidance using Hawkins wire from 2017 Sep to 2019 Dec and RFID from Jan 2020 to Feb 2022.
Bracketing technique was used for extensive disease, allowing targeted localization(54). The wires were marked with numbers (wire 1, wire 2 and so on) and its exact location in relation to the breast lesion was recorded. For lateral intercostal artery perforator or lateral thoracic artery perforator flap procedure, lateral approach for wire localisation was avoided to prevent damage to the vessels, similarly inferior approach avoided for anterior intercostal artery perforator flaps. In these circumstances the bracketing was challanging in some cases of wire localization as shortest skin to lesion distance was not chosen in order to preserve the vessels which made targeting difficult as the wire had to traverse a much greater depth then would be ideal.
In order to achieve the good surgical margins the surgeons required multiple measurements to create a 3D interpretation in their mind of the abnormal tissue.
Directly after the procedure, cranio-caudal (CC) and medio-lateral (ML) mammograms were obtained to confirm accurate placement (Figure 1). Skin marking with a permanent pen was also done to indicate site and depth of lesion if the localization was performed on the day of surgery to aid the site of surgical excision. In most cases the localization was done to bracket the extreme ends of the lesion either medial and lateral or superior and inferior.
The radiology report was written after careful examination of the pre and post localisation images. The distance between the lesion ( marker clip) and localization device, its depth from the skin and in case of wire, the lenghth of wire outside the skin was recorded. RFID tag unique number was also documented.
Figure 1: 51yr old with 31 BMI and BRA size 38 DD with multifocal DCIS measuring 50mm on pre op imaging, post op specimen weighed 150gm, tumerectomy with partial breast reconstruction lateral intercostal artery perforator flap. Margins clear. Mediolateral (A) and Craniocaudal (B) mammograms showing two bracketing wires marking the medial and lateral extent of disease. Specimen X ray shows radiologically clear margins (C).
Figure 2: 50yr old with 24 BMI, Bra size 34 B/C with Grade 1 invasive carcinoma measuring 31mm on pre op imaging, post op specimen weighed 58gm, tumerectomy and partial breast reconstruction with lateral intercostal artery perforator flap. Margins clear. Mediolateral and Craniocaudal mammograms showing two RFID tags marking the medial and lateral extent of disease.
Figure 3: 63yr old with 24 BMI, Bra size 36 DD with invasive carcinoma measuring 66 mm on pre op imaging, post op specimen weighed 129gm, tumerectomy and partial breast reconstruction with chest wall perforator flap. Margins clear. Mediolateral (A) and Craniocaudal (B) mammograms showing two RFID tags marking the medial and lateral extent of disease with specimen X rays shows radiologically clear margins (C,D)
Post operative specimen
The surgical specimen was X-rayed and reported by the radiologists. A verbal report was communicated to the surgeon at the same time and report included the presence of clips, tags or wires and radial margin adequacy. If any part of the abnormality was close to the margin, extra tissue was taken as a shave, where appropriate. Aim was to excise 1cm of macroscopically normal breast tissue.(49). It is our routine practice to take cavity shaves following tumerectomy.(50)
Results:
Between sept 2017 to Feb 2022, a total of 76 patients underwent more than one localisation procedure. The average size of pre operative tumour was 65.47 mm (total extent) with a range of (14-108 mm). 35 (45.45%) had multifocal/multicentric tumours and 41 (53.25%) had extensive calcifications. The breakdown of demographics and tumour details are shown in Table 1.
Table 1: Clinico-pathological characteristics of the study population?
|
Characteristic |
Number (percentage) |
|
Pathology |
|
|
?39/76 (51.3%) 12/76 (15.8%) 24/76 (31.6%) 1/76 (1.3%) |
|
Stage |
|
|
26/76 (34.2%) 14/76 (18.4%) 30/76 (39.5%) 6/76 (7.9%) |
|
Nodal status? |
|
|
15/76 (19.7%) 36/76 (47.4%) 25/76 (32.9%) |
|
Grade distribution: |
|
|
5/76 (6.6%) 36/76 (47.4%) 11/76 (14.5%) 24/76 (31.5%) |
|
Hormone receptor positivity: |
|
|
48/76 (63.2%) 13/76 (17.1%) 15/76 (19.7%) |
|
Her 2 Neu status: |
|
|
12/76 (15.8%) 40/76 (52.6%) 24/76 (31.6%) |
|
Neoadjuvant chemotherapy: |
|
|
12/76 (15.8%) 40/76 (52.6%) 24/76 (31.6%) |
|
ACE (Adult Co-morbidity Evaluation) score: |
|
|
36/76 (47.3%) 13/76 (17.1%) 2/76 (2.7%) 25/76 (32.9%) |
51 (68%) had wire guided localizations (39 patients had multiple wires inserted while 12 had a single wire with an ultrasound skin mark). 24 (32%) had radio-frequency tags placed for localization (21 patients had multiple tags placed while 3 patients had single tag in association with an ultrasound guided skin mark).
98.68% (75/76) localizations were accurate as judged by post localization mammography, i.e with in 10mm of target area. Only in 1 patient ultrasound guided tag placement was inaccurate and this was converted to stereo guided procredure, which led to accurate tag placement.
Wire and tag retrieval rate was 100 % and none of them were migrated. No patient had any localisation related complication; except that one patient needed an additional stereo guided localization procedure as ultrasound guided tag placement was not accurate.
The average specimen weight was 144.8 gms (range 23.6-671.2 gms) 67/76 (88%) had clear excision margins and 9/76 (12%) required re-excision. None were converted to mastectomy.
11/76 (14.47%) of the patients had postoperative complications, most of these were minor and were managed conservatively (seroma-2, haematoma-2 and wound breakdowns-2).
Discussion
It is undemanding to resect a unifocal or palpable tumour with less tumour to breast ratio, however for multifocal, non-palpable tumours and DCIS, it is valuable to know the likely radial margins for resection. Detailed planning with the radiology team cannot be overstressed for adequate tumorectomy and to avoid high rate of margin re-excision. Firstly, it is vital to assess the patients’ suitability for breast conserving surgery with or without an oncoplastic procedure. Secondly, an appropriate localisation method needs to be planned to assist surgeons during operation, so as to create a 3-D picture of abnormal tissue. If the tumour is more than 3 cm then we recommend a bracketing technique with more than one localisation. It is not uncommon to have more than 2 localisations sometimes, especially, if the tumour appears to be triangular in shape.
Localisation for oncoplastic procedures requires added time to plan, perform and report these cases. In our study, accurate localization was achieved in 98.68% of the cases. Retrieval rate was 100%. All cancers were confirmed on the final histology and 25 (33%) patients had complete pathological response after neoadjuvant chemotherapy. So, the success rate for multiple localisation for non-palpable tumour was 98.68%. These results are in agreement with other published literature.(27,28,29,30)
Although Wires have been the mainstay of pre-operative localisation, they have many disadvantages including vasovagal reaction, migration within and outside the breast, transection of certain types of wires during surgery with pieces being retained in the breast (23,24,25,26). The procedure in itself is unpleasant and requires greater compliance from patients, as wire must remain in position until surgery is performed. Moreover, this localization has to be performed on the day of surgery to avoid displacement and therefore requires adequate coordination between breast radiologists and surgeons. This can lead to inconvenience and inefficient utilisation of the theatre list. Sometimes, wire localisation could limit the surgical approach and potentially affect cosmetic outcome. For certain oncoplastic procedures such as chest wall perforator flap especially during one stage procedure(51,53)one should avoid lateral approach otherwise this might compromise lateral intercostal vessels.
Newer techniques have developed over a period of time to mitigate some of the risks of standard wire localisation techniques including non-target tissue removal. Some reported issues which are similar to any localisation techniques are haematomas(39,39) resulting in difficulty in obtaining signal from the device(40) and migration of device on release of breast from compression during the stereotactic guidance(41,43). In our study we report no serious complications due to localisation. 11/76 (14.47%) patients had postoperative complications. No patient had migration of tags or wires. Only 1 patient needed further localisation with stereo-tag because of inaccurate placement of tag by ultrasound.
