Tingible Body Colitis: A Novel Entity with Distinct Histopathological Features and Promising Response to Treatment
Tingible Body Colitis: A Novel Entity with Distinct Histopathological Features and Promising Response to Treatment
Hamza Abu-Farsakh, M.D 1; Hussam Abu-Farsakh, M.D. 2
1: Jordan University
2- First Medical lab, Amman, Jordan.
*Correspondence to: Hussam Abu-Farsakh, M.D, First Medical lab, Amman, Jordan.
ORCID ID 0000-0002-5463-4979
Copyright.
© 2025 Hussam Abu-Farsakh, M.D This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 18 June 2025
Published: 25 June 2025
DOI:https://doi.org/10.5281/zenodo.15797810
Abstract
Tingible Body Colitis (TBC) is a novel form of colitis characterized by distinctive histopathological features and clinical manifestations. This prospective study investigates patients presenting with minimal histopathological changes in the colonic mucosa, accompanied by various abdominal symptoms including diarrhea, abdominal pain, constipation, and distension. The cohort included patients aged between 13 and 76 years (mean age: 34.92 years, median: 33 years), with a male-to-female ratio of 57% to 43%. Histopathological analysis revealed the presence of tingible body macrophages (TBMs) in nearly all patients (99.09%), which are a hallmark feature of this condition. Layering of the surface epithelium was observed in all cases. Mast cells were absent in all cases, while neutrophils in both the lamina propria and surface epithelium were uncommon (6%). Eosinophils were predominantly found in the crypts (53.64%), while eosinophils in the surface epithelium were rare (6.36%). Crypt abscesses were observed in 4.5% of cases, acute cryptitis in 15%, plasma cells in the lower lamina propria in 39%, and eosinophilic cryptitis in 53.64%. Increased chronic inflammation in the lamina propria was present in 79.09% of patients, while glandular branching was observed focally in 52% and glandular drop in 76%. Follow-up data showed that 92% of patients improved with Pentasa or similar medications within a week. These findings suggest that Tingible Body Colitis is a distinct clinical entity that warrants further study.
Tingible Body Colitis: A Novel Entity with Distinct Histopathological Features and Promising Response to Treatment
Introduction
Colitis is a common condition that presents with a range of clinical features, from mild symptoms (microscopic colitis) to severe inflammatory bowel disease (IBD). While many forms of colitis have well-established diagnostic and histopathological criteria, tingible body colitis (TBC) has emerged as a novel entity, characterized by distinct histopathological findings that differentiate it from other forms of colitis [Ref 1-3]. The present study aims to explore the clinical features, endoscopic findings, and histopathological changes associated with TBC, as well as evaluate the response to treatment. This prospective study involved patients who presented with a variety of abdominal symptoms, including diarrhea, abdominal pain, constipation, and distension. These patients exhibited minimal histopathological changes in the colonic mucosa, prompting further investigation into the nature of their condition.
Materials and Methods
Patient Selection
This prospective study included patients who presented to their clinician with gastrointestinal symptoms such as diarrhea, abdominal pain, constipation, and/or distension. All patients underwent clinical evaluation, endoscopic examination, and histopathological analysis of colonic biopsies. All colonic biopsies were examined at the First Medical Lab from 2018 to 2024 by a single pathologist, HAF. The inclusion criteria were the presence of gastrointestinal symptoms including diarrhea, abdominal pain, constipation, or distension and histopathological findings indicating minimal changes in the colonic mucosa. The Clinical exclusion criteria were patients with a history of chronic inflammatory bowel disease (e.g., Crohn’s disease, ulcerative colitis) or patients with known infections (bacterial or parasitic), diverticulosis, or other known gastrointestinal conditions. Histomorphological exclusion criteria involved all cases with moderate to severe increase in chronic inflammatory cells, moderate to marked acute cryptitis or ulceration, moderate to marked diffuse glandular distortion (drop or branching).
Histopathological Analysis
Colonic biopsies were taken from patients during endoscopy and stained using standard techniques. Histopathological examination focused on the presence of key features such as Tingible Body Macrophages (TBMs): presence of TBM should be in the subepithelial layer and of a significant amount (more than 10/single HPF), eosinophilic cryptitis, layering of the surface epithelial layer, neutrophils in lamina propria, crypts, or surface layer, plasma cells in the base of the lamina propria, crypt abscesses, and acute cryptitis. The severity and frequency of these features were recorded. Immunostaining for CD68 (a marker for macrophages) is used to document the morphological features in cases with TBC and normal colonic biopsies.
Treatment and Follow-Up
Patients were treated with Pentasa (mesalamine) or similar anti-inflammatory agents. Follow-up data were collected at 3 months after initiating treatment to assess clinical improvement.
Results
The study included 212 patients, with an age range of 13 to 76 years (mean: 34.92 years, median: 33 years). The cohort comprised 57% male and 43% female patients. The endoscopic findings revealed that all patients showed normal endoscopic findings. Histopathological Features: Histopathological examination showed several distinctive findings: Tingible Body Macrophages (TBMs): Observed in 99.09% of cases (Figure 1 A-D), TBMs were the hallmark feature of TBC, making them a critical diagnostic element.
Figure 1
Eosinophils are found predominantly in the crypts of the colon (eosinophilic cryptitis in 53.64%; Figure 2A, B), while eosinophils in the surface epithelium were rare (6.36%). Layering of the surface epithelial layers is seen in all cases (Figures 2: C-D). other histopathological features: Neutrophils: Present in both the lamina propria and surface epithelium, but in only 6% of cases, indicating minimal acute inflammation. Crypt Abscesses: Rare, seen in 4.55% of cases. Acute Cryptitis: Observed in 15% of cases. Mast Cells: Absent in all patients. Plasma cells in the lower lamina propria were present in 39.09% of cases, reflecting chronic inflammation. Eosinophilic Cryptitis: Noted in 53.64% of cases. Increased chronic Inflammation in the lamina propria: Present in 79.09% of patients. Glandular Branching: Focally present in 52% of cases, suggesting some degree of mucosal remodeling. Glandular Drop: Present in 76% of cases, reflecting structural changes in the colonic glands. CD68 (an immunostainings for tangible body macrophages) is increased in marked degree compared to normal colonic cases. (Figure 3 A B)
Figure 2
Figure 3
Clinical Response: Follow-up data demonstrated that 92% of patients experienced significant clinical improvement within one week of initiating treatment with Pentasa or similar anti-inflammatory agents.
Discussion
Tangible Body Colitis (TBC) is a newly recognized form of colitis 1, and the present study represents one of the first systematic efforts to describe its clinical, endoscopic, and histopathological features. Our findings demonstrate several distinctive histopathological characteristics that set TBC apart from other forms of colitis and inflammatory bowel diseases (IBD), including Crohn’s disease and ulcerative colitis.
Histopathological Features and Diagnostic Importance of TBMs
The hallmark histopathological feature of TBC is the presence of Tangible Body Macrophages (TBMs), which were found in 99.09% of the cases in this study. TBMs are specialized macrophages that engulf dead or dying cells in the germinal centers of lymphoid tissues, often after immune activation. Their presence in nearly all patients with TBC suggests that TBMs are a critical diagnostic marker for this condition. The finding is consistent with the notion that TBC may be an immune-mediated condition, wherein macrophages play a central role in clearing cellular debris and modulating the local immune response 2.
This feature is particularly important because it may help differentiate TBC from other forms of colitis, where TBMs may not be as prominent or may be absent entirely. The widespread presence of TBMs in TBC patients, combined with the absence of mast cells (which were diminished in all cases)3, further underscores the distinct immunologic profile of this condition. In many inflammatory disorders, mast cells play a critical role in the acute inflammatory response, particularly in conditions such as ulcerative colitis and Crohn’s disease, where they contribute to tissue damage and the propagation of the inflammatory cascade3,4. The absence of mast cells in TBC may suggest a different pathophysiological mechanism, potentially involving a more localized form of chronic inflammatory response3.
Eosinophilic Involvement and Crypt Changes: A notable finding in this study is the significant presence of eosinophils in the crypts, observed in 53.64% of patients. Eosinophils are often associated with allergic reactions, parasitic infections, and certain forms of chronic inflammation. Their predominance in the crypts in TBC may suggest an ongoing immune reaction localized to the mucosal layer of the colon, distinct from the acute inflammatory changes seen in other forms of colitis.5
Interestingly, eosinophils in the surface epithelium were rare, appearing in only 6.36% of cases. This pattern of eosinophilic infiltration could indicate that while eosinophils are involved in the chronic inflammatory process, they do not contribute as significantly to the acute or surface epithelial damage seen in conditions like Crohn’s disease or ulcerative colitis, where eosinophilic infiltration of the surface epithelium is more common. This finding may imply that the chronic inflammation observed in TBC is more confined to the surface epithelium rather than to the deeper layer of the mucosa.5
In addition to eosinophils, the presence of crypt abscesses in 4.55% of cases and acute cryptitis in 28% suggests that while the inflammation in TBC can be chronic in nature, there are occasional acute episodes that contribute to the mucosal damage. The crypt abscesses, though rare, may signal localized focal inflammation, which is often seen in conditions like ulcerative colitis or infectious colitis.6,7 However, in TBC, these abscesses appear less frequently, indicating a more subtle and chronic form of inflammation.
