Follow-up Analysis of the Pulmonary Function Test in COVID-19 Pneumonia after Three and Six-Month Post Recovery.
Follow-up Analysis of the Pulmonary Function Test in COVID-19 Pneumonia after Three and Six-Month Post Recovery.
Bhagwan Mantri 1*, Syed Majid2, Deepika Ughade3, Nivart Shenuoda4, Akash Kashyap5
1. Bhagwan Mantri Associate Professor Pulmonology Department, Subharti Medical College, India.
2. Syed Majid Specialist Internal Medicine IBHO Hospital, Emirates Health services UAE.
3. Deepika Ughade Assistant Professor DY Patil Hospital Mumbai, India.
4. Nivart Shenuoda, Specialist Pulmonologist IBHO Hospital, Emirates Health services UAE.
5. Akash Kashyap, Senior resident Moolchand hospital Delhi.
Correspondence to: Bhagwan Mantri Associate Professor Pulmonology Department, Subharti Medical College, India.
Copyright
© 2024 Bhagwan Mantri. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 02 June 2025
Published: 16 June 2025
Follow-up Analysis of the Pulmonary Function Test in COVID-19 Pneumonia after Three and Six-Month Post Recovery.
Introduction
The World Health Organization (WHO) officially declared the SARS-CoV-2 outbreak a Public Health Emergency of International Concern on January 30, 2020 and a global pandemic on March 11, 2020.1 Earlier in late December 2019, an outbreak of a mysterious pneumonia characterized by fever, dry cough, fatigue, and occasional gastrointestinal symptoms happened in a seafood wholesale wet market, the Huanan Seafood Wholesale Market, in Wuhan, Hubei, China.2 The pathogen of the outbreak was identified as a novel beta-coronavirus, named 2019 novel coronavirus (2019-nCoV) which recalled the terrible memory of the severe acute respiratory syndrome (SARS-2003, caused by another beta-coronavirus) that occurred in 2003.3 On Feb 11, 2020, WHO renamed the disease as coronavirus disease 2019 (COVID-19).That same day, the Coronavirus Study Group (CSG) of the International Committee on Virus Taxonomy posted a manuscript on bioRxiv in which they suggested designating 2019-nCoV as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).4
This novel virus is round or elliptic and often pleomorphic form and a diameter of approximately 60–140 nm,5 genetically similar to bat coronaviruses, and shares about 79% and 50% of its genetic sequence with the coronaviruses responsible for severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS), respectively. Similar to the etiological agents in previous human coronavirus outbreaks (severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS).6,7The World Health Organization (WHO) declared COVID-19 a global pandemic on March 11, 2020. Since then, India has witnessed three waves. Compared to the first wave, the second wave has had severe consequences worldwide regarding cases and mortality. Even in India, mortality was higher in the second wave than in the first wave . As of March 11, 2023, morethan 446 million RT-PCR-confirmed cases had been diagnosed in India, with more than 5.3 lakh deaths attributed to the COVID-19 infection.8
COVID-19 patients may present with a spectrum of symptoms ranging from asymptomatic, mild upper respiratory tract symptoms to severe pneumonia and multiorgan failure. Fever, fatigue, cough and expectoration are the most frequent presenting symptoms, but muscle soreness, anorexia, chest tightness, dyspnea, nausea, vomiting, diarrhea, headache also occurred frequently among Covid-19 patients. The lung is the most common organ affected in SARS-CoV-2 infection. The predominant pattern of lung abnormalities during illness is ground-glass opacity.9,10,11 CoV2 may have a restrictive or obstructive defect on a spirometry during recovery and an abnormal pulmonary function test. Current evidence suggests that pulmonary fibrosis could complicate infection by SARS-COV2. Fibrosis could be viewed as a consequence of a disordered wound healing process and may be directly related to the severity of the illness.