Use of tag from Dec 2020, has improved our theatre efficiency and time management in our services, as well as patients’ quality of life; since these do not necessarily require insertion on the day of surgery(48).
Nonetheless, good communication prior to surgical date with well-illustrated diagrams and annonated mammographic images is necessary to mitigate the risks, as the radiologist planning the procedure with the surgeon may not be the one performing the localization.
The primary aim of our localization of multifocal and non-palpable tumours is removal of lesions with margins free of disease. To facilitate the same, the intra-op specimen X ray, frozen section or margin probe will aid to ensure adequate margin excision. (33,34,35)
Although there is no consensus across the globe, about what defines the clear margin, we accept 1mm free from the tumour as acceptable margin for both invasive and pre invasive tumours (37). Our study shows margin excision in oncoplastic breast surgery with multiple localisation techniques is comparable to other studies irrespective of localisation techniques. Some studies have shown that the presence of ductal carcinoma in situ is one of the factors that increases the number of cases with positive surgical margins regardless of the localization method used(31,46,47). In our study the margin excision rate for tumours with both invasive and DCIS components and for pre-invasive tumours was 12%. When comparing non wire localisation techniques and wire localization techniques, some studies have shown positive margin or re-excision rates is slightly lower with former, however, it was not statically significant (36,40,42,44,45).
In our study the margin excision rates for wire localisation was 14.8% (8/54 patients) and for RFID was 4.54% (1/22 patients) which was not statistically significant (P value- 0.1).
The limitation of this study is that it is from a single centre where a high volume of non-palpable lesions undergo breast conserving surgery with oncoplastic technique, unlike in other centres where many of these patients would have been treated with mastectomy. Experienced radiological and surgical team is imperative to localise these lesions to provide the extreme oncoplastic services for breast conservation respectively. The future aim is to study the cost effectiveness of RFID localisation technique and patient reported outcome to have some objective measurement for cosmetic outcome for both wire and RFID localisation.
Conclusion
A multidisciplinary team approach is essential to facilitate extreme oncoplastic breast conserving surgery for the management of large non-palpable lesions using bracketing image guided techniques. This allows large area of pre-invasive and multifocal disease resection, whilst maintaining a good aesthetic outcome and patient satisfaction, thus improving the quality of life. Several reports have shown lower re-excision and mastectomy rates.
Newer non wire localization devices such as RFID are promising and more effective with similar outcomes compared to wire guided localisation techniques.
References
1. Losken A, Hart AM, Chatterjee A. Updated Evidence on the Oncoplastic Approach to Breast Conservation Therapy. Plast Reconstr Surg. 2017 Nov;140(5S Advances in Breast Reconstruction):14S-22S. doi: 10.1097/PRS.0000000000003951. PMID: 29064918.
2. Clough KB, Lewis JS, Couturaud B, Fitoussi A, Nos C, Falcou MC. Oncoplastic techniques allow extensive resections for breast-conserving therapy of breast carcinomas. Ann Surg. 2003 Jan;237(1):26-34. doi: 10.1097/00000658-200301000-00005. PMID: 12496527; PMCID: PMC1513973.
3. Engel J, Kerr J, Schlesinger-Raab A, Sauer H, Hölzel D. Quality of life following breast-conserving therapy or mastectomy: results of a 5-year prospective study. Breast J. 2004 May-Jun;10(3):223-31. doi: 10.1111/j.1075-122X.2004.21323.x. PMID: 15125749.
4. Corsi, F.; Sorrentino, L.; Bossi, D.; Sartani, A.; Foschi, D. preoperative localization and surgical margins in conservative breast surgery. Int. J. Surg. Oncol. 2013, 2013, 793819. [CrossRef]
5. Munhoz AM, Montag E, Arruda E, Aldrighi CM, Filassi JR, Piato JR, Prado LC, Aldrighi JM, Gemperli R, Ferreira MC. Immediate reconstruction following breast-conserving surgery: management of the positive surgical margins and influence on secondary reconstruction. Breast. 2009 Feb;18(1):47-54. doi: 10.1016/j.breast.2008.10.005. Epub 2008 Dec 24. PMID: 19110425.
6. Eaton BR, Losken A, Okwan-Duodu D, Schuster DM, Switchenko JM, Mister D, Godette K, Torres MA. Local recurrence patterns in breast cancer patients treated with oncoplastic reduction mammaplasty and radiotherapy. Ann Surg Oncol. 2014 Jan;21(1):93-9. doi: 10.1245/s10434-013-3235-8. Epub 2013 Oct 1. PMID: 24081796.
7. Chakravorty A, Shrestha AK, Sanmugalingam N, Rapisarda F, Roche N, Querci Della Rovere G, Macneill FA. How safe is oncoplastic breast conservation? Comparative analysis with standard breast conserving surgery. Eur J Surg Oncol. 2012 May;38(5):395-8. doi: 10.1016/j.ejso.2012.02.186. Epub 2012 Mar 20. PMID: 22436560.
8. Singletary SE. Surgical margins in patients with early-stage breast cancer treated with breast conservation therapy. Am J Surg. 2002 Nov;184(5):383-93. doi: 10.1016/s0002-9610(02)01012-7. PMID: 12433599.
9. Park, C.C.; Mitsumori, M.; Nixon, A.; Recht, A.; Connolly, J.; Gelman, R.; Silver, B.; Hetelekidis, S.; Abner, A.; Harris, J.R.; et al. Outcome at 8 years after breast-conserving surgery and radiation therapy for invasive breast cancer: Influence of margin status and systemic therapy on local recurrence. J. Clin. Oncol. 2000, 18, 1668–1675. [CrossRef] [PubMed] J. Pers. Med. 2021, 11, 99 12 of 14
10. Franceschini G, Sanchez AM, Di Leone A, Magno S, Moschella F, Accetta C, Natale M, Di Giorgio D, Scaldaferri A, D'Archi S, Scardina L, Masetti R. Update on the surgical management of breast cancer. Ann Ital Chir. 2015 Mar-Apr;86(2):89-99. PMID: 25951853.
11. Franceschini G, Mason EJ, Grippo C, D'Archi S, D'Angelo A, Scardina L, Sanchez AM, Conti M, Trombadori C, Terribile DA, Di Leone A, Carnassale B, Belli P, Manfredi R, Masetti R. Image-Guided Localization Techniques for Surgical Excision of Non-Palpable Breast Lesions: An Overview of Current Literature and Our Experience with Preoperative Skin Tattoo. J Pers Med. 2021 Feb 4;11(2):99. doi: 10.3390/jpm11020099. PMID: 33557072; PMCID: PMC7913802.
12. Hayes MK. Update on Preoperative Breast Localization. Radiol Clin North Am. 2017 May;55(3):591-603. doi: 10.1016/j.rcl.2016.12.012. PMID: 28411682.
13. Jeffries DO, Dossett LA, Jorns JM. Localization for Breast Surgery: The Next Generation. Arch Pathol Lab Med. 2017 Oct;141(10):1324-1329. doi: 10.5858/arpa.2017-0214-RA. PMID: 28968154.
14. Hall FM, Kopans DB, Sadowsky NL, Homer MJ. Development of wire localization for occult breast lesions: Boston remembrances. Radiology. 2013 Sep;268(3):622-7. doi: 10.1148/radiol.13121943. PMID: 23970507.
