A Comparative Study of the Effectiveness of Oral Nifedipine versus Intravenous Labetalol in controlling hypertension in Severe Pre-eclampsia
A Comparative Study of the Effectiveness of Oral Nifedipine versus Intravenous Labetalol in controlling hypertension in Severe Pre-eclampsia
Dr. Maitri Snehal Shah1, Dr. Manvi Verma2*, Dr. Sunil Sharma3, Dr. Himani Jivani4
1. Senior Resident, Department of Obstetrics & Gynaecology, MGM Institute of Health Sciences, Kamothe, Navi Mumbai
2. Assistant Professor, Department of Obstetrics & Gynaecology, MGM Institute of Health Sciences, Kamothe, Navi Mumbai
3. Professor & Head of Department, Department of Obstetrics & Gynaecology, MGM Institute of Health Sciences, Kamothe, Navi Mumbai
4. Junior Resident, Department of Obstetrics & Gynaecology, MGM Institute of Health Sciences, Kamothe, Navi Mumbai.
*Correspondence to: Dr Manvi Verma, Assistant Professor, Department of Obstetrics and Gynecology, MGM Institute of Health Sciences, Kamothe, Navi Mumbai
Copyright
© 2025 Dr. Maitri Snehal Shah. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 12 May 2025
Published: 19 May 2025
Abstract:
Background: To study and compare the effectiveness, efficacy and side effects between oral nifedipine and intravenous labetalol in controlling hypertension in acute cases of severe pre-eclampsia.
Methods: A Prospective Comparative study of pregnant patients with blood pressure > 160/110 mm Hg after 20 weeks of gestation was conducted in the Department of Obstetrics and Gynaecology at MGM Medical College & Hospital, Navi Mumbai over a period of one and a half years from 1st August 2022 to 31st December 2023. A total of 100 cases were studied, 50 receiving oral nifedipine and the other 50 receiving intravenous labetalol.
Results: Though intravenous labetalol and oral nifedipine both are equally efficacious first line anti-hypertensive drugs, oral Nifedipine was observed to have a quicker onset of action, with lesser number of doses required to reach target blood pressure with convenient route of administration. Thus, it can be preferred in settings where intravenous access is challenging, remote or low resource settings or patients who prefer oral medication. Additionally, nifedipine works as a uterine relaxant, shown to increase urine output, has no effect on pulmonary vasculature, has lesser side effects and costs 0.5% of the overall cost for intravenous labetalol.
Conclusion: Oral nifedipine could be the better choice between the two for treating hypertensive emergencies however, the decision should be individualized, considering patient-specific factors, clinical setting, and resource availability.
Keywords: Oral nifedipine, Intravenous labetalol, Hypertensive emergency, Severe pre-eclampsia.
A Comparative Study of the Effectiveness of Oral Nifedipine versus Intravenous Labetalol in controlling hypertension in Severe Pre-eclampsia
Introduction
Hypertensive disorders of pregnancy, including chronic hypertension (with or without superimposed preeclampsia/eclampsia), gestational hypertension, preeclampsia (with or without severe features), HELLP syndrome, and eclampsia, present a significant risk of morbidity to both mother and fetus. (1) The prevalence of Hypertensive disorders in pregnancy in India has been found to be 11%. (2) Severe hypertension in pregnancy is defined as a sustained systolic blood pressure of 160 mmHg or higher, or a diastolic blood pressure of 110 mmHg or higher, and requires hospital assessment. (3)
Hypertensive disorders of pregnancy have been associated with adverse outcomes like placental abruption, postpartum hemorrhage, prematurity and fetal growth restriction. Various risk factors like increased maternal age, family history of hypertension in pregnancy, body mass index >30 ?kg/m2, hyperglycemia , In-vitro fertilization and polycystic ovary syndrome have been found to be associated. (4) Severe hypertension in pregnancy needs immediate management to control blood pressure and the drug agents used are Hydralazine, Nifedipine and labetalol. (5) The aim of the present study was to compare the effectiveness of oral nifedipine versus intravenous labetalol in controlling hypertension in severe pre-eclampsia. The objectives were to study the complications, feto-maternal outcome and cost-efficacy.