In people recovering from COVID-19, there is concern regarding potential long- term pulmonary sequelae and associated impairment of functional capacity. There is also increasing recognition that between 30–60% of individuals report protracted symptoms, such as fatigue and dyspnea, following resolution of their acute illness; variably termed the ‘long-COVID’ or the post-COVID syndrome. 12,13 Exertional dyspnea and exercise intolerance are prominent clinical features reported in this syndrome,14and there is increasing interest in how best to objectively characterize and followed-up this issue with objective tests. Similar concerns were apparent following the first SARS pandemic with SARS-CoV-1 infection, in 2003. Pulmonary function testing (PFT) in patients following SARS-CoV-1 pneumonitis often revealed impaired in gas transfer indices and a disconnect between this impairment and exercise limitation on cardio-pulmonary exercise testing (CPET). 15 The literature on previous coronavirus infections reports that patients may experience persistent impairment in respiratory function after being discharged. Our objective was to determine the prevalence of restrictive pattern, obstructive pattern and altered diffusion in patient’s post-COVID-19 infection and to describe the different evaluations of respiratory function used with these patients.
.
Material and Methods
The aim of our study was to assess pulmonary function test in patients who had recovered from COVID-19 pneumonia, after discharging the patients, we had planned to assessed FEV1,FVC, FEV1/FVC ,TLCO and DLCO at 3rd and 6th month during OPD follow up.
Table 1 classification restrictive disease
|
FEV1 % predicted |
Stage |
|
>80% |
Mild |
|
50-79% |
Moderate |
|
30-49% |
Severe |
|
<30% |
Very severe |
This prospective cohort study was conducted from November 2020 to March 2022 at Moolchand Hospital, Lajpat Nagar-3, New Delhi, after obtaining approval from the institutional ethics committee. Informed consent was obtained from each patient enrolled in the study. A total of 96 patients were included based on specific inclusion criteria: adult patients diagnosed with COVID-19 who were admitted to the ICU due to hypoxemia, confirmed by HRCT chest, and subsequently discharged after recovery.
Patients with a history of pulmonary parenchymal or airway diseases, such as interstitial lung disease or COPD, were excluded. Following discharge, all patients were scheduled for a follow-up appointment in the outpatient department for pulmonary function testing (PFT). The PFTs were conducted using a standardized PFT machine. Data analysis was performed using SPSS software (version 2022), applying ANOVA for comparisons
Result
Ninety-six patients were included in this study, out of them 51% (n=49) were male and 49% (N= 47) are female. All patient had been distributed in five categories on the basis of age (Table 1). Among 96 patients 3.1% (n=3) belonged to age group of 18-28 years, while in the second group which belong to age group of 29–38-year 25% (n= 24) case were there, age group of 39-48 had 44.8 % (n=43) cases, in fourth group there was 25% (n=24) which belong to age group of 49-59 years, remaining all case included in 5th group.
Fig 1
Clinically patients were divided in three groups with mild ,moderate and severe illness on basis of preset criteria( table of criteria) labelled as group A,B and C respectively. All patient those who have sign and symptom of viral pneumonitis but they did not received supplemental oxygen considered mild infection(Group A), total no of cases were 58.3%(n= 57) in this group, 25% (n= 24 )patient with moderate illness(Group B) required supplementary oxygen but did not receive respiratory support, remaining 16.7% (n= 15) cases required invasive or non-invasive ventilation(Group C).
Table 2: Comparison of FEV1 (raw data) at various follow up group according to Covid grading severity
|
FEV1 |
|
N |
Mean |
Std. Deviation |
|
|
|
|
|
F value |
p value |
post hoc |
||||
|
IMMEDIATE POST RECOVERY |
GROUP A |
56.00 |
2.45 |
0.65 |
67.031 |
.000*, SIG |
A>B>C |
|
GROUP B |
24.00 |
1.49 |
0.14 |
||||
|
GROUP C |
16.00 |
0.96 |
0.19 |
||||
|
Total |
96.00 |
1.96 |
0.79 |
||||
|
THREE MONTH |
GROUP A |
56 |
2.8889 |
.70553 |
43.26 |
0.0001*, SIG |
A>B>C |
|
GROUP B |
24 |
2.0363 |
.37109 |
||||
|
GROUP C |
16 |
1.4981 |
.26423 |
||||
|
Total |
96 |
2.4440 |
.80120 |
||||
|
SIX MONTH |
GROUP A |
51 |
3.4186 |
.84592 |
24.166 |
0.0001*, SIG |
A>B>C |
|
GROUP B |
18 |
2.6600 |
.44998 |
||||
|
GROUP C |
16 |
2.0850 |
.38231 |
||||
|
Total |
85 |
3.0069 |
.88522 |
||||
A pulmonary function test was performed for all patients immediately after recovery but before discharge, repeated PFT was done after three months of discharge, while third PFT performed after six months of discharge. FVC, FEV1, FEV1/FVC DLCO used for pulmonary function test parameters. Immediately after post recovery the mean ± SD FEV1 was 1.96 ± 0.79, after three months it was 2.44
± 0.80, while 3rd PFT at six months after discharge SD FEV1 was 3.01 ± 0.89. This comparison was done by using Anova test, the difference was found to be significant with minimum seen at immediate after Operation recovery with gradually and significantly increased add six-month. Difference was found maximum in group A, which was significantly higher than group B, which was further significantly higher than seen in group C.
Immediate post recovery mean ± SD FVC was 2.58± 0.90(76.97%±17.15), after three months ± SD FVC was 3.09 ±0.87(88.04% ±14.79) while at six months of discharge ± SD FVC showed 4.36±6.83(92.53%±22.34). Before discharge, and at three months the FVC was found to be significantly different across groups of COVID grading severity when compared using one-way Anova test.
Table 3: Comparison of FVC (raw data) at various follow up group according to Covid grading severity
|
FVC |
|
N |
Mean |
Std. Deviation |
|
|
|
|
|
F value |
p value |
post hoc |
||||
|
IMMEDIATE POST RECOVERY |
GROUP A |
56 |
3.0816 |
.81442 |
40.132 |
.000*, SIG |
A>B>C |
|
GROUP B |
24 |
2.0600 |
.29314 |
||||
|
GROUP C |
16 |
1.6144 |
.42228 |
||||
|
Total |
96 |
2.5817 |
.89813 |
||||
|
THREE MONTH |
GROUP A |
56 |
3.5241 |
.80643 |
30.009 |
0.0001*, SIG |
A>B>C |
|
GROUP B |
24 |
2.7038 |
.49180 |
||||
|
GROUP C |
16 |
2.1463 |
.42044 |
||||
|
Total |
96 |
3.0894 |
.87279 |
||||
|
SIX MONTH |
GROUP A |
51 |
4.0212 |
.90990 |
1.279 |
0.284,nS |
NA |
|
GROUP B |
18 |
3.2239 |
.61090 |
||||
|
GROUP C |
16 |
6.7238 |
15.81412 |
||||
|
Total |
85 |
4.3611 |
6.82914 |
||||
Post recover, before discharge mean ± SD FEV1/FVC% predicted was 75.57± 12.07,after three months the mean ± SD FEV1/FVC %was 80.90 ±9.43,while at six month of discharge mean SD FEV1/FVE 5 was 85.11 17.66, the comparison was done by using repeated measure of Anova, the difference was found to be significant, with minimum seen at immediate After discharge with gradual recovery which was significantly increased at six-month.
Table 4: Comparison of FVC (% predicted) at various follow up group according to Covid grading severity
|
FEV1/FVC |
|
N |
Mean |
Std. Deviation |
|
|
|
|
|
F value |
p value |
Post Hoc |
||||
|
IMMEDIATE POST RECOVERY |
GROUP A |
56 |
80.3157 |
6.50014 |
21.844 |
.000*, SIG |
A>B>C |
|
GROUP B |
24 |
73.7433 |
11.99391 |
||||
|
GROUP C |
16 |
61.6956 |
15.87312 |
||||
|
Total |
96 |
75.5693 |
12.06696 |
||||
|
THREE MONTH S |
GROUP A |
56 |
84.5659 |
5.82184 |
14.464 |
0.0001*, SIG |
A>B>C |
|
GROUP B |
24 |
77.6075 |
10.40559 |
||||
|
GROUP C |
16 |
73.0206 |
11.81765 |
||||
|
Total |
96 |
0.9021 |
9.43424 |
||||
|
SIX MONTHS |
GROUP A |
51 |
88.4175 |
13.43963 |
5.043 |
0.009*, sig |
A>B>C |
|
GROUP B |
18 |
86.3439 |
8.99040 |
||||
|
GROUP C |
16 |
73.1594 |
29.35969 |
||||
|
Total |
85 |
85.1062 |
17.66268 |
||||
Post recovery before discharge, the mean ± SD DLCO% predicted was 12.90 ±2.18(55.91%±10.74), after three months the mean ± SD DLCO% was 16.37 ± 2.42(71.09% ±11.67) and after six month of discharge mean ± SD DLCO% predicted was 22.01±5.87(87.54%±8.35). The comparison was done using repeated major of Anova, the difference was found to be significant, with minimum seen at immediately post recovery, which gradually and significantly increased at six- month.
