Giant Pancreatic Pseudocyst an Uncommon Diagnosis in the Gambia. Experience with Two Cases over a Year
Giant Pancreatic Pseudocyst an Uncommon Diagnosis in the Gambia. Experience with Two Cases over a Year
Dr. Luis Vazquez Lopez MD. *1, Dr. Annia Ramos Perez MD. 2, Dr. Firas Alireqsousi MD3, Dr. Lamin Sanyang MD4. Dr. Rosa Maria Martinez Ortega5
1,2 General Surgeon
3 Obstetrics/Gynecologist.
4. MD
5. Associate Professor of Epidemiology and Biostatistics.
*Correspondence to: Dr. Luis Vazquez Lopez MD, Department of Surgery, KGH.
https://orcid.org/0000-0002-4395-1522
Copyright
© 2025 Dr. Luis Vazquez Lopez MD. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 16 April 2025
Published: 26 April 2025
DOI:https://doi.org/10.5281/zenodo.15386510
Abstract
Pancreatic pseudocysts are a well-known complication of acute or chronic pancreatitis, with a higher incidence in the latter. Currently several classification systems are in use that are based on the origin of the pseudocyst, their relation to pancreatic duct anatomy and a possible pseudocyst–duct communication. Diagnosis is accomplished most often by CT scanning, by endoscopic retrograde cholangiopancreatography (ERCP) or by ultrasound, and rapid progress in the improvement of diagnostic tools has enabled detection with high sensitivity and specificity. There are different therapeutic strategies: endoscopic transpapillary or transmural drainage, percutaneous catheter drainage, or open surgery. The feasibility of endoscopic drainage is highly dependent on the anatomy and topography of the pseudocyst but provides high success and low complication rates. Percutaneous drainage is used for infected pseudocysts. However, its usefulness in chronic pancreatitis-associated pseudocysts is questionable. Internal drainage and pseudocyst resection are frequently used as surgical approaches with a good overall outcome, but a somewhat higher morbidity and mortality compared with endoscopic intervention. We therefore conclude that pseudocyst treatment in chronic pancreatitis can be effectively achieved by both endoscopic and surgical means.
The prevalence of pancreatic pseudocysts in acute pancreatitis has been reported to range from 6% to 18.5%. The prevalence of pancreatic pseudocysts in chronic pancreatitis ranges 20% to 40%. The incidence of pseudocysts is low ranging from 1.6 to 4.5% or 0.5 to 1 per 100000 adults per year. The authors report their personal experience with pancreatic pseudocysts that they encountered over 1 year of surgical practice in one of the busiest hospitals in the Gambia and discuss various management options.
There are no randomized studies for the management protocols for pancreatic pseudocysts. Therefore, today we must rely on best clinical practice, but still certain advice may be given. First, it is important to differentiate acute from chronic pseudocysts for management, but at the same time does not miss cystic neoplasia’s.
Conservative treatment should always be considered the first option (pseudocysts should not be treated just because they are there). However, if intervention is needed, a procedure that is well known should always be considered first. The results of percutaneous or endoscopic drainage are probably more dependent on the experience of the interventionist than the choice of procedure and if surgery is needed, an intern anastomosis can hold sutures not until several weeks (if possible 6 weeks). Keywords: pancreas; pancreatic pseudocyst; acute pancreatitis; chronic pancreatitis; pancreatic fluid collection; pancreatic local complication.
Giant Pancreatic Pseudocyst an Uncommon Diagnosis in the Gambia. Experience with Two Cases over a Year
Introduction
A pancreatic pseudocyst is defined as a fluid collection within or adjacent to the pancreas that becomes completely encapsulated with a mature, nonepithelialized, fibrous, inflammatory wall. This process of acute pseudocyst formation requires at least 4 weeks(1, 2). Pancreatic pseudocysts are typically homogeneous with minimal or no necrosis present and without a significant solid component on contrast enhanced computed tomography (CT) imaging(3).
In the following two decades, there was a discrepancy in the nomenclature being used among surgeons, gastroenterologists, and radiologists due to the increasing availability of high-quality cross-sectional imaging. An improved understanding of the nature and pathophysiology of PFCs then led to a revision of definitions in 2012 (the revised Atlanta classification)(4). As shown in Table 1, the 2012 revised Atlanta classification of acute pancreatitis classifies PFCs as acute peripancreatic fluid collection, acute necrotic collection, pancreatic pseudocyst, and walled-off necrosis.(1, 4-6)
A pancreatic pseudocyst is usually diagnosed by ultrasound, computed tomography (CT) scan, or magnetic resonance imaging (MRI). Transabdominal ultrasonography should be utilized as the first step in diagnosing pancreatic pseudocysts. High-resolution endoscopic ultrasound (EUS) detects cystic lesions less than 2 cm in diameter and appears to be of high diagnostic sensitivity.(1) In addition to being an important tool for the diagnosis of pseudocysts, endoscopic retrograde cholangiopancreatography (ERCP) is equally important for endoscopic therapy.