Materials & Methods
Study Setting:
This was a prospective comparative study of pregnant patients with blood pressure > 160/110 mm Hg after 20 weeks of gestation conducted in the Department of Obstetrics and Gynaecology at MGM Medical College & Hospital, Navi Mumbai over a period of one and a half years from 1st August 2022 to 31st December 2023.
Sampling and sample size:
A total number of 100 hypertensive pregnant women with a BP >160/110 mm Hg were divided into two groups- Group A with 50 cases receiving oral nifedipine and Group B with 50 cases receiving intravenous labetalol
Sample size was based on the formula,
N= z2*p*(1-p)
D2
Where,
P= Incidence i.e. 10% (0.10)s
Z=1.96 (constant for 5% precision)
D=6.1% allowable error
Thus, n= 1.96*1.96*0.10*0.90 / 0.06*0.06
N=96.04 = 96
A total sample size of 100 patients was selected. Total sample size = n + defaulters = 100.
Methodology
After obtaining ethical approval from the institutional ethics committee, the subjects who fulfilled the inclusion and exclusion criteria were recruited in the study after informed consent. Information was collected through a pre-tested structured proforma.
Inclusion Criteria:
Pregnant women with blood pressure >160/110 mm Hg with >20 weeks of gestation with or without proteinuria and willing to give written informed consent were included.
Exclusion Criteria:
Patients with chronic hypertension, asthma, cardiogenic shock, cardiac failure, pulmonary oedema, chronic obstructive pulmonary disease, renal disease, bradycardia; exposure to either medication within the preceding 24 hours; with absolute contraindication to labetalol or nifedipine and refusing to give written consent were excluded from the study.
Patients underwent detailed history, clinical examination and laboratory investigations. Routine haematological, biochemical and radiological tests were done (CBC, LFT, RFT, BG, PT-INR, Serology, TSH, OGCT, Ultrasonogram). Urine output and vitals were monitored. Fundus examination was done. Cardiotocography and ultrasound scan for fetal well-being was done. Patients were divided into Group A (Oral Nifedipine) and Group B (Intravenous Labetalol) with 50 patients each. In a semi- recumbent position, oral nifedipine was given (10mg tablet up to five doses) and Intravenous Labetalol was given in doses of 20, 40, 80, 80 and 80 mg; 20 minutes apart until a target Blood pressure of <150/100 mm Hg was obtained. Future anti-hypertensive agents were given based on individual case requirements.
Statistical Analysis:
The data was entered in Microsoft Excel and analysed statistically using SPSS-25. Quantitative variables were expressed as mean value ± standard deviation or median ± interquartile range. Qualitative data was expressed as percentages and proportions. Appropriate statistical test was used to infer association between 2 variables and a p value of < 0.05 was considered statistically significant.
Results
Demographic & Baseline Characteristics
In group A, 25 patients were in the age group of 18 to 25 years and 26 to 37 years each respectively. In group B, 26 patients were in the age group of 18 to 25 years and 24 patients were in the age group of 26 to 37 years. The difference between two groups in terms of age was not found to be statistically significant (p = 0.84).
28 patients in group A and 30 patients in group B were primigravida. 22 patients in group A and 20 patients in group B were multigravida. The difference between two groups was not statistically significant (p = 0.68).
Vital parameters such as systolic blood pressure, diastolic blood pressure, pulse rate, haemoglobin and serum creatinine levels were comparable across both groups at the time of admission and there was no statistically significant difference between them in the two groups (p >0.05).
Efficacy
Table 1 shows the comparison of number of doses required to control blood pressure. The mean number of doses required to control blood pressure in Group A was 2.28 (2.28 +/- 1.05 SD) which was lower than the mean number of doses required to control blood pressure in group B-2.74 (2.74 +/- 1.04 SD). The difference between the two groups was statistically significant (p = 0.03).
The average time taken in nifedipine administered patients was 45.60 minutes (45.60 +/- 21.01 SD) while it was 54.80 minutes (54.80 +/- 20.92 SD) in intravenous labetalol. (Table 2) The difference between two groups was found to be statistically significant (p = 0.04).