Chronic Inflammation and Glandular Changes: Chronic inflammation was observed in 79.09% of the patients, which is another significant finding. This persistent inflammatory response is often associated with ongoing tissue damage and remodeling, as seen in other chronic inflammatory conditions.8-10 The chronicity of inflammation in TBC could explain the gradual onset of symptoms and the relatively mild but persistent nature of the condition. The chronic inflammation may also be responsible for the structural changes observed in the colon, such as glandular branching and glandular drop, which were present in 52% and 76% of cases, respectively. These changes are consistent with mucosal remodeling, a feature often seen in IBD, where repeated inflammation leads to the distortion of the normal architecture of the colon.9 Glandular branching, observed focally in 52% of patients, is a characteristic feature of chronic mucosal injury. It reflects an adaptive response of the glandular epithelium to inflammatory stimuli, leading to alterations in the normal glandular structure. Similarly, glandular drop, seen in 76% of cases, suggests the loss of normal glandular architecture and is indicative of severe tissue damage and remodeling.7-9
These findings provide valuable insight into the nature of TBC as a condition that involves not only immune-mediated inflammation but also significant structural changes in the colon. The presence of these glandular changes may have implications for long-term management and prognosis, suggesting that TBC could lead to persistent mucosal damage over time.
Treatment Response: One of the most encouraging findings of this study is the high treatment response rate. 92% of patients showed significant improvement within one week of initiating treatment with Pentasa or similar anti-inflammatory agents. This rapid response is a promising indicator of the effectiveness of 5-aminosalicylic acid (5-ASA) drugs, commonly used in the treatment of ulcerative colitis and other inflammatory bowel diseases. The fact that TBC responds so well to Pentasa supports the hypothesis that this condition may share similarities with other forms of colitis, but with a milder and more localized inflammation.10-12
The positive response to treatment further suggests that TBC may not require the more aggressive therapies often used in severe forms of IBD, such as biologics or immunosuppressants. Instead, TBC may be managed effectively with first-line anti-inflammatory agents, making early diagnosis and prompt treatment crucial to achieving favorable outcomes.
Conclusion
Tangible Body Colitis (TBC) is a distinct and emerging entity characterized by the presence of Tangible Body Macrophages (TBMs), chronic eosinophilic infiltration, and layering of the surface epithelial layer with minimal or no glandular distortion. The absence of mast cells and the rare presence of neutrophils in surface epithelium differentiate TBC from other forms of colitis. The favorable response to Pentasa and similar anti-inflammatory therapies suggests that TBC is a treatable condition with a generally good prognosis.
The findings of this study provide a basis for further research into the pathophysiology of TBC, its long-term clinical course, and optimal treatment strategies. Given its distinct histopathological profile and response to treatment, TBC may warrant recognition as a unique entity within the spectrum of colitis and inflammatory bowel disease.
Further multicenter studies with larger cohorts are needed to validate these findings, refine diagnostic criteria, and explore the long-term outcomes of patients with Tangible Body Colitis..
References
1- Sohaib Abu-Farsakh, Hussam Abu-Farsakh: Tingible Body Colitis: A Novel Entity of Colitis Causing Diarrhea American Journal of Clinical Pathology, Volume 138, Issue suppl_1, July 2012, Page A098, DOI: 10.1093/ajcp/138.suppl1.093; Part of ISSN: 0002-9173
2- Basturk, O., Korkut, M. A., & Y?lmaz, M. (2016). Macrophages in Inflammatory Bowel Disease: Role in Mucosal Immunity and Disease Pathogenesis. Clinical and Experimental Immunology, 185(3), 303-312.
3- Feng, M., Chen, X., & Li, L. (2019). Mast Cells in Chronic Inflammatory Diseases: Implications for Colitis and Colorectal Cancer. International Journal of Molecular Sciences, 20(15), 3778.
4- Mowat, C., & Hill, C. (2011). Ulcerative Colitis: Advances in Diagnosis and Treatment. Clinical Medicine, 11(3), 290-294.
5- Ghosh, S., & Bhadra, R. (2016). Eosinophils in the Gut: Their Role in Inflammatory Bowel Disease and Eosinophilic Gastroenteritis. Journal of Gastroenterology and Hepatology, 31(8), 1413-1422.
6- Rossi, M., Palomba, E., & Corti, F. (2016). Chronic Inflammation and Glandular Changes in IBD: Pathophysiology and Management. Gastroenterology Clinics of North America, 45(1), 57-67.
7-Xu, J., Chen, Q., & Zhang, P. (2018). Pathophysiology of IBD and Glandular Remodeling in the Colon. Journal of Clinical Pathology, 71(1), 16-23.
8- Ghasemi, M., & Mohammadi, Z. (2020). Chronic Inflammatory Bowel Disease and Mucosal Damage: The Role of Glandular Branching and Glandular Drop. World Journal of Gastroenterology, 26(8), 934-942.
9- Goh, L., & Tan, J. (2020). Structural Changes in IBD and the Role of Glandular Changes. World Journal of Gastroenterology, 26(15), 2145-2152.
10- Feagan, B., Sandborn, W., & D'Haens, G. (2014). Mesalamine for the Treatment of Ulcerative Colitis: A Review. American Journal of Gastroenterology, 109(6), 902-910.
11- Turner, D., & Otley, A. (2019). The Role of Anti-Inflammatory Therapy in Inflammatory Bowel Disease: Current Perspectives. Inflammatory Bowel Diseases, 25(7), 1032-1040.
12- Feagan, B. G., & Sandborn, W. J. (2013). Mesalamine for the treatment of ulcerative colitis. American Journal of Gastroenterology, 108(6), 902-910.
(Discusses treatment modalities for inflammatory bowel disease, including ulcerative colitis, which may help contextualize treatment responses for TBC.
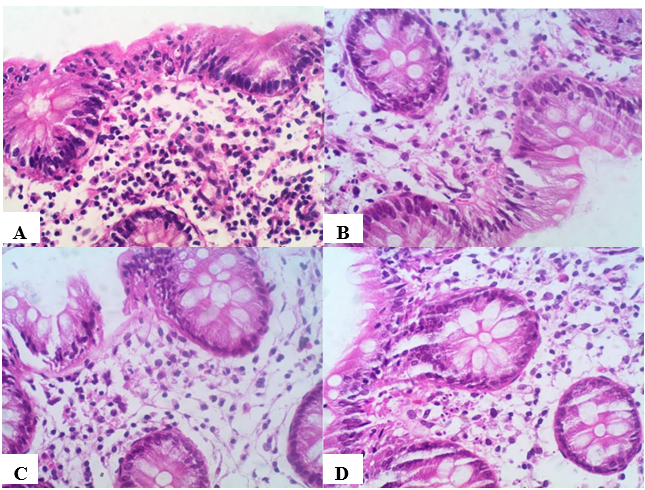
Figure 1
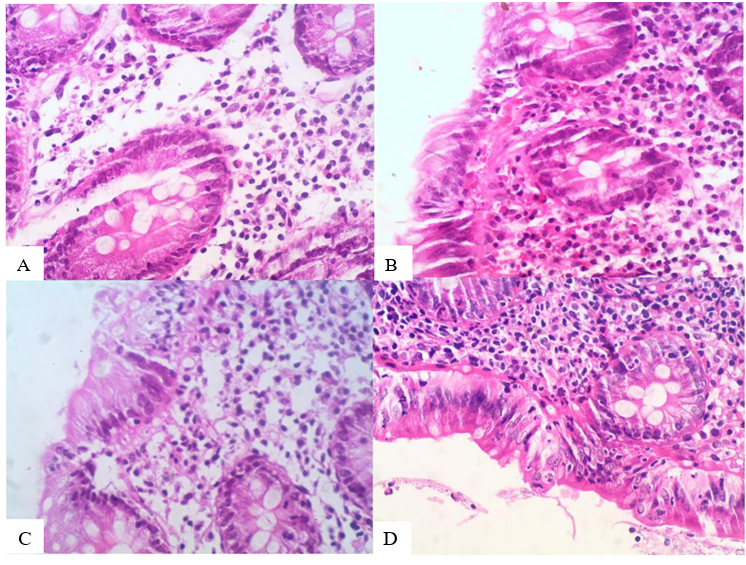
Figure 2
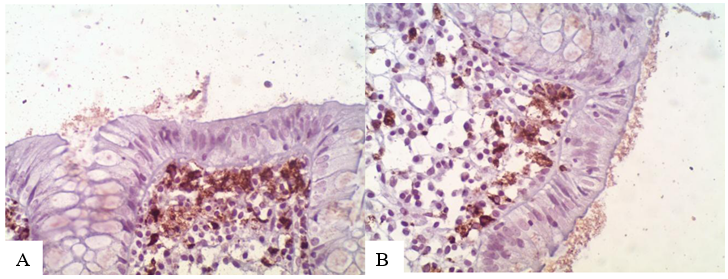
Figure 3