PPC can be treated with a variety of methods: percutaneous catheter drainage, endoscopic ultrasonography assistance, laparoscopic surgery, or open internal drainage. But when and how to choose the best surgical method is still unclear. Open internal drainage surgery is the classic procedure for the treatment of PPC, it is indicated for a significant number of cases.(7)
The prevalence of pancreatic pseudocysts in acute pancreatitis has been reported to range from 6% to 18.5%. The prevalence of pancreatic pseudocysts in chronic pancreatitis ranges 20% to 40%. The incidence of pseudocysts is low ranging from 0.5 to 1 per 100000 adults per year.
There are no studies of the incidence/prevalence of this entity in Africa. There are no published studies of patients with this pathology in The Gambia.
The authors report their personal experience with Giant Pancreatic Pseudocysts (two cases) that they have encountered over a year of surgical practice in one of the busiest hospitals in the Gambia (KGH), with a prevalence in 3 years of 1.61 and discuss various management options. Table 2
Review Criteria
This review entails publications referring to the classification of pancreatic pseudocysts, epidemiology and diagnostic tools, as well as therapeutic options for pancreatic pseudocysts. Only full papers were considered for the review. Based on a search in PubMed the MeSH terms ‘pancreatic pseudocysts and classification’, ‘diagnosis’ and ‘endoscopic, “percutaneous and surgical treatment’ were used, either alone or in combination.
Case presentation CASE # 1
Age: 31
Sex: Male
Main Complaint: Upper abdominal swelling for about 8 months.
HPC: The swelling started small from the RUQ then progressively increased to the epigastrium and LUQ. It was associated with indigestion but no pain, nausea or vomiting. He denies the history of abdominal trauma but confirmed an episode of severe upper abdominal pain few months before he noticed the swelling but he never seeks medical attention.
PMH: none
Surgeries: none Medical allergies: none Transfusions: none
No alcohol intake or smoke
Family history: hypertension.
O/E (abdomen): Full, moves with respiration, no area of tenderness. There is a palpable mass occupying the entire upper abdomen with about 11cm below the costal margin, not painful, not fixed to the anterior abdominal wall but fixed to the underlying structures.
Stable in no obvious distress, not pale, anicteric, acyanose, afebrile to touch. INVESTIGATIONS
• Hb: 15.5 g/dl
• WBC: 4
• Plat: 203
Abdominal Ultrasound: There is an epigastric simple cyst measuring 17x11cm with thickened wall measuring 5.8mm suggesting pancreatic pseudocyst.
Figure 1
• Contrasted Enhanced CT scan: There is a large midline oval cystic mass in the lesser sac compressing and displacing the stomach supero-anteriorly measuring 21x11 cm. It has smooth thin wall and homogeneous content with no solid areas. The mass is seeing closely related to the pancreatic head, pancreatic body and tail normal appearance. Diagnosis: Pancreatic pseudocyst.
Figure 2
• Endoscopy: unremarkable.
• LFT, RFT and Coagulation Profile: unremarkable.
SURGERY
Findings: Large pancreatic pseudocyst rejecting the stomach upwards located at the lesser sac but with the maximum projection at the transverse mesocolon. Wall thickness: 7mm, smooth, no solid areas or nodularity. About 1.5L of clear yellowish fluid drained. Sample of the fluid collected for: Amylase levels, cytology and MCS.
Procedure: Exploratory Laparotomy using a Hanrahan incision, a Transmesocolic Pseudocysto- Jejunostomy in 1 layer of Vicryl 2.0 was performed.
The patient was discharged without complications 8 days after the surgery.
Figure 3
Investigations from the content of the pseudocyst:
MCS: negative Cytology: negative
Amylase levels from the pseudocyst fluid: not available
CASE # 2
Age: 1G Sex: Male
Main Complaint: Abdominal pain and vomiting.
HPC: the patient refers to abdominal pain located at the epigastrium for the past 2 months, initially it was a mild pain, but it has become worse over time. It was associated with vomiting that started being postprandial but then it progressed to be more frequent vomiting basically everything he ate. The patient also noticed a mass in the upper abdomen progressively increasing the size over time. He denies fever or any other symptoms.
PMH: blunt abdominal trauma around 6 months before the beginning of the symptoms. He never went to the hospital.
Family history: none
Allergies: none
Previous surgeries: none
Transfusions: none
No alcohol consumption or smoking
Family history: hypertension.
O/E (abdomen): Full, moves with respiration, no area of tenderness. There is a palpable mass occupying the entire upper abdomen with about 11cm below the costal margin, not painful, not fixed to the anterior abdominal wall but fixed to the underlying structures.
Stable in no obvious distress, not pale, anicteric, acyanose, afebrile to touch. INVESTIGATIONS
- Hb: 15.5 g/dl
- WBC: 4
- Plat: 203
Abdominal Ultrasound: There is an epigastric simple cyst measuring 17x11cm with thickened wall measuring 5.8mm suggesting pancreatic pseudocyst.
Figure 4
SURGERY
FINDINGS: Retro gastric Pancreatic Pseudocyst with a thick wall of 8mm approximately, containing
+- 1.5L of clear and yellowish fluid. The Stomach completely collapsed.
Procedure: Exploratory Laparotomy + “Juracz” Pseudocysto-Gastrostomy. (Trans gastric approach). Sample of the fluid was collected for MCS, Cytology and Amylase)
Figure 5, Figure 6, Figure 7
Differential Diagnosis
Pancreatic cystic neoplasm:
Particularly if multiloculated, may display significant denudation of the lining epithelium.
• Intraductal papillary mucinous neoplasms (IPMNs)
• Serous cystic neoplasms:
Even if completely denudated, it displays a subepithelial network of delicate capillary channels
• Mucinous cystic neoplasms (MCNs):
Even if completely denudated, it displays ovarian type Stroma
• Cystic neuroendocrine neoplasms:
Display a neuroendocrine epithelial lining, positive at immunohistochemistry for chromogranin A and synaptophysin.
• Solid pseudopapillary neoplasms:
Present neoplastic tissue, with necrotic / hemorrhagic cystic changes and tumor cells that are positive at immunohistochemistry for beta catenin (nuclear), LEF1 and CD10.
• Cystic pancreatic ductal adenocarcinoma:
Display malignant ductal epithelial lining, which is positive at immunohistochemistry for CK7 and CK8/18.
Acute necrotic collection:
• Differs from pseudocyst because it is not well defined, and it is filled by a commixture of fluid and necrosis, with different density.
• Appears during the clinical manifestation of the necrotizing pancreatitis.
Walled off necrosis:
• Differs from pseudocyst because it is filled by a commixture of fluid and necrosis, with different density.
Infective cysts:
• Secondary bacterial infection may produce cysts.
• Rarely, Echinococcus spp. may cause hydatid cyst in the pancreas and it should be suspected in endemic region.
Cystic lymphangiomas:
• Lesion associated with prominent lymphoid tissues that demonstrate ectatic spaces lined by endothelial type cells.
Lymphoepithelial cysts:
• Radiologically and sonographically indistinguishable from pseudocyst.
• Smears show abundant anucleated squamous cells, keratinous debris, nucleated squamous cells and it is possible to recognize squamous cell block with preserved granular layer.
Cysts associated with paraduodenal pancreatitis:
• Differential diagnosis is given by the clinicopathological context.
Squamoid cysts of the pancreas:
• Extremely uncommon cyst that is lined exclusively by squamous epithelium, lacking in solid areas.
Acinar cystic transformation of the pancreas:
• Cystic lesion lined by acinar epithelium
• Not excluded the presence of foci of mucinous or squamous epithelium
Congenital cysts of the pancreas:
• Rare cysts that arise by developmental errors of pancreatic ducts, presumably related to localized obstruction of the duct in the utero
• Typically, these cysts are symptomatic before the second year of age and histologically, they present nonmucin producing cuboidal, columnar or flattened cells
Enteric duplication cysts:
• Rare congenital malformations that are most diagnosed in children
• These cysts display gastric or intestinal type lining epithelium and a well- developed, bilayered muscular wall.
Ciliated foregut cysts:
• Cytology is characterized by amorphous debris, rare macrophages and ciliated columnar cells and detached ciliary tufts, which distinguish ciliated foregut cyst from pseudocyst.
Epidermoid cysts:
• Presence of squamous epithelium and keratin flakes.
Colliquated metastasis:
• Renal cell carcinoma is the most common source of metastasis to the Pancreas.
• Other malignancies that involve the pancreatic district are melanoma, pulmonary, mammary, gastric and colonic adenocarcinoma
Outcome and follow-up
Case 1: The patient was discharged without complications 8 days after the surgery, and is doing well on follow-up.
Case 2: The patient was discharged without complications 10 days after the procedure. Currently asymptomatic.
|
Collection type |
Definitions |
|
Acute peripancreatic fluid collection |
Peripancreatic fluid seen within the first 4 wk after onset of interstitial edematous pancreatitis, with no associated peripancreatic necrosis or features of a pseudocyst |
|
Pancreatic pseudocyst |
An encapsulated collection of fluid with a well- defined inflammatory wall usually outside the pancreas with minimal or no necrosis. It usually takes more than 4 weeks for it to mature after the onset of interstitial edematous pancreatitis and contains amylase- rich fluid with no solid debris |
|
Acute necrotic collection |
A collection containing variable amounts of both fluid and necrosis associated with necrotizing pancreatitis within the first 4 wk. The necrosis can involve pancreatic parenchyma and/or the peripancreatic tissues |
|
Walled-off necrosis |
A mature, encapsulated collection of pancreatic and/or peripancreatic necrosis with a well-defined inflammatory wall. It usually matures 4 weeks after the onset of necrotizing pancreatitis |
Table 1. Definitions of pancreatic fluid collections according to the 2012 Atlanta classification(5).
|
YEAR |
ADMISSIONS Dep OF SURGERY at KGH |
AVERAGE PER YEAR |
No DE CASES WITH PP IN 3 YEARS |
PREVALENCE/1000 ADMISSIOS |
|
2021 |
1234 |
1240 |
2 |
1.61 |
|
2022 |
1346 |
|||
|
2023 |
1140 |
Table 2.
Epidemiology and Etiology
Pancreatic pseudocysts affect up to 1 per 100000 adults per year, and their incidence ranges from 1.0%-4.5% among patients with pancreatitis, regardless of etiology The etiology of pancreatic pseudocysts parallels that of pancreatitis. In countries where there is high alcohol consumption, alcoholic pancreatitis is the most common cause, accounting for 59%-78% of all pancreatic pseudocysts. However, alcohol itself does not lead to a greater predisposition to pseudocyst formation(5). About one-third of pseudocysts are located near the head of the gland, and the remaining two-thirds occur in the tail.(8) The pathogenesis of a pseudocyst formation in chronic pancreatitis is not well understood, but it may involve acute exacerbation of underlying pancreatitis with new calculus or protein plug formation in the pancreatic ducts.(8)
Pathophysiology
A pancreatic pseudocyst is a consequence of inflammatory or obstructive processes within the pancreas, which result in pancreatic duct disruption and the leakage of enzyme-rich pancreatic juice into the retroperitoneum, with ensuing autodigestion of the surrounding tissue.(4, 5)This enzymatic activity promotes further inflammation and produces a cyst wall with abundant granulation tissue but no true epithelial lining.(1) Subsequently, against the background of the inflammatory process, this fluid accumulation is separated by the deposition of fibrin and the formation of a granulation shaft, leading to the formation of a fibrous connective tissue wall.(5) The persistence of an acute fluid collection beyond 4 weeks and formation of a well-defined wall define a pancreatic pseudocyst Thus, in the course of its development, pancreatic pseudocysts go through several stages in the early period: 1- Progression of the inflammatory infiltrate; 2- acute fluid accumulation; and 3- formation of an encapsulated fluid accumulation, rich in pancreatic enzymes and confined to fibrous peritoneal tissue and/or the retroperitoneal space and serous membrane of adjacent organs.(1) The absence of epithelium allows differentiation of pseudocysts from true cysts and cystic neoplasms.(5)
Classification
In 1991, D’Egidio and Schein proposed a classification of pancreatic pseudocysts according to whether they develop the following acute or chronic pancreatitis, with an emphasis on the presence of any pancreatic ductal pathology.(5) Type I pseudocyst is defined as a post-necrotic pseudocyst associated with acute pancreatitis. Type II pseudocyst is a post-necrotic pseudocyst occurring after an episode of acute-on-chronic pancreatitis, without a ductal stricture (though there may be a pseudocyst-duct communication). Furthermore, type III pseudocyst is defined as a “retention” pseudocyst that occurs due to chronic pancreatitis and is associated with ductal stricture and pseudocyst-duct communication.(1, 5, 9) In 2002, Nealon and Walser (14) proposed a more simple classification based on the pancreatic duct anatomy from endoscopic retrograde cholangiopancreatography (ERCP), which is provided in Table 3.(5, 10)
Table 3 Classification of pancreatic pseudocyst concerning the pancreatic ductal anatomy(1, 10)
In 2015, using a large retrospective study of 893 patients with pancreatic pseudocysts, Pan et al proposed a new classification based on the anatomical location and clinical manifestation of the pseudocysts, along with the relationship between the cyst and the pancreatic duct. Table 4
|
Type |
Description |
|
I |
<5 cm without symptoms, complications and neoplasia |
|
II |
Suspected of cystic neoplasia |
|
III IIIA IIIB |
Pseudocyst located in the pancreatic uncinate process Communication with pancreatic duct (+) Communication with pancreatic duct (-) |
|
IV
IVA IVB IVC |
Pseudocyst located in head, neck or body of pancreas Communication with pancreatic duct (+) Distance of cyst to gastrointestinal wall is <1 cm Neither IVa nor IVb applies |
|
V Va Vb |
Pseudocyst located in the pancreatic tail Splenic vein involvement or upper gastrointestinal bleeding Distance from the cyst to gastrointestinal wall is <1 cm, without splenic vein involvement or upper gastrointestinal bleeding |
|
|
|
Table 4: Classification of pancreatic pseudocyst.(10)
Discussion
When the main pancreatic duct or one of the ductal branches becomes disrupted, pancreatic secretions collect in the retroperitoneum or peripancreatic tissues. Most of these accumulations resolve spontaneously, but in some cases, the fluid collection persists, and a wall of fibrous or inflammatory tissue forms around the fluid reservoir. This wall lacks an epithelial lining, hence the use of the term pseudocyst.(11) Various reasons can lead to pancreatic duct discontinuity and pancreatic juice's leak into the abdominal cavity, which was considered a main pathogenesis of pancreatic pseudocyst currently.(2, 7) Although the clinical manifestations of pancreatic pseudocysts are diverse, the basic principle of treatment is mainly to solve abnormal drainage of pancreatic juice. In more than 2 thirds of patients, pseudocysts spontaneously disappeared, so the first treatment option for this disease is conservative treatment based on observation and follow- up.(7) The clinical presentations of pancreatic pseudocysts vary widely from completely asymptomatic to pancreatic and bile duct obstruction.(11, 12) The most common clinical manifestations are abdominal pain, nausea and vomiting, early satiety, weight loss and obstructive jaundice(9). The location of the pancreatic pseudocyst is extremely important, both in symptomatology and management(11)
The American College of Gastroenterology released guidelines on the evaluation of pancreatic cysts in 2018 and offered strengths of recommendation for those guidelines. MRI or MR cholangiopancreatography (MRCP) are the imaging modalities of choice when confirming the diagnosis of a pancreatic pseudocyst, and this usually follows indications of an encapsulated fluid mass on ultrasound (US).(2) CT or US can be alternatives for patients unable to undergo MRIs.(1, 13)
Conservative management includes octreotide and bromhexine hydrochloride to decrease pancreatic secretions along with supportive management that includes a low?fat diet, antibiotics, and in certain conditions nil orally with TPN. These methods, however, require strict compliance and follow?up. With the PP less than 5 cm in diameter, spontaneous resolution is more likely in children compared to adults with rare recurrences.(9)
Drainage of pseudocyst can be performed by open laparotomy, laparoscopy, percutaneous drainage, or endoscopically with or without EUS guidance.(1, 14) Ideally, any intervention should be delayed to around six weeks after the inciting pancreatitis episode, in the absence of complicating factors, to allow the pseudocyst wall to thicken and mature. Some surgeons continue to adhere to the ‘Rule of 6’ for the treatment of pancreatic pseudocysts (cysts >6 cm or duration >6 weeks), based on the assumptions that 6 weeks is sufficient time for (A) the pseudocyst to resolve spontaneously if it will resolve at all and (B) the pseudocyst wall to mature to be sturdy enough to hold sutures postoperatively(11). It is important to note that pseudocysts of any size or duration require more immediate intervention if it is thought to be the cause of life-threatening illness.(8, 15)
In this case report, two cases of giant pancreatic pseudocyst are presented, one of them with a history of acute pancreatitis and the other case with a history of blunt abdominal trauma, this last one a common cause in children, however this is a cause to keep in mind in adults, as one of the cases presented. (16) Ultrasound scans and Ct scans were used as alternatives to accomplish the diagnosis since the cases were unable to undergo MRIs. (16)
Indications for therapeutic intervention of pancreatic pseudocysts.
Complicated pancreatic pseudocysts (one criterion sufficient)
• Compression of large vessels (clinical symptoms or seen on CT scan)
• Gastric or duodenal outlet obstruction
• Stenosis of the common bile duct due to compression
• Infected pancreatic pseudocysts
• Hemorrhage into pancreatic pseudocyst
• Pancreatico-pleural fistula
Symptomatic pancreatic pseudocyst
• Satiety
• Nausea and vomiting
• Pain
• Upper gastrointestinal bleeding (10–20%)
Endoscopic drainage
Beginning with endoscopic drainage (ED), there are two approaches we have reviewed. ED of a pseudocyst is done either via a transpapillary or transmural route, and the latter is considered a cystogastrostomy (CG)(17). EUS is used to determine the pseudocyst's communications and location. A transpapillary pseudocyst drainage uses endoscopic retrograde cholangiopancreatography (ERCP) guidance to place a stent through the ampulla of Vater and into the pancreatic duct, assuming there is communication between the duct and the pseudocyst.(13) (18)Success depended on the length and location of the stent, as well as the degree of pancreatic duct obstruction (partial vs. complete); there were higher rates of resolution in stenting of partial duct obstructions (88%) versus complete duct obstruction (12%).(4, 13)
Cystogastrostomy
CG is transmural drainage that uses endoscopy with EUS guidance to locate, confirm, puncture, stent, and drain a pseudocyst that is directly in contact with either the gastric or duodenal wall.(13) A retrospective study at the West China Hospital of Sichuan University compared CG with Roux-en- Y cystojejunostomy (RCJ) and concluded that there was no significant difference (p=0.467) in recurrence rates between the two methods of cyst drainage.(13)
Percutaneous drainage
Percutaneous drainage (PD) involves either simple percutaneous aspiration or catheter placement and is guided by CT or US.(13) The use of PD as a form of noninvasive pseudocyst management has fallen out of favor in recent years. The outcomes of noninvasive drainage techniques were compared in one study involving 164 patients: 55 of whom had PD while 109 underwent ED. In the 109 patients who underwent ED, treatment success was considerably higher compared to those managed by PD (70% vs. 31%), with success being defined as the “complete resolution or a decrease in the size of the pancreatic fluid collections (PFC) to less than 2 cm on follow-up CT”.(4, 13)
Roux-en-Y cystojejunostomy
RCJ is a laparoscopic technique that creates an anastomosis between the proximal Roux end of the jejunum (created after dividing the jejunum ~25-30 cm distal to the gastroduodenal junction) and the most dependent part of the pseudocyst. This can be accessed either through the transverse mesocolon or the gastrocolic omentum [13]. Finally, an entero-entero anastomosis is created to allow for gastric contents to drain into the jejunum about 40 cm distal to the Roux end.(13)
Laparoscopic cystogastrostomy
Laparoscopic cystogastrostomy typically involves accessing the pseudocyst through the posterior aspect of the gastric compartment.(19) Indications for the procedure are symptomatic cysts or patients experiencing complications secondary to cyst formation. Regarding complications secondary to the procedure, one analysis looked at the different post-surgical complications in 347 patients who underwent PD and 248 patients who underwent laparoscopic drainage (LD). The rates of intraabdominal abscess were 9.8% in PD vs. 5.6% in LD; the sepsis rates were 31.4% in PD and 13.7% in LD, and respiratory failure rates were 17.3% in PD and 9.7% in LD.(13, 14)
Surgical drainage
Historically surgical drainage, which includes cystogastrostomy, Cystoduodenostomy, or cystojejunostomy, was the method of choice for pancreatic pseudocyst drainage.(6, 8) Surgical drainage is indicated for patients with pancreatic pseudocysts that cannot be treated with endoscopic techniques and patients who fail endoscopic treatment. Definitive treatment depends on the location of the cyst(20). Pancreatic pseudocysts closely attached to the stomach should be treated with a cystogastrostomy, some pseudocysts are not in contact with the stomach or duodenum. The surgical treatment for these patients is a Roux-en-Y cystojejunostomy. Surgical cyst enterostomy is successful in achieving immediate cyst drainage in over 90% of cases. Following initial resolution, recurrent pseudocyst formation may occur in up to 12% of cases during long-term follow-up, depending on the location of the cyst and underlying cause of the disease.(13) A retrospective study done in 2009 comparing endoscopic drainage with surgical drainage (open or laparoscopic) found a similar rate of complications, but surgical drainage was associated with a higher treatment success rate. In conclusion, surgical drainage is a safe alternative to other less invasive procedures and is reserved for cysts that need a wider stoma for drainage, resection of pancreatic necrosis, or cysts of unknown etiology.(8)
Despite recent developments in minimally invasive techniques and further progress in CT- and ultrasound-guided therapy, surgical drainage is still a principal method in the management of pancreatic pseudocysts. It traditionally includes internal and external drainage and excision. A surgical approach can be indicated in patients with a) complicated pseudocysts, i.e. infected and necrotic pseudocysts; b) pseudocysts associated with pancreatic duct stricture and a dilated pancreatic duct; c) suspected cystic neoplasia; d) coexistence of pseudocysts and bile duct stenosis; and e) complications such as compression of the stomach or the duodenum, perforation and hemorrhage due to erosion of arteries or pseudoaneurysms. Timing of surgical intervention depends on maturation of the cyst wall. In chronic pancreatitis pseudocysts can be treated without any delay under the assumption that maturation of the cyst wall has already taken place and can thus withstand sutures, whereas optimal timing in acute or traumatic pseudocysts is more difficult.
1. Surgical internal drainage
Internal drainage is the method of choice for uncomplicated mature pseudocysts. Depending on the topographic anatomy, pseudocyst gastrostomy is done for cysts directly adherent to the posterior wall of the stomach. Small (<4 cm) pseudocysts in the head and the uncinated process of the pancreas are eligible for pseudocystoduodenostomy and pseudocystojejunostomy can be performed for all other cysts including extremely large (>15 cm) cysts. There is controversy as to whether pseudocyst gastrostomy and pseudocystoduodenostomy are equivalent in their outcome: pseudocyst gastrostomy has been reported to be simple, quick and less prone to infections, but tends to be associated with more frequent upper gastrointestinal bleedings. Pseudocystojejunostomy seems to be more popular, and results are somewhat better than for pseudocyst gastrostomy. Newell et al. found no significant difference in cyst recurrence, morbidity or mortality between cystogastrostomy and cystojejunostomy but the duration of the operation and blood loss were less after cystogastrostomy. This was the method we used in both cases, with excellent results, no complications, and short hospital stays. Following up for over a year without post operatory complications.
2. Pseudocyst resection
Resection is an alternative procedure to internal drainage for chronic pseudocysts and indications include painful chronic pancreatitis, multiple cysts, gastrointestinal hemorrhage from pseudoaneurysms, common bile duct or duodenal obstruction and technical inability to drain pseudocysts located in the uncinated process. Resection is performed by different operation methods including partial left-sided pancreatectomy preserving the spleen if possible, or by partial right-sided pancreatectomy (Whipple's procedure, pylorus-preserving pancreatoduodenectomy, Beger's operation or Frey's procedure).