Table 1 Comparison of number of doses required to control blood pressure given as per the schedule decided
|
Variable |
Groups |
t-statistics, p-value* |
|||
|
Group A |
Group B |
||||
|
Mean |
SD |
Mean |
SD |
||
|
Number of doses required |
2.28 |
1.05 |
2.74 |
1.04 |
- 2.19, 0.03 |
* p-value <0.05 statistically significant; Independent samples t-test applied
Table 2 Comparison of time taken to control blood pressure between groups
|
Variable |
Groups |
t-statistics, p-value* |
|||
|
Group A |
Group B |
||||
|
Mean |
SD |
Mean |
SD |
||
|
Time taken to control (in minutes) |
45.60 |
21.01 |
54.80 |
20.92 |
- 2.23, 0.04 |
* p-value <0.05 statistically significant; Independent samples t-test applied
Complications
Table 3 shows the comparison of complications between groups in the present study. No patients in both groups had any complication of hypotension. Headache as a complication was present in 4 patients in group A while it was seen in 5 patients in group B. The association between headache and type of drug administered was not statistically significant (p = 1.00). Dizziness was found to be present in higher proportion (24%) of patients in group B as compared to 8% of patients in group A. The association was found to be statistically significant (p = 0.03) with dizziness more commonly associated with patients administered with intravenous labetalol as compared to nifedipine. Palpitations were found to be present in 2 patients in group A and no patients in group B. There was no statistically significant association between palpitations and type of drug administered (p = 0.49). Tachycardia was present in 12 patients in group A as compared to no patients in group B. The association was found to be statistically significant (p = 0.001) with it being more common in patients administered with nifedipine as compared to labetalol. Nausea or vomiting was present in 10 patients in group B as compared to no patients in group A. The association between nausea/vomiting and type of drug administered was found to be statistically significant (p = 0.03). Breathing difficulty and bradycardia were found to be present in 6 and 8 patients respectively in group B as compared to no patients in group A. The association was found to be statistically significant (p = 0.03 and p = 0.001).
Table 3 Comparison of complications between groups
|
Complications |
Groups |
?2, p-value* |
|
|
Group A n (%) |
Group B n (%) |
||
|
Hypotension |
|||
|
Absent |
50 (100) |
50 (100) |
- |
|
Headache |
|||
|
Present |
04 (08) |
05 (10) |
Fisher’s Exact, 1.00 |
|
Absent |
46 (92) |
45 (90) |
|
|
Dizziness |
|||
|
Present |
03 (06) |
12 (24) |
6.35, 0.03 |
|
Absent |
47 (94) |
38 (76) |
|
|
Palpitations |
|||
|
Present |
02 (04) |
00 (00) |
Fisher’s Exact, 0.49 |
|
Absent |
48 (96) |
50 (100) |
|
|
Tachycardia |
|||
|
Present |
12 (24) |
00 (00) |
13.63, 0.001 |
|
Absent |
38 (76) |
50 (100) |
|
|
Nausea/Vomiting |
|||
|
Present |
00 (00) |
10 (20) |
11.11, 0.001 |
|
Absent |
50 (100) |
40 (80) |
|
|
Breathing difficulty |
|||
|
Present |
00 (00) |
06 (12) |
Fisher’s Exact, 0.03 |
|
Absent |
50 (100) |
44 (88) |
|
|
Bradycardia |
|||
|
Present |
00 (00) |
08 (16) |
Fisher’s Exact, 0.001 |
|
Absent |
50 (100) |
42 (84) |
|
|
Total |
50 (100) |
50 (100) |
|
* p-value <0.05 statistically significant; Chi square / Fisher’s Exact test applied
Feto-maternal Outcome
Figure 1 shows comparison of feto-maternal outcome between Group A & Group B. ICU admission following delivery was seen in 2 patients in group A as compared to 3 patients in group B. However, there was no association found between ICU admission and type of drug administered (p = 1.00). Regarding the gestational age at delivery (Figure 1), term delivery was seen in 34 and 39 patients in group A and B respectively. Preterm delivery was seen in 16 and 11 patients in group A and B respectively. However, the association between mode of delivery, time of delivery status and type of drug administered was not statistically significant (p = 0.26).