15. Bick U, Trimboli RM, Athanasiou A, Balleyguier C, Baltzer PAT, Bernathova M, Borbély K, Brkljacic B, Carbonaro LA, Clauser P, Cassano E, Colin C, Esen G, Evans A, Fallenberg EM, Fuchsjaeger MH, Gilbert FJ, Helbich TH, Heywang-Köbrunner SH, Herranz M, Kinkel K, Kilburn-Toppin F, Kuhl CK, Lesaru M, Lobbes MBI, Mann RM, Martincich L, Panizza P, Pediconi F, Pijnappel RM, Pinker K, Schiaffino S, Sella T, Thomassin-Naggara I, Tardivon A, Ongeval CV, Wallis MG, Zackrisson S, Forrai G, Herrero JC, Sardanelli F; European Society of Breast Imaging (EUSOBI), with language review by Europa Donna–The European Breast Cancer Coalition. Image-guided breast biopsy and localisation: recommendations for information to women and referring physicians by the European Society of Breast Imaging. Insights Imaging. 2020 Feb 5;11(1):12. doi: 10.1186/s13244-019-0803-x. PMID: 32025985; PMCID: PMC7002629.
16. Rose A, Collins JP, Neerhut P, Bishop CV, Mann GB. Carbon localisation of impalpable breast lesions. Breast. 2003 Aug;12(4):264-9. doi: 10.1016/s0960-9776(03)00105-x. PMID: 14659311.
17. Luini A, Zurrida S, Galimberti V, Paganelli G. Radioguided surgery of occult breast lesions. Eur J Cancer. 1998 Jan;34(1):204-5. doi: 10.1016/s0959-8049(97)00376-6. PMID: 9624261.
18. Pavlicek W, Walton HA, Karstaedt PJ, Gray RJ. Radiation safety with use of I-125 seeds for localization of nonpalpable breast lesions. Acad Radiol. 2006 Jul;13(7):909-15. doi: 10.1016/j.acra.2006.03.017. PMID: 16777565.
19. Harvey JR, Lim Y, Murphy J, Howe M, Morris J, Goyal A, Maxwell AJ. Safety and feasibility of breast lesion localization using magnetic seeds (Magseed): a multi-centre, open-label cohort study. Breast Cancer Res Treat. 2018 Jun;169(3):531-536. doi: 10.1007/s10549-018-4709-y. Epub 2018 Feb 16. PMID: 29453521; PMCID: PMC5953977.
20. Cox C.E., Garcia-Henriquez N., Glancy M.J., Whitworth P., Cox J.M., Themar-Geck M., Prati R., Jung M., Russell S., Appleton K., et al. Pilot study of a new nonradioactive surgical guidance technology for locating nonpalpable breast lesions. Ann. Surg. Oncol. 2016;23:1824–1830. doi: 10.1245/s10434-015-5079-x.
21. Franceschini G, Mason EJ, Grippo C, et al. Image-Guided Localization Techniques for Surgical Excision of Non-Palpable Breast Lesions: An Overview of Current Literature and Our Experience with Preoperative Skin Tattoo. J Pers Med. 2021;11(2):99. Published 2021 Feb 4. doi:10.3390/jpm11020099
22. Mango V.L., Wynn R.T., Feldman S., Friedlander L., Desperito E., Patel S.N., Gomberawalla A., Ha R. Beyond wires and seeds: Reflector-guided breast lesion localization and excision. Radiology. 2017;284:365–371. doi: 10.1148/radiol.2017161661.
23. Van Susante J., Barendregt W., Bruggink E. Migration of the guide-wire into the pleural cavity after needle localization of breast lesions. Eur. J. Surg. Oncol. (EJSO) 1998;24:446–448. doi: 10.1016/S0748-7983(98)92527-6.
24. Azoury F.M., Sayad P., Rizk A. Thoracoscopic management of a pericardial migration of a breast biopsy localization wire. Ann. Thorac. Surg. 2009;87:1937–1939. doi: 10.1016/j.athoracsur.2008.10.069.
25. Volders J.H., Haloua M.H., Krekel N.M.A., Meijer S., Van Den Tol P.M. Current status of ultrasound-guided surgery in the treatment of breast cancer. World J. Clin. Oncol. 2016;7:44–53. doi: 10.5306/wjco.v7.i1.44.