Table 5: Comparison of DLCO (raw data) at various follow up group according to Covid grading severity
|
DLCO |
|
N |
Mean |
Std. Deviation |
|
|
|
|
|
F value |
p value |
Post Hoc |
||||
|
IMMEDIATE POST RECOVERY |
GROUP A |
56 |
13.6154 |
2.12166 |
8.426 |
.000*, SIG |
A>B, C |
|
GROUP B |
24 |
11.9288 |
2.23830 |
||||
|
GROUP C |
16 |
11.8344 |
1.17026 |
||||
|
Total |
96 |
12.8969 |
2.18323 |
||||
|
THREE MONTH S |
GROUP A |
56 |
16.7804 |
2.51482 |
2.128 |
0.125,Ns |
NA |
|
GROUP B |
19 |
15.6695 |
2.58360 |
||||
|
GROUP C |
16 |
15.7825 |
1.50469 |
||||
|
Total |
91 |
16.3730 |
2.41804 |
||||
|
SIX MONTHS |
GROUP A |
47 |
21.7117 |
1.52156 |
1.372 |
0.260,ns |
NA |
|
GROUP B |
17 |
20.8871 |
1.61059 |
||||
|
GROUP C |
16 |
24.0719 |
12.85771 |
||||
|
Total |
80 |
22.0085 |
5.86931 |
||||
Discussion
On March 11, 2020, the World Health Organization (WHO) declared coronavirus disease 2019 (COVID-19) a pandemic, with 20% of infected patients requiring hospitalization and 6% requiring critical care and invasive ventilatory assistance. 16
According to early epidemiological reports, 8.2% of all cases had rapid and progressive respiratory failure, which was similar to acute respiratory distress syndrome (ARDS). 17
COVID-19 is characterized by extensive injury to alveolar epithelial and endothelial cells, as well as secondary fibroproliferation, implying the possibility of chronic vascular and alveolar remodeling, which could lead to lung fibrosis and/or pulmonary hypertension. PFTs, such as spirometry, diffusion capacity, and lung volumes, are the most common types of objective functional respiratory evaluations. 18-20
In people recovering from COVID-19, there was concern regarding potential long- term pulmonary sequelae and associated impairment of functional capacity. Spirometry indices appear to be generally well preserved, but that a defect in diffusing capacity (DLCO) is a prevalent abnormality identified on follow- up lung function; decreased diffusion capacity present in 20–30% of those with mild to moderate disease and 60% in those with severe disease, also reductions in total lung capacity were commonly reported.21 In similar study Devasahayam et.el suggest potential long-term pulmonary sequelae and associated impairment of functional capacity among Indian Patients. In this study they have shown that post- COVID-19 lung damage leads to significant impairment of lung function, quality of life and effort tolerance. 22Julia Tarraso and colleagues in their study concluded that significant percentage of individuals would develop pulmonary sequelae after COVID 19 pneumonia, regardless of severity of the acute process.23
In our study we performed Pulmonary function test for all patients immediately after recovery but before discharge, PFT repeated after three months of discharge, while third PFT performed after six months of discharge. FVC, FEV1, FEV1/FVC DLCO were used for pulmonary function test parameters. Most of the patients who have moderate to severe illness, and they require supplemental oxygen and respiratory support have significant impairment of pulmonary function immediately post recovery, fortunately most of the patients improved clinically and their pulmonary function test showed significant improvement in FVC, FEV1, FEV1/FVC DLCO24.
Our study showed that lung function measured by spirometry was lower Immediately post discharge and gradually showed improvement in repeat spirometry 3 Months and 6 months post recovery. Our results were supported by the previous studies indicating that the spirometry values including %predicted of FVC and FEV1 showed a gradual Improvement in post COVID-19 pneumonia in 1 Year follow up
The strength of our study is that we have studied follow up of the diffusion capacity of lungs which also showed significant improvement after 3 months and 6 months post covid 19 recoveries, which emphasize on fact that early CT Chest findings which was suggestive of early fibrosis is uncertain though we did not follow these patients with CT Scan.