IUGR was seen in 12 patients in group A as compared to 15 patients in group B. (Figure 1) However, the association between IUGR status and drug administered was not statistically significant (p = 0.50). 1 patient had intrauterine foetal death (IUFD) in group A as compared to no patient in group B. No association was found between birth status and type of drug administered. (p=1.00)
In both groups, 20 neonates were admitted to NICU following delivery. No statistically significant association was found between NICU admission and type of drug administered (p = 1.00).
Fig.1 :Comparison of feto-maternal outcome
Cost Efficacy
The mean cost requirement in the nifedipine group was 6.84 rupees (6.84 +/- 3.15 SD) which was significantly lower than the mean cost requirement in the labetalol group- 1240 rupees (1240 +/- 762.38 SD). The difference between the two groups was statistically significant (p = 0.001). The cost included tablet cost for nifedipine and injection cost for labetalol.
Discussion
Baseline Characteristics
The difference between two groups in terms of age was not found to be statistically significant (p = 0.84). In a similar study by Biswas SK et al, mean ages for Nifedipine and Labetalol groups were similar (p value=0.54; >0.05). (6) In a similar study by Kaur T et al, the mean age of patients was 25.85±3.10 years and 24.92±3.12 in labetalol group and nifedipine group, respectively, which was quite similar. (7) 28 patients in group A and 30 patients in group B were primigravida, the difference between two groups in terms of parity was also not statistically significant (p = 0.68). Similar findings were noted by Lohnan LC et al where 35 patients (47.3%) were nullipara, and the mean gestational age was 34.3±3.4 weeks with 39 patients (52.7%)being 34 weeks and beyond. (8)
There was no statistically significant difference found between two groups in terms of vital parameters at admission (p >0.05). Baseline characteristics like mean age, mean weight, heart rate, systolic and diastolic pressures were similar in both labetalol and nifedipine groups in a similar study by Biswas SK et al. (6)
Efficacy
The mean number of doses required to control blood pressure in group A was 2.28 (2.28 +/- 1.05 SD) which was lower than the mean number of doses required to control blood pressure in group B- 2.74 (2.74 +/- 1.04 SD). The difference between the two groups was statistically significant (p = 0.03). The average time taken in nifedipine administered patients was 45.60 (45.60 +/- 21.01) minutes while it was 54.80 (54.80 +/- 20.92) minutes in intravenous labetalol. The difference between two groups was found to be statistically significant (p = 0.04).
However, in a study by Wasim et al, it was found that both drugs are equally efficacious to control blood pressure in patients with severe preeclampsia. It was found that labetalol took 22.6± 13.5 minutes and Nifidepine took 22.09± 11.7 minutes to achieve target BP (p>0.05) and whereas labetalol required 2.3± 1.58 doses; Nifidepine required 2.2± 1.58 doses to control BP (p>0.05). (9). Similar findings were observed by Shahnaz et al. (10)
Similar to our study, in a systematic review by Lin L et al, the nifedipine group required fewer doses to achieve the target blood pressure (MD 0.62, 95%CI 0.36 to 0.88, p < .00001) and also patients who received oral nifedipine reached their target blood pressure more rapidly than those who received intravenous labetalol (MD 7.64, 95%CI 4.08–11.20, p < .0001). (11) Similarly, Kumari et al concluded that Oral Nifedipine controls hypertension more rapidly and with fewer doses compared to IV Labetalol. (12) Similar findings were observed by Sahai et al that though both drugs are efficacious, nifedipine controls hypertension more rapidly with less number of doses and was associated with a significant increase in urinary output. (13)
However, Kaur T et al found out that intravenous labetalol is faster in controlling blood pressure in pregnant women with preeclampsia than oral nifedipine and that it may be used as a first-line drug in the acute control of blood pressure in a hypertensive emergency.. (7) Thus more studies are needed to assess which dug is better.
Complications
In the present study, headache was seen in four patients in group A and five patients in group B; which was not statistically significant (p = 1.00). Dizziness was found in 24% of patients in labetalol group as compared to 8% of patients in nifedipine group which was statistically significant. (p = 0.03) Palpitations were present in two patients in group A and no patients in group B, which was not statistically significant (p = 0.49). Tachycardia was present in 12 patients in group A as compared to no patients in group B, which was statistically significant (p = 0.001) with it being more common in patients administered with nifedipine as compared to labetalol. Nausea or vomiting was present in 10 patients in group B as compared to no patients in group A which was again statistically significant (p = 0.03). Breathing difficulty and bradycardia was present in six and eight patients respectively in group B as compared to no patients in group A which was again statistically significant (p = 0.03 and p = 0.001).
No association found between ICU admission and type of drug administered (p = 1.00) (2 in Group A versus 3 in Group B). ICU admission was more related to maternal complications of pre-eclampsia.
Biswas SK et al had similar findings that maternal and neonatal ICU admission, maternal eclampsia, decrease in maternal urine output, maternal heart failure, maternal stroke, maternal nausea and vomiting, maternal dizziness and/headache, maternal palpitation, fetal heart rate abnormality, 5 minutes Apgar score <7 also showed no statistically significant difference between the groups. (6) Kaur et al and Lin L et al also observed no significant difference in adverse effects between the two groups. (7,11) Similarly Kumari et al found no significant difference between side effects and fetomaternal outcome. (12)
Foetal Outcome
IUGR was seen in 12 patients in group A as compared to 15 patients in group B which was not statistically significant (p = 0.50).
One patient had intrauterine foetal death (IUFD) in group A as compared to no patient in group B. There was no association found between birth status and type of drug administered. In both groups, 20 neonates were admitted to NICU following delivery (not statistically significant (p = 1.00)). In a systematic review by Lin L et al, there were no significant difference in the neonatal complications, including NICU admission, 5 min Apgar score < 7, neonatal deaths, FHR abnormality. (11)
Cost-effectiveness
The mean cost requirement in the nifedipine group was 6.84 rupees, and the mean cost requirement in the labetalol group (1240 rupees) which was statistically significant (p = 0.001). In a similar study Azmat B et al the cost of treatment per participant in the IV Labetalol group averaged $550 (46,200 rupees), whereas in the Oral Nifedipine group, it averaged $600 (50,400 rupees). Thus nifedipine is more cost-effective than labetalol. (14)
Conclusion
Our findings indicate that both medications are first-line and effective in reducing blood pressure in severe pre-eclampsia but oral Nifedipine has a quicker onset of action, with lesser number of doses required. Intravenous Labetalol, on the other hand, demonstrated a predictable blood pressure control, making it a good choice for in-hospital management.
Ethical Approval: The Study Was Conducted In Accordance With The Ethical Standards Of The Institutional And/Or National Research Committee And With The 1964 Helsinki Declaration And Its Later Amendments Or Comparable Ethical Standards. The Institutional Ethical Approval Letter For The Study Has Been Attached.
Conflict Of Interest: The Authors Declare That They Have No Conflict Of Interest
Informed Consent: Informed Consent Was Obtained From All Individual Participants Included In The Study
Funding: The Logistic Support Was Provided By MGM Hospital Kamothe, No Other Source Of Funding
References
1. Luger RK, Kight BP. Hypertension In Pregnancy. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430839/
2. Dhinwa M, Gawande K, Jha N, Anjali M, Bhadoria AS, Sinha S. Prevalence of Hypertensive Disorders of Pregnancy in India: A Systematic Review and Meta-Analysis. Journal of Medical Evidence. May-Aug 2021; 2(2): 105-12.
3. Wiles K, Damodaram M, Frise C. Severe hypertension in pregnancy. Clin Med (Lond). 2021 Sep;21(5):e451-e456.
4. Mathew R, Devanesan BP, Srijana, Sreedevi NS. Prevalence of hypertensive disorders of pregnancy, associated factors and pregnancy complications in a primigravida population. Gynecology and obstetrics clinical medicine. 2023; 3(2): 119-23.