26. Hayes M.K. Update on preoperative breast localization. Radiol. Clin. N. Am. 2017;55:591–603. doi: 10.1016/j.rcl.2016.12.012.
27. Chan B.K., Wiseberg-Firtell J.A., Jois R.H., Jensen K., Audisio R.A. Localization techniques for guided surgical excision of non-palpable breast lesions. Cochrane Database Syst. Rev. 2015:CD009206. doi: 10.1002/14651858.CD009206.pub2.
28. Lovrics P.J., Cornacchi S., Vora R., Goldsmith C., Kahnamoui K. Systematic review of radioguided surgery for non-palpable breast cancer. Eur. J. Surg. Oncol. (EJSO) 2011;37:388–397. doi: 10.1016/j.ejso.2011.01.018.
29. Medina-Franco H, Abarca-Pérez L, García-Alvarez MN, et al. Radioguided occult lesion localization (ROLL) versus wire-guided lumpectomy for non-palpable breast lesions: a randomized prospective evaluation. J Surg Oncol. 2008;97(2):108–111. doi: 10.1002/jso.20880.
30. Ngô C, Pollet AG, Laperrelle J, et al. Intraoperative ultrasound localization of nonpalpable breast cancers. Ann Surg Oncol. 2007;14(9):2485–2489. doi: 10.1245/s10434-007-9420-x.
31. Nagashima T, Hashimoto H, Oshida K, et al. Ultrasound demonstration of mammographic detected microcalcifications in patients with ductal carcinoma in situ of the breast. Breast Cancer. 2005;12:216–220. doi: 10.2325/jbcs.12.216.
32. Liberman L., Kaplan J., Van Zee K.J., Morris E.A., LaTrenta L.R., Abramson A.F., Dershaw D.D. Bracketing wires for preoperative breast needle localization. Am. J. Roentgenol. 2001;177:565–572. doi: 10.2214/ajr.177.3.1770565.
33. Bathla L, Harris A, Davey M, Sharma P, Silva E (2011) High resolution intra-operative two-dimensional specimen mammography and its impact on second operation for re-excision of positive margins at final pathology after breast conservation surgery. Am J Surg 202(4):387–394
34. St John ER, Al-Khudairi R, Ashrafian H, Athanasiou T, Takats Z, Hadjiminas DJ, Darzi A, Leff DR (2017) Diagnostic accuracy of intraoperative techniques for margin assessment in breast cancer surgery: a meta-analysis. Ann Surg 265(2):300–310
35. Versteegden DPA, Keizer LGG, Schlooz-Vries MS, et al. Performance characteristics of specimen radiography for margin assessment for ductal carcinoma in situ: a systematic review. Breast Cancer Res Treat. 2017;166(3):669–679. doi: 10.1007/s10549-017-4475-2.
36. Garzotto F, Comoretto RI, Michieletto S, et al. Preoperative non-palpable breast lesion localization, innovative techniques and clinical outcomes in surgical practice: A systematic review and meta-analysis. Breast. 2021;58:93-105. doi:10.1016/j.breast.2021.04.007
37. Moran M.S., Schnitt S.J., Giuliano A.E., Harris J.R., Khan S.A., Horton J. Society of surgical oncology–American society for radiation oncology consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. J Clin Oncol. 2014;32:1507–1515. doi: 10.1200/JCO.2013.53.3935.
38. Mango V.L., Wynn R.T., Feldman S., Friedlander L., Desperito E., Patel S.N. Beyond wires and seeds: reflector-guided breast lesion localization and excision. Radiology. 2017;284:365–371. doi: 10.1148/radiol.2017161661.
39. Tayeh S., Gera R., Perry N., Michell M., Malhotra A., Mokbel K. The use of magnetic seeds and radiofrequency identifier tags in breast surgery for non-palpable lesions. Anticancer Res. 2020;40:315–321. doi: 10.21873/anticanres.13955.
