Metastasizing Pleomorphic Adenoma of the Salivary Gland Involving the Sternum: A Case Report
Metastasizing Pleomorphic Adenoma of the Salivary Gland Involving the Sternum: A Case Report
Dr Shripal Jani1, Dr Shehnaz Kantharia2, Dr Aditya Shah3, Dr Hetal Parikh4, Dr Rajesh Kantharia *
1- Dr Shripal Jani, MDS, Fellowship Oral Oncosurgery, Consultant Oral oncosurgeon, Bankers Superspeciality Hospital, Manjalpur, Baroda 390011.
2- Dr Shehnaz Kantharia, MS ( ENT ), PGDCR, MBA, ENT and Head and Neck Surgeon, Head of Clinical Research, Bankers Superspeciality Hospital, Manjalpur, Baroda 390011.
3- Dr Aditya Shah, MDS ( Oral and Maxillofacial surgery ), Clinical Observer, Dept of Head and Neck Oncosurgery, Bankers Superspeciality Hospital, Manjalpur, Baroda, 390011.
4- Dr Hetal Parikh, MD, FRCPath (Histopathology), Consultant Histopathologist, Unipath Laboratory, Baroda.
*Correspondence to: Dr Rajesh Kantharia, MS, Fellowship Head And Neck Oncosurgery (Tata Memorial Hospital), Program Director Oncology Services, Head and Neck Oncosurgeon, Bankers Superspeciality Hospital, Manjalpur, Baroda 390011.
Copyright.
© 2025 Dr Rajesh Kantharia This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 15 April 2025
Published: 21 April 2025
DOI:https://doi.org/10.5281/zenodo.15622012
Abstract:
BACKGROUND: The acinar-ductal units of the three pairs of main salivary glands (submandibular, sublingual, and parotid) and small salivary glands are encircled by myoepithelial cells, which are functionally altered epithelial cells that produce extracellular matrix. The majority of salivary gland tumors are benign; large glands account for 70% of cases, while small glands account for 25%. Salivary gland tumors make up 2 to 6.5% of all head and neck neoplasms. Tumors in the salivary glands can be cancerous at different rates—most often in the sublingual gland, less so in the submandibular gland, and least commonly in the parotid gland. Among adults, pleomorphic adenoma is the most commonly diagnosed salivary gland tumor, which is often a benign with a well-defined circumference and a modest growth rate. Despite being malignant, metastatic pleomorphic adenoma (MPA) shares identical histological features with benign pleomorphic adenoma (PA). This report details a case of pleomorphic adenoma of the salivary gland exhibiting metastasis to the sternum in an geriatric patient.
Metastasizing Pleomorphic Adenoma of the Salivary Gland Involving the Sternum: A Case Report
Introduction
There are three major pairs of salivary glands in the human body: parotid, submandibular, and sublingual. Minor salivary glands are found along the lateral borders of the tongue, palate, lips, and buccal mucosa. There are notable histological similarities between minor salivary glands and seromucous glands of the respiratory tract, including those in the nasal cavity, larynx, and bronchi.(1)
The acinar-ductal unit is the basic anatomy of all salivary glands. Either mucous cells, serous cells, or both make up acin. The excretory, intercalated, and striated ducts make up the ductal unit. Myoepithelial cells around acini and intercalated ducts. Despite having a dual epithelium and smooth muscle phenotype and producing extracellular matrix, myoepithelial cells are functionally modified epithelial cells.(1)
Benign tumors of the salivary glands are found in 25% of minor salivary glands and 70% of major salivary glands. Salivary glands account for 2–6.5% of all head and neck neoplasms, with 70 to 90% of sublingual glands, 41 to 45% of submandibular, and 15 to 35% of parotid gland tumors being malignant.(2)
The most prevalent salivary gland tumor in adults is called pleomorphic adenoma (PA). They can occur anywhere throughout the upper aerodigestive tract, although most of them start in the parotid gland.(3) It is often a benign tumor that grows slowly and is well-circumscribed.(5) PA can metastasize without malignant transformation, but it can also malignantly change into ex-pleomorphic adenoma.(4)
The three histologic variants of malignant mixed tumors of the salivary glands include metastasizing pleomorphic adenoma, carcinoma ex pleomorphic adenoma, carcinosarcoma.(5)
The rare malignant tumor known as metastatic pleomorphic adenoma (MPA) is histologically distinct from PA but produces secondary tumors in other locations. (4) MPAs are a type of PA that differs genetically from the standard PLAG1/HMGA2 fusions and is more likely to exhibit malignant behavior. (3)
Here, we present a case of pleomorphic adenoma of the salivary gland with a sternal mass confirming metastasizing pleomorphic adenoma on biopsy.
Case Report
A 71-year-old female presented at Bankers Superspeciality Hospital complaining of swelling over the right side of the face and upper neck region, with localised swelling over the sternum. She was asymptomatic 5 months back as she gradually noticed swelling over her right side of her face and upper neck region, which gradually increased in size. In the past (2018), she had surgery at a local hospital, query for parotid surgery; no documents were available ( surgery note and histopathological report). On inspection and palpation, 8*7cm hard proliferative, lobulated growth present over the right side upper neck region, not fixed to underlying structures, crossing midline of the face with no ulceration present over skin (Image 1). No neck nodes were palpable. No sign of facial palsy present. Localised mass of 6*5cm was present over the right side sternum, which was fixed to the underlying bone (Image 2). No skin ulceration was present over sternum region and no axillary nodes were palpable. The performance status of the patient was ECOG 1. Computed tomography of Neck and chest (Plain + Contrast) suggestive of large heterogeneously enhancing lobulated solid cystic lesion seen in right submandibular and upper internal jugular regions and right submental region anterior to right sternocleidomastoid muscle, size of 94(W)*76(AP)*79.6(CC)mm, with mild compression on right IJV. Approx 78*75*92mm soft tissue lesion involving sternum causing osteolytic destruction extending into the anterior mediastinum displacing heart and other structures posteriorly. A few variable-sized soft tissue density nodules scattered throughout both lungs (Image 3 & Image 4). Trucut Biopsy from Right submandibular region s/o pleomorphic adenoma ( composed of epithelial tubules and myoepithelial cells in abundant chondromyxoid stroma) (Image 5 & Image 6). Trucut Biopsy from sternal mass suggestive of malignant tumor, likely to be sarcoma, chondrosarcoma is a possibility. Further Immunohistochemistry was done which suggestive of lesional cells with chondroid type appearance and matrix with positive for AE1/3, CK7 and S100 with KI-67 2-3% favouring Benign metastasizing pleomorphic adenoma (Image 7 & Image 8). PETCT scan was done, which suggestive of FDG avid heterogenously enhancing lobulated soft tissue mass involving right submandibular and submental region 8.6(AP)*7.3(TS)*8.9(CC) ; SUV max 8.6. Medially involving the Right submandibular gland Indenting onto right SCM and strap muscles, Superioly lesion is infiltrating inferior lobe of right parotid gland, overlying skin is involved with no obvious erosion. FDG avid lytic destructive lesion are noted involving sternum, left fifth rib, D12 and L3 vertebrae. Sternal lytic destructive lesion is present with soft tissue compenent with calcification of 8.2*7.2*10.5cm; SUV max 5.9. Soft tissue component is extending into anterior mediastinum and is seen infiltrating right middle lobe lung parenchyma. Left fifth rib is assosciated with FDG avid enhancing soft tissue component 2.6*2.3cm; SUVmax 6.5 (Figure 9). On PETCT Scan report with skeletal metastasis, in multidisciplinary tumor board discussion, the case was discussed and decided to give palliative chemotherapy.
Figure 1: Pleomorphic Adenoma
Figure 2: Sternal Mass
Figure 3: Computed Tomography with contrast of Head and Neck
Figure 4: Computed Tomography with contrast of Chest
Figure 5: Ductal elements and background chondromyxoid stroma and myoepithelial cells
Figure 6: Bone with background PA
Figure 7: P63 Immunohistochemistry myoepithelial cells
Figure 8: AE1&3 highlights epithelial cell closure
Figure 9: Positon Emission Tomography- Computed Tomography
(please go through attached pdf to view all images)
Discussion
The most common noncancerous tumor affecting the salivary glands is pleomorphic adenoma, also known as a mixed tumor, and it is most often treated with surgery aimed at complete cure. Patients between the ages of 30 and 50 most frequently present with a painless lump. Three types of cancer can develop from PA: metastasizing pleomorphic adenoma (MPA), carcinosarcoma, and carcinoma ex-pleomorphic adenoma (CEPA). MPA makes up 1% of all malignant PA, making the later two extremely uncommon.(4)
MPA is a very uncommon type of tumor. The death rate linked to MPA may reach 22%, despite its apparent benign nature. Based on a study of case reports by Nouraei and colleagues, the 5-year disease-specific survival rate for patients with MPA is 58%, while the disease-free survival rate is 50%. Before distant metastases were identified, the majority of MPA patients (81%) had experienced at least one local PA recurrence.(8) As our case of local recurrence of PA from parotid to distant metastasis at the sternum.
Local recurrence and distant metastasis were significantly linked to incomplete surgical excision of the main PA and tumor seeding into the bloodstream during surgical resection.(4) Our patient also gives similar history of previous surgery followed by recurrence and distant metastasis.
The cornerstone of MPA treatment is metastasectomy, where possible. According to log-rank analysis, it provided a notable survival advantage over nonoperative treatment.(8) But our patient had skeletal metastasis with anterior mediastinum involvement which was a factor for contraindicating surgery.
The tumor's melanocytic or myoepithelial origin is indicated by the glial cella's S100 marker for melanocytes and the AE1/3 marker for epithelial cells' pancytokeratin. When it comes to salivary gland cancer, Ki-67's high proliferative activity is the most reliable indicator of a poor prognosis. Compared to patients with a Ki-67 score more than 15%, those with a value of 15% or less had higher survival rates.(5) In our case sternal mass biopsy with IHC shows positive for AE1/3 and S100 cells and also Ki-67 was 2-3% suggestive of Metastasizing pleomorphic adenoma.
In their case report, Seo Young Choi et al. recommended a right upper lobe lobectomy due to a single metastasizing pulmonary pleomorphic adenoma.(6) In their study, Tetsuji Yamaguchi et al. described a case of submandibular gland pleomorphic adenoma that had spread to the lung and bone. They managed the perihilar and bone lesions using chemotherapy (2400 mg of 5-fluorouracil and 340 mg of nedaplatin) combined with external-beam radiation therapy at a dose of 45 Gy. Four months later, they performed a left-complete pneumonectomy and resection of the sternum.(7) As per TBC discussion we started chemotherapy of Inj Doxurubicin 60mg + Inj cisplatin 60mg and it is to be given for 6 cycles.
Conclusion
The exact Etiology of Metastasizing Pleomorphic Adenoma is not known. Literature review says that most cases occurred after surgery or invasive procedures. It is hypothesized that surgical manipulation can lead to tumor cell migration to blood vessels and this hematogenous spread can lead to metastasis. In our case too, local recurrence and sternal metastasis occurred after initial surgical excision of the Pleomorphic Adenoma of the Parotid. We can assume that there may have been cellular migration from the index tumor into the blood vessels or lymphatics.
Conflict of Interest
None
Ethical Approval
None required
References
1) Ghannam MG, Singh P. Anatomy, head and neck, salivary glands. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025.
2) Alsanie I, Rajab S, Cottom H, Adegun O, Agarwal R, Jay A, et al. Distribution and frequency of salivary gland tumours: An international multicenter study. Head Neck Pathol [Internet]. 2022;16(4):1043–54. Available from: http://dx.doi.org/10.1007/s12105-022-01459-0
3) Wasserman, J. K., Dickson, B. C., Smith, A., Swanson, D., Purgina, B. M., & Weinreb, I. (2019). Metastasizing pleomorphic adenoma: Recurrent PLAG1/HMGA2 rearrangements and identification of a novel HMGA2-TMTC2 fusion. The American Journal of Surgical Pathology, 43(8), 1145–1151. https://doi.org/10.1097/PAS.0000000000001280
4) Knight, J., & Ratnasingham, K. (2015). Metastasising pleomorphic adenoma: Systematic review. International Journal of Surgery (London, England), 19, 137–145. https://doi.org/10.1016/j.ijsu.2015.04.084
5) Nagao T, Sato E, Inoue R, Oshiro H, H Takahashi R, Nagai T, et al. Immunohistochemical analysis of salivary gland tumors: application for surgical pathology practice. Acta Histochem Cytochem [Internet]. 2012;45(5):269–82. Available from: http://dx.doi.org/10.1267/ahc.12019
6) Choi, S. Y., Kwon, W., Hong, I. S., & Jung, S. H. (2016). Multimodality imaging of metastasizing pleomorphic adenoma presenting as a solitary pulmonary nodule without local tumor recurrence: A case report. Journal of the Korean Society of Radiology, 74(3), 204. https://doi.org/10.3348/jksr.2016.74.3.204
7) Yamaguchi, T., Ashizawa, K., Nagaoki, K., Abiru, H., Uetani, M., Tagawa, T., Nagayasu, T., & Hayashi, T. (2009). Metastasizing pleomorphic adenoma of the submandibular gland with metastasis to the lung and sternum: CT and MR imaging findings. European Journal of Radiology Extra, 72(2), e69–e72. https://doi.org/10.1016/j.ejrex.2009.07.004.
8) Santaliz-Ruiz LE Iv, Morales G, Santini H, Sánchez-Santiago M, Arroyo A. Metastasizing pleomorphic adenoma: a fascinating enigma. Case Rep Med [Internet]. 2012;2012:148103. Available from: http://dx.doi.org/10.1155/2012/148103
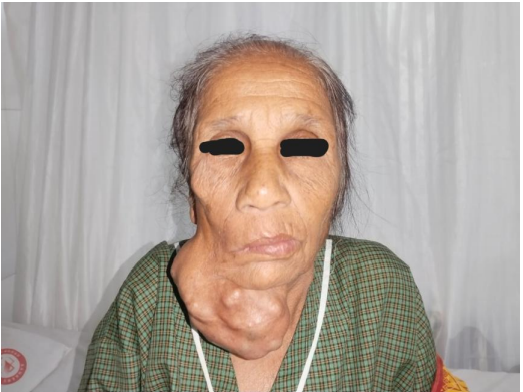
Figure 1
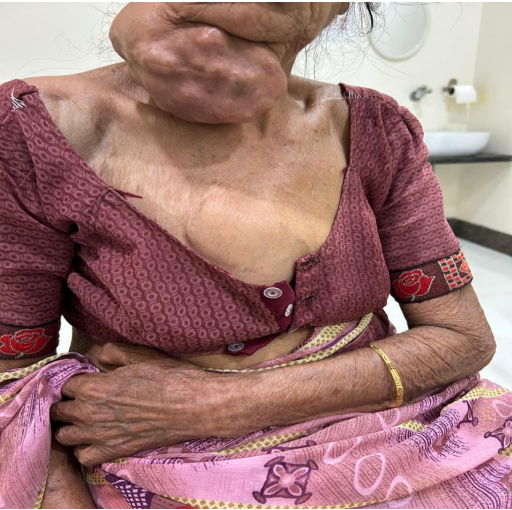
Figure 2
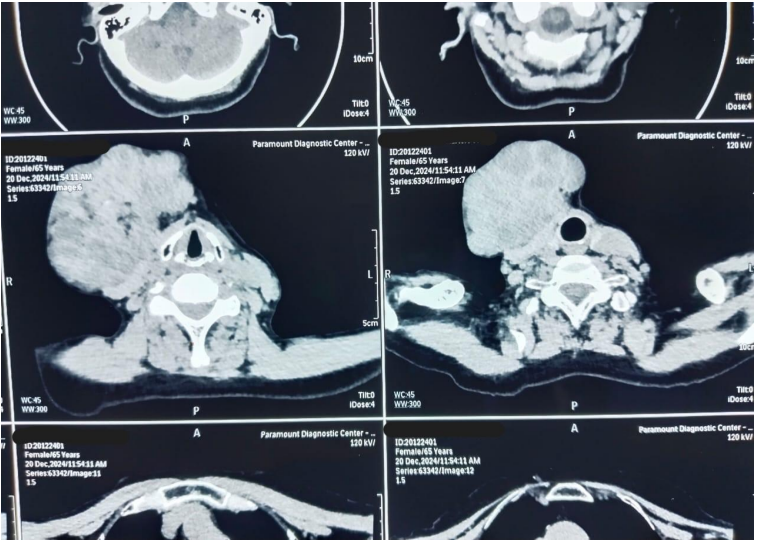
Figure 3
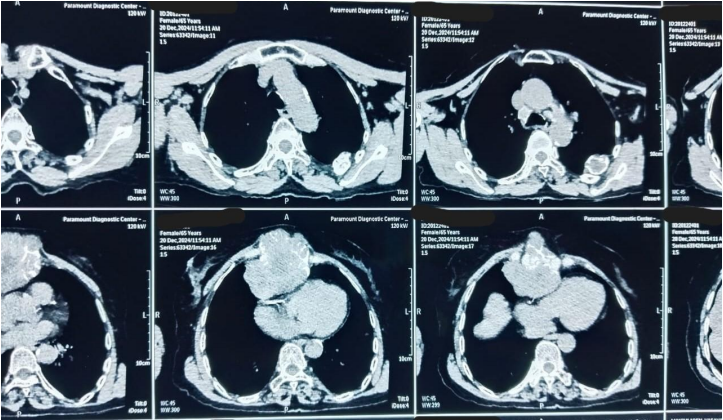
Figure 4
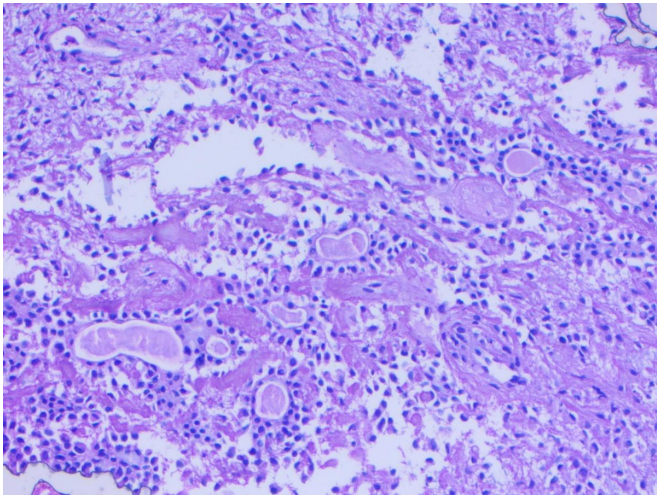
Figure 5
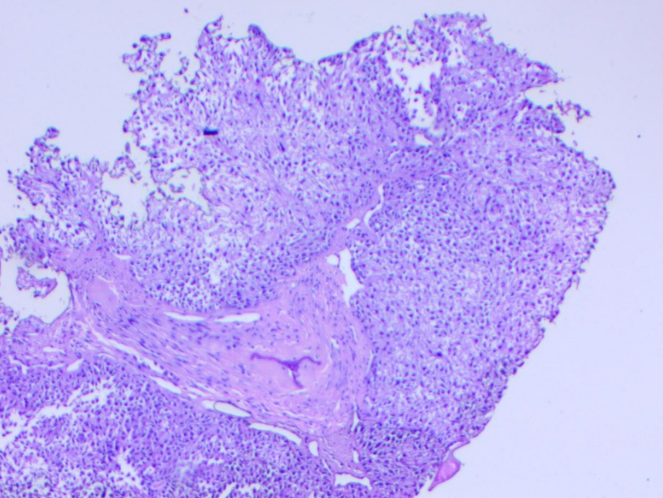
Figure 6
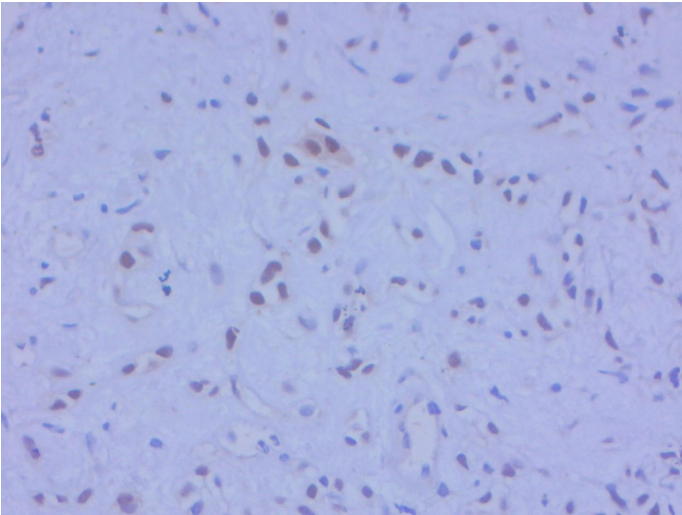
Figure 7
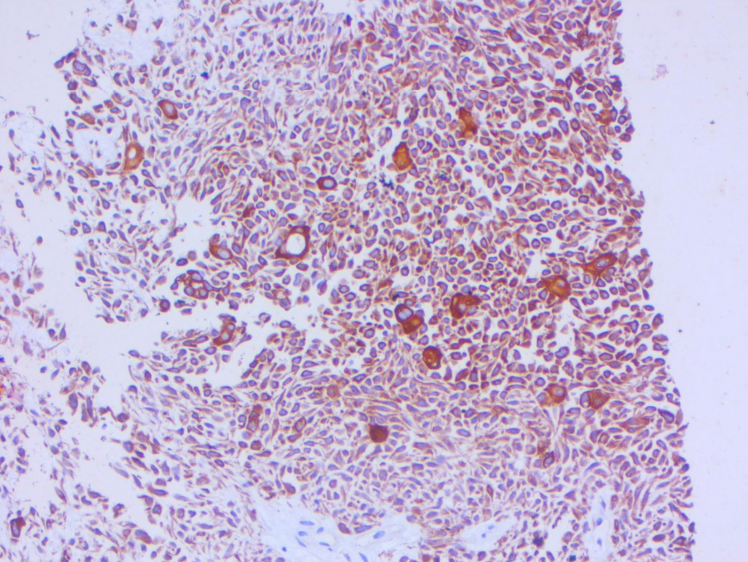
Figure 8
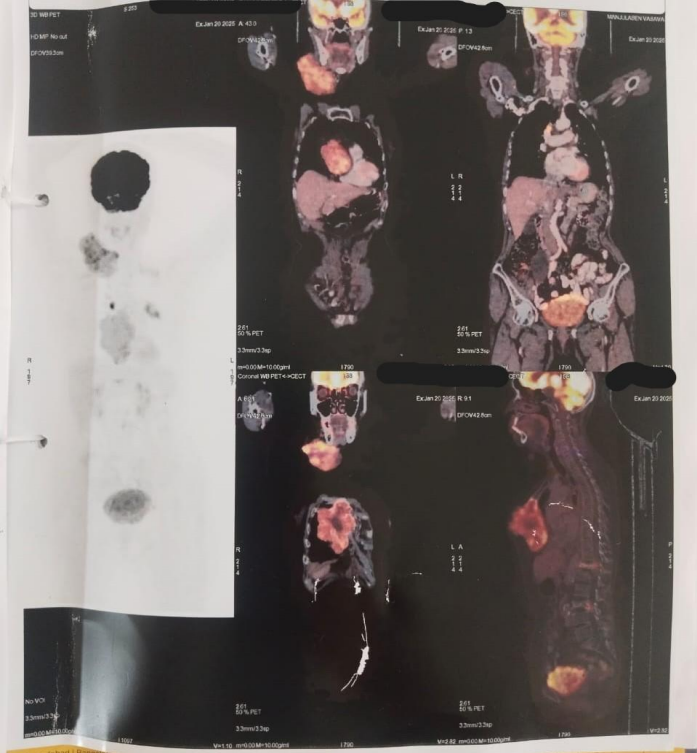
Figure 9