5. Beech A, Mangos G. Management of hypertension in pregnancy. Aust Prescr. 2021 Oct;44(5):148-52.
6. Biswas SK, Raha SK. Oral Nifedipine versus Intravenous Labetalol for Acute Blood Pressure Control in Severe Hypertension of Pregnancy: A Study at Faridpur Medical College Hospital. Faridpur Med Coll J. 2021 Oct 3;16(1):25-9.
7. Kaur T, Kumari K, Rai P, Gupta V, Pandey S, Saini S. A Comparative Study of Oral Nifedipine and Intravenous Labetalol for Acute Hypertensive Management in Pregnancy: Assessing Feto-Maternal Outcomes in a Hospital-based Randomized Control Trial. Int J MCH AIDS. 2024 May 31;13.
8. Lohnan LC, Bitrus J, Opara FC, Momoh MC, Oyeniran OF, Chibuzo, S. Comparison of labetalol and nifedipine in control of blood pressure in severe pre-eclampsia in Dalhatu Araf Specialist Hospital. Int J Reprod Contracept Obstet Gynecol. 2025. 14(4):1054–9.
9. Wasim T, Agha S, Saeed K, Riaz A. Oral Nifidepine versus IV labetalol in severe preeclampsia: A randomized control trial. Pak J Med Sci. 2020 Sep-Oct;36(6):1147-52.
10. Shahnaz I , Ali E ,Das S, Hoque IU. A comparative study on efficacy of intravenous labetalol and oral nifedipine for control of blood pressure in severe pre-eclampsia in a tertiary care hospital. Int J Acad Med Pharm.2024; 6 (4); 945-51
11. Li L, Xie W, Xu H, Cao L. Oral nifedipine versus intravenous labetalol for hypertensive emergencies during pregnancy: a systematic review and meta-analysis. J Matern Fetal Neonatal Med. 2023 Dec 15;36(2):2235057.
12. Kumari A, Sinha A. Comparative Study of Efficacy and Safety of Oral Nifedipine and Intravenous Labetalol for Hypertensive Emergencies in Pregnancy. Int J Pharm Clin Res. 2024; 16(7); 1170-4.
13. Sahai R, Nidhi A, Ranjan R, Lal S. Comparative study of oral nifedipine versus intravenous labetalol in severe hypertension in pregnancy: A randomized controlled study. Indian J Obstet Gynecol Res2020; 75-80.
14. Azmat B, Sharma A, Singh P. IntraVenous Labetalol vs Oral Nifedipine in Acute Severe Hypertension of Pregnancy–A Randomized Controlled Trial. J Cardiovasc Dis Res. 2018 April 30; 9: 159-64.
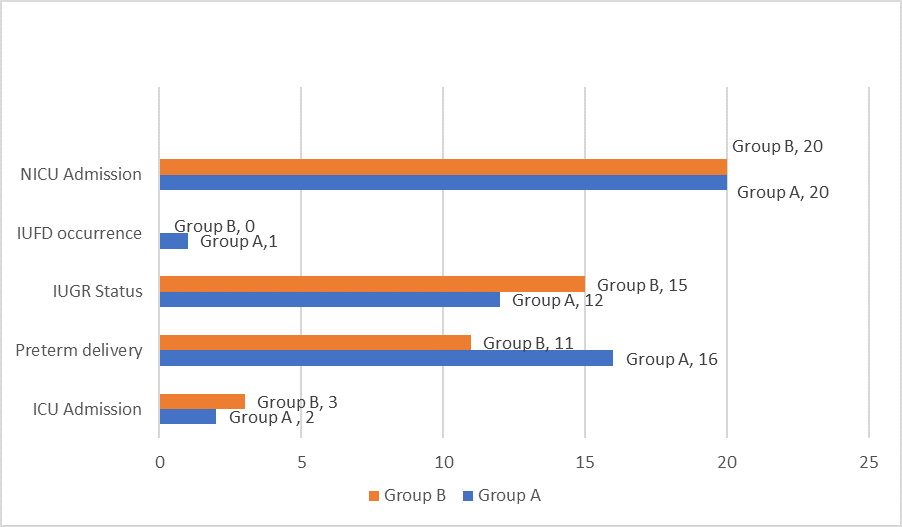
Figure 1