40. Zacharioudakis K., Down S., Bholah Z., Lee S., Khan T., Maxwell A.J. Is the future magnetic? Magseed localisation for non palpable breast cancer. A multi-centre non randomised control study. Eur J Surg Oncol. 2019;45 doi: 10.1016/j.ejso.2019.06.035. 2016–21.
41. Lamb L.R., Bahl M., Specht M.C., D'Alessandro H.A., Lehman C.D. Evaluation of a nonradioactive magnetic marker wireless localization program. Am J Roentgenol. 2018;211:940–945. doi: 10.2214/AJR.18.19637.
42. Patel S.N., Mango V.L., Jadeja P., Friedlander L., Desperito E., Wynn R. Reflector-guided breast tumor localization versus wire localization for lumpectomies: a comparison of surgical outcomes. Clin Imag. 2018;47:14–17. doi: 10.1016/j.clinimag.2017.07.020.
43. Fung W., Wong T., Chau C., Yu E.L., Chan T., Chan R.L. Safety and efficacy of magnetic seed localisation of non-palpable breast lesions: pilot study in a Chinese population. Hong Kong Med J. 2020 doi: 10.12809/hkmj208559.
44. Srour M.K., Kim S., Amersi F., Giuliano A.E., Chung A. Comparison of wire localization, radioactive seed, and Savi scout ® radar for management of surgical breast disease. Breast J. 2020;26:406–413. doi: 10.1111/tbj.13499.
45. Lee M.K., Sanaiha Y., Kusske A.M., Thompson C.K., Attai D.J., Baker J.L. A comparison of two non-radioactive alternatives to wire for the localization of non-palpable breast cancers. Breast Canc Res Treat. 2020;182:299–303. doi: 10.1007/s10549-020-05707-1.
46. Langhans L, Jensen M.-B, Talman M.-L.M, Vejborg I, Kroman N, Tvedskov T.F. Reoperation rates in ductal carcinoma in situ vs invasive breast cancer after wire-guided breast-conserving surgery. JAMA Surg. 2017; 152: 378https://doi.org/10.1001/jamasurg.2016.4751
47. Laws A, Brar M.S, Bouchard-Fortier A, Leong B, Quan M.L. Intraoperative margin assessment in wire-localized breast-conserving surgery for invasive cancer: a population-level comparison of techniques. Ann Surg Oncol. 2016; 23: 3290-3296https://doi.org/10.1245/s10434-016-5401-2
48. Wazir U, Tayeh S, Perry N, Michell M, Malhotra A, Mokbel K. Wireless breast localization using radio-frequency identification tags: the first reported European experience in breast cancer. In Vivo. 2020; 34: 233-238https://doi.org/10.21873/invivo.11765
49. Fischer, J. E., Ellison, E. C., Upchurch, G. R., Galandiuk, S., Gould, J. C., Klimberg, V., Henke, P., Hochwald, S. N., & Tiao, G. M. (2018). Fischer’s mastery of surgery, seventh edition. Wolters Kluwer Health Adis (ESP).
50. Chagpar AB, Killelea BK, Tsangaris TN, Butler M, Stavris K, Li F, Yao X, Bossuyt V, Harigopal M, Lannin DR, Pusztai L, Horowitz NR. A Randomized, Controlled Trial of Cavity Shave Margins in Breast Cancer. N Engl J Med. 2015 Aug 6;373(6):503-10. doi: 10.1056/NEJMoa1504473. Epub 2015 May 30. PMID: 26028131; PMCID: PMC5584380
51. Geeta Shetty, Arwa Ashoor, Shaista Zafar, Ishita Laroyia, Josiah Moki Mwendwa, Melissa Tan. Ambulatory surgery for partial breast reconstruction with pedicled chest wall perforator flaps. Journal of Plastic, Reconstructive & Aesthetic Surgery. Volume 76, January 2023, Pages 67-70
52. Asgeirsson KS, McCulley SJ, Pinder SE, Macmillan RD. Size of invasive breast cancer and risk of local recurrence after breast-conservation therapy. Eur J Cancer. 2003 Nov;39(17):2462-9. doi: 10.1016/s0959-8049(03)00605-1. PMID: 14602132.