3. Laparoscopic surgery
Due to continuing progress in laparoscopic techniques minimally invasive surgery offers new modalities in the treatment of pancreatic pseudocysts. Although laparoscopic pseudocystogastrostomy and pseudocystojejunostomy result in adequate internal drainage and minimal morbidity, experience is limited and long-term outcome of relevant studies is awaited
4.External drainage
External drainage is indicated for immature cysts with infected contents and for ruptured cysts. It hardly ever applies to patients with chronic pancreatitis unless the pancreatic cyst has developed after a superimposed attack of necrotizing pancreatitis
Learning points.
Pancreatic pseudocysts commonly occur in patients with a history of pancreatitis. Always consider blunt abdominal trauma as a possible cause.
Giant pancreatic pseudocysts, those measuring 10 cm or more in major diameter, are rare and can be a surgical challenge.
Giant pancreatic pseudocysts can be treated through surgical anastomosis between the pseudocyst wall and the stomach, duodenum or jejunum.
Cystojejunostomy is the anastomosis of choice when surgically decompressing a giant pancreatic pseudocyst, since it allows for dependent drainage.
Conclusions
Giant hemorrhagic pancreatic pseudocysts have not been directly reviewed as there is limited literature, if any, on their occurrence.(13) However, after careful review, we do propose some therapeutic approaches to giant pancreatic pseudocysts and hemorrhagic pancreatic pseudocysts. Using pseudocyst recurrence rates and complication rates as measures of success in pseudocyst drainage techniques, laparoscopic CG has the lowest of both rates when compared to PD and endoscopic CG; this makes laparoscopic CG a successful approach and method to manage pseudocysts, especially those that have favorable proximity between the pseudocyst and gastric wall.(13)
ACKNOWLEDGMENT
We want to thank the participating institutions for helping throughout the manuscript.
References
1. Zhu H, Du Y, Wang K, Li Z, Jin Z. Consensus guidelines on the diagnosis and treatment of pancreatic pseudocyst and walled-off necrosis from a Chinese multiple disciplinary team expert panel. Endosc Ultrasound. 2024;13(4):205-17.
2. Groskreutz D, Ahmad Y, Vargas J, Assaker R. Interventional management and diagnostic follow-up of a large pancreatic pseudocyst: A case report. Radiol Case Rep. 2023;18(1):60-3.
3. .
4. Zhang HY, He CC. Early endoscopic management of an infected acute necrotic collection misdiagnosed as a pancreatic pseudocyst: A case report. World journal of gastrointestinal surgery. 2024;16(2):609-15.
5. Koo JG, Liau MYQ, Kryvoruchko IA, Habeeb TA, Chia C, Shelat VG. Pancreatic pseudocyst: The past, the present, and the future. World journal of gastrointestinal surgery. 2024;16(7):1986- 2002.
6. Son TQ, Hoc TH, Huong TT, Dinh NQ, Van Tuyen P. A ruptured pancreatic pseudocyst causes acute peritonitis with clinical characteristics of a gastrointestinal tract perforation. Journal of surgical case reports. 2022;2022(5):rjac164.
7. Ye J, Wang L, Lu S, Yang D, Hu W, Lu H, et al. Clinical study on cystogastrostomy and Roux- en-Y-type cystojejunostomy in the treatment of pancreatic pseudocyst: A single-center experience. Medicine. 2021;100(10):e25029.
8. Misra D, Sood T. Pancreatic Pseudocyst. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Tanuj Sood declares no relevant financial relationships with ineligible companies.2025.
9. Suleman M, Tadayo J, Tendwa I, Amsi P, Tsandiraki J, Lodhia J. Pancreatic pseudocyst as a cause for gastric outlet obstruction. Clinical case reports. 2023;11(3):e7122.
10. Tan JH, Chin W, Shaikh AL, Zheng S. Pancreatic pseudocyst: Dilemma of its recent management (Review). Experimental and therapeutic medicine. 2021;21(2):159.
11. Wang GC, Misra S. A giant pancreatic pseudocyst treated by cystogastrostomy. BMJ case reports. 2015;2015.
12. Martinez Hernandez A, Granel Villach L, Bellver Oliver M, Moya Herraiz A. Giant pancreatic pseudocyst. An atypical presentation. Dig Liver Dis. 2022;54(2):280-1.
13. Sabir S, Peace S, Ho C, Oi K, Le K. Giant Hemorrhagic Pancreatic Pseudocyst: A Case Report and Guidelines for Care. Cureus. 2022;14(8):e28398.
14. Jain AK, Singh SK, Jain S, Agrawal P. Endoscopic Ultrasound-Guided Pancreatic Pseudocyst Drainage in Children: A Case Series. Journal of Indian Association of Pediatric Surgeons. 2022;27(4):478-80.
15. Janoska-Gawronska A, Pietrukaniec M, Kopys-Sikora M, Niezgoda M, Holecki M. A fatal course of pancreatic pseudocyst rupture: reflections on the management of the patient. Authors' reply. Pol Arch Intern Med. 2023;133(10).