However, this study has certain limitations. Firstly, it was conducted at a single center, which may limit the generalizability of the results to other settings or populations. Secondly, we assessed outcomes only up to 6 months post-recovery. To better understand the long-term effects of post-COVID-19 pneumonia, a longer follow-up period—ideally up to two years—would be necessary.
Limitation
This study has certain limitations. Firstly, it was conducted at a single center, which may limit the generalizability of the results to other settings or populations. Secondly, we assessed outcomes only up to 6 months post-recovery. To better understand the long-term effects of post-COVID-19 pneumonia, a longer follow-up period—ideally up to two years—would be necessary And age and gender match control group could have lucidly provided better understanding of PFT in these cohort covid-19 group.
Conclusion
This study showed that Severe COVID-19 patients had a higher rate of abnormal spirometry when compared to patients with mild symptoms .6 Months follow-up study in COVID-19 pneumonia Patients, we demonstrated a gradual improvement in spirometry FEV1, FVC, and DLCO over time; and, this improvement was more significant in severe category. We recommend a follow-up program for all post COVID-19 pneumonia patients for early detection of any complications in the lung function, and to find the appropriate way for rehabilitation and treatment
References
1. World Health Organization WHO Announces COVID-19 Outbreak a Pandemic http://www.euro.who.int/en/heps//:coronavirus.jhu.edu/map.alth-topics/health- emergencies/coronavirus-covid-19/news/news/2020/3/who-announces-covid-19- outbreak-a-pandemic (Accessed 1 May 2020) March 12.
2. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020 DOI: 10.1016/S0140- 6736(20)30183-5.
3. J Chin Med Assoc. 2020 Mar; 83(3): 217–220. Published online 2020 Feb 12. doi: 10.1097/JCMA.0000000000000270 PMCID: PMC7153464 PMID: 32134861
4. The outbreak of COVID-19: An overview Yi-Chi Wu,a Ching-Sung Chen,a and Yu-Jiun Chana,b,c,*
5. Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020;382:727–33.
6. Guan W.-j., Ni Z.-y., Hu Y., et al. Clinical characteristics of 2019 novel coronavirus infection in China. 2020: https://doi.org/10.1101/2020.02.06.20020974.
7. Fu L, Wang B, Yuan T, Chen X, Ao Y, Fitzpatrick T, Li P, Zhou Y, Lin YF, Duan Q, Luo G, Fan S, Lu Y, Feng A, Zhan Y, Liang B, Cai W, Zhang L, Du X, Li L, Shu Y, Zou H. Clinical characteristics of coronavirus disease 2019 (COVID-19) in China: A systematic review and meta-analysis. J Infect. 2020 Jun;80(6):656-665.
8. Government of India: #IndiaFightsCorona COVID-19. [ Mar; 2023 ]. 2023. https://www.mygov.in/covid-19
9. Huang Y, Tan C, Wu J, Chen M, Wang Z, Luo L, Zhou X, Liu X, Huang X, Yuan S, Chen C. Impact of coronavirus disease 2019 on pulmonary function in early convalescence phase. Respiratory research. 2020 Dec;21(1):1-0.
10. Liang L, Yang B, Jiang N, Fu W, He X, Zhou Y, Ma WL, Wang X. Three-month follow-up study of survivors of coronavirus disease 2019 after discharge. Journal of Korean medical science. 2020 Dec 7;35(47).
11. Torres-Castro R, Vasconcello-Castillo L, Alsina-Restoy X, Solis-Navarro L, Burgos F, Puppo H, Vilaró J. Respiratory function in patients post-infection by COVID-19: a systematic review and meta-analysis. Pulmonology. 2021 Jul-Aug;27(4):328-337.