53. Soumian S, Parmeshwar R, Chandarana M, Marla S, Narayanan S, Shetty G. Chest wall perforator flaps for partial breast reconstruction: Surgical outcomes from a multicenter study. Arch Plast Surg. 2020 Mar;47(2):153-159. doi: 10.5999/aps.2019.01186. Epub 2020 Mar 15. PMID: 32203992; PMCID: PMC7093273.
54. Liberman L, Kaplan J, Van Zee KJ, Morris EA, LaTrenta LR, Abramson AF, Dershaw DD. Bracketing wires for preoperative breast needle localization. AJR Am J Roentgenol. 2001 Sep;177(3):565-72. doi: 10.2214/ajr.177.3.1770565. PMID: 11517048.
55. Asgeirsson KS, Rasheed T, McCulley SJ, Macmillan RD. Oncological and cosmetic outcomes of oncoplastic breast conserving surgery. Eur J Surg Oncol. 2005 Oct;31(8):817-23. doi: 10.1016/j.ejso.2005.05.010. PMID: 16043322.
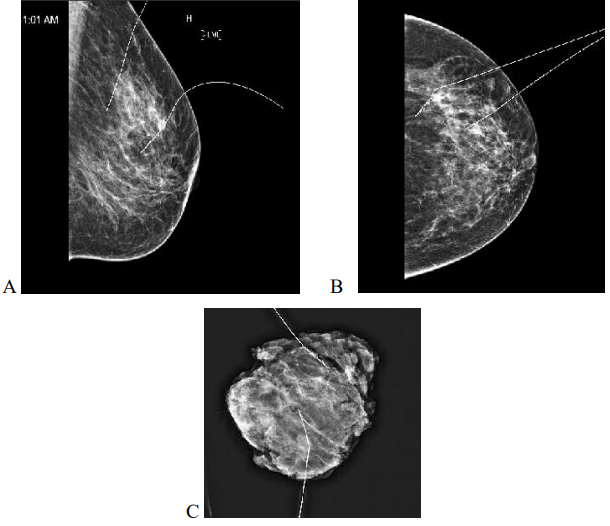
Figure 1
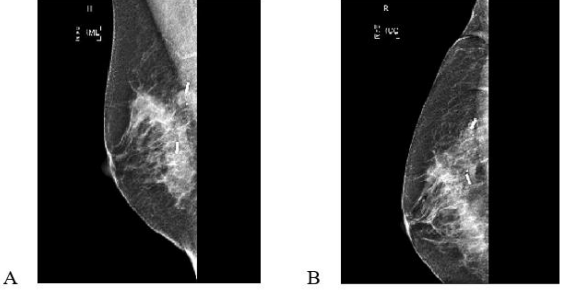
Figure 2
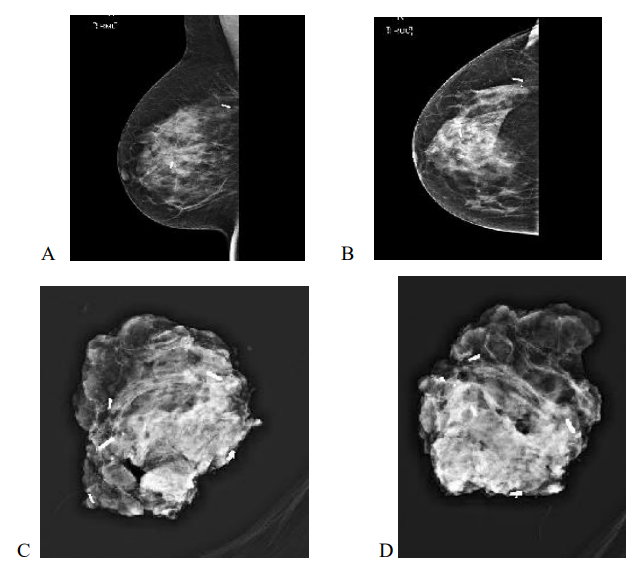
Figure 3