16. Jaiswal V, Naz S, Ishak A, Batra N, Quinonez J, Mukherjee D, et al. A rare case of pediatric pancreatic pseudocyst. Clinical case reports. 2022;10(5):e05879.
17. Ogura T, Ueno S, Nishioka N, Okuda A, Nishikawa H. Endoscopic Ultrasound-Guided Pancreatic Pseudocyst Drainage Using a Novel Dilation Device with Double Guidewire Insertion Function (with Video). Dig Dis Sci. 2024;69(5):1517-9.
18. Elfert K, Chamay S, Santos LD, Mohamed M, Beran A, Jaber F, et al. Increasing trend of endoscopic drainage utilization for the management of pancreatic pseudocyst: insights from a nationwide database. Clin Endosc. 2024;57(1):105-11.
19. Naik P, Sahoo MR, Nayak J. Comparison of surgical outcome between conventional laparoscopic cystogastrostomy method and plication of the edge of anterior gastrotomy in patients with pancreatic pseudocyst: A retrospective study at two tertiary care centres. Journal of minimal access surgery. 2022;18(2):181-5.
20. Gupta R, Singh R, Bhandari A. Minimally Invasive Open Cystogastrostomy for Giant Pancreatic Pseudocyst in Pediatric Patients. Journal of Indian Association of Pediatric Surgeons. 2024;29(1):81-3..
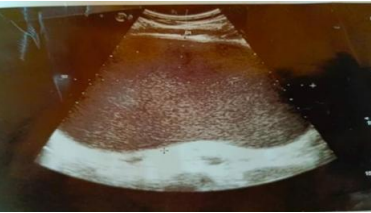
Figure 1
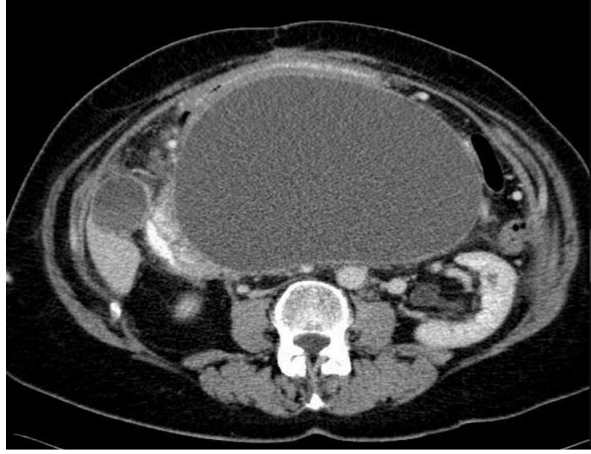
Figure 2
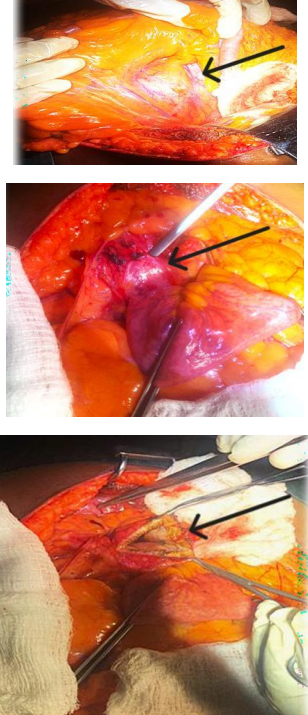
Figure 3
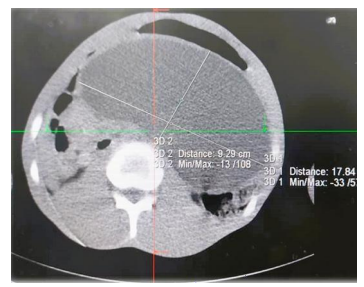
Figure 4
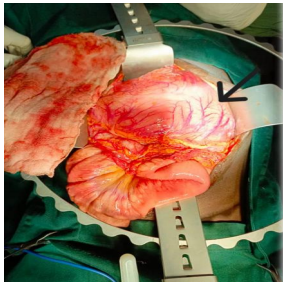
Figure 5
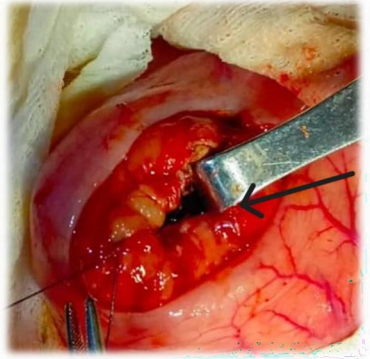
Figure 6
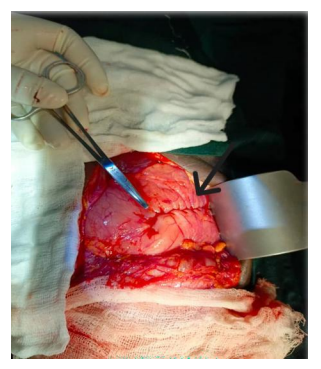
Figure 7