12 T. Sonnweber, S. Sahanic, A. Pizzini, A. Luger, C. Schwabl, B. Sonnweber, et al. Cardiopulmonary recovery after COVID-19 – an observational prospective multi-center trial Eur Respir J, 57 (2020)
Available from: http://erj.ersjournals.com/lookup/doi/10.1183/13993003.03481-2020
13 B. Van den Borst, J. Peters, M. Brink, Y. Schoon, C. Bleeker-Rovers, H. Schers, et al. Comprehensive health assessment three months after recovery from acute COVID-19 Ann Med, 0 (2020), pp. 1-14, 10.1080/07853890.2020.1840620
14. P. Clavario, V. de Marzo, R. Lotti, C. Barbara, A. Porcile, C. Russo, et al.
Assessment of functional capacity with cardiopulmonary exercise testing in non-severe COVID-19 patients at three months follow-up medRxiv (2020), 10.1101/2020.11.15.20231985
15. H. Ahmed, K. Patel, D. Greenwood, S. Halpin, P. Lewthwaite, A. Salawu, et al.
Long-term clinical outcomes in survivors of severe acute respiratory syndrome and Middle East respiratory syndrome coronavirus outbreaks after hospitalization or ICU admission: a systematic review and meta-analysis J Rehabil Med, 52 (2020)
16. Namendys-Silva ˜ SA. ECMO for ARDS due to COVID-19. Hear Heart Lung. 2020;49:348---9
17. M o X, Jian W, Su Z, Chen M, Peng H, Peng P, et al. Abnormal pulmonary function in COVID-19 patients at time of hospital discharge. Eur Respir J. 2020;55:2001217.
18. Venkataraman T, Frieman MB. The role of epidermal growth factor receptor (EGFR) signaling in SARS coronavirus-induced pulmonary fibrosis. Antiviral Res. 2017;143:142- 50
19. Pellegrino R, Viegi G, Brusasco V, Crapo RO, Burgos F, Casaburi R, et al. Interpretative strategies for lung function tests. Eur Respir J. 2005;26:948---68.
20. Bao C, Liu X, Zhang H, Li Y, Liu J. Coronavirus Disease 2019 (COVID-19) CT Findings: a systematic review and meta-analysis. J Am Coll Radiol. 2020;17:P701---709. 9. British Thoracic Society. British Thora
21. https://www.sciencedirect.com/science/article/pii/S2468867321000407
22. Devasahayam Jesudas ChristopherID 1*, Barney T. J. Isaac1, Flavita Benna John1, Deepa ShankarID 2, Prasanna Samuel3, Richa Gupta4, Balamugesh ThangakunamID
23. 1 year after COVID-19: a prospective follow-up Julia Tarraso1, Belen Safont1, Juan A. Carbonell?Asins2, Estrella Fernandez?Fabrellas3, José N. Sancho?Chust4, Elsa Naval5, Beatriz Amat6, Susana Herrera7, José A. Ros8, Juan J. Soler?Cataluña9, Jose A. Rodriguez?Portal10, Ada L. Andreu11, Margarita Marín12, Juan L. Rodriguez?Hermosa13, Cruz Gonzalez?Villaescusa1, Joan B. Soriano14, Jaime Signes?Costa1* and COVID-FIBROTIC study team
24. COVID-19 and the effects on pulmonary function following infection: A retrospective analysis
Author links open overlay panel Kristyn L. Lewis, Scott A. Helgeson, Mehmet M. Tatari, Jorge M. Mallea, Hassan Z. Baig, Neal M. Patel
25. Two-year diffusion capacity trajectory in COVID-19 pneumonia survivors
Marlise P de Roos 1,?, Rick M Heijnen 1, Nynke G Dijkstra 1, Kees Brinkman 2, Paul Bresser 3.
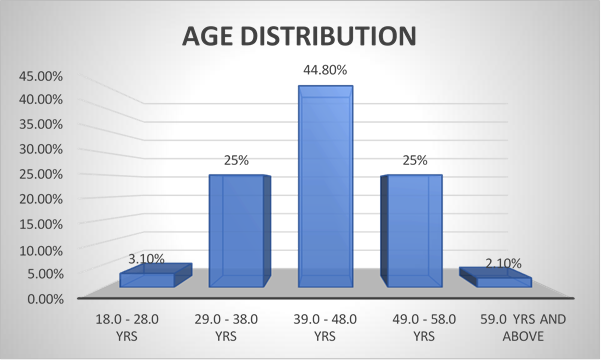
Figure 1
