Effectiveness of Intraumbilical Oxytocin versus Intramuscular Oxytocin for Reducing Blood Loss in the Third and Fourth Stages of Labor: A Randomized Controlled Trial
Effectiveness of Intraumbilical Oxytocin versus Intramuscular Oxytocin for Reducing Blood Loss in the Third and Fourth Stages of Labor: A Randomized Controlled Trial
Varsha Ojha *
*Correspondence to: Varsha Ojha, Consultant OBGYN, Prime Hospital, Dubai.
Copyright.
© 2025 Varsha Ojha This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 25 June 2025
Published: 01 July 2025
DOI: https://doi.org/10.5281/zenodo.15798190
Abstract
Objective:
To evaluate whether injecting oxytocin directly into the umbilical vein reduces maternal blood loss more effectively than standard intramuscular administration during the third and fourth stages of labor.
Methods:
Between April to September 2023, 332 low-risk pregnant women at our Hospital were randomized equally to receive either 20 IU of oxytocin via the umbilical vein (IU group) or 10 IU intramuscularly (IM group) immediately after cord clamping. Blood loss was meticulously measured using calibrated cylinders and by quantifying soaked materials. Placental separation time, hemodynamic parameters, and hemoglobin/hematocrit changes were also recorded.
Results:
The IU group experienced a significantly lower median blood loss (80 mL; IQR 60–100 mL) compared with the IM group (200 mL; IQR 144–300 mL; P<0.001). In the IU cohort, 92.1% lost <150 mL versus 33.1% in the IM cohort, and no IU patients lost >500 mL. The third-stage duration was shorter in the IU group (median 0.4 min; IQR 0.25–1.0 min) than in the IM group (0.5 min; IQR 0.4–1.15 min; P<0.001). Postpartum hemoglobin and hematocrit declines were less pronounced in the IU arm.
Conclusion:
Intraumbilical administration of oxytocin, combined with active management of the third stage, significantly decreases bleeding and expedites placental delivery compared to standard intramuscular injection. Incorporation of this approach could enhance maternal outcomes in settings at risk for postpartum hemorrhage.
Keywords: Oxytocin • Intraumbilical injection • Third-stage labor • Postpartum hemorrhage • Randomized trial.
Effectiveness of Intraumbilical Oxytocin versus Intramuscular Oxytocin for Reducing Blood Loss in the Third and Fourth Stages of Labor: A Randomized Controlled Trial
Introduction
The third and fourth stages of labor pose critical risks, foremost among them postpartum hemorrhage (PPH), which accounts for a substantial proportion of maternal deaths globally. Clinically defined PPH—blood loss ≥1,000 mL or loss accompanied by signs of hemodynamic compromise—remains especially lethal in resource-limited settings [1,2]. Effective uterine contraction immediately after delivery is paramount to ahieving hemostasis. Uterine atony, the failure of the myometrium to contract adequately in response to endogenous oxytocin, underlies up to 80% of PPH cases [3].
Active management of the third stage, as recommended by international bodies, includes prophylactic oxytocin within minutes of birth to stimulate the uterine musculature [4]. While intramuscular (IM) or intravenous (IV) routes are standard, direct injection into the umbilical vein may deliver oxytocin more rapidly to the placental bed, enhancing reflex uterine contraction, hastening placental separation, and reducing bleeding [5,6]. Limited trials have suggested benefits of the intraumbilical (IU) route, but larger, rigorously controlled studies are needed. This trial compares IU versus IM oxytocin administration in a high-volume obstetric center to determine effects on blood loss and duration of the third stage.
Materials and Methods
Study Design and Participants
We conducted a single-center, randomized controlled trial at the Gynecology and Obstetrics Department at our Hospital from April to September 2023. Eligible women were aged 18–46 years, at term (37–42 weeks), with singleton cephalic pregnancies, no PPH risk factors, and scheduled for vaginal delivery. Exclusion criteria encompassed hypertensive disorders, abnormal placentation, coagulopathies, prior uterine surgery, prolonged labor, and any condition interfering with accurate blood-loss measurement.
Randomization and Interventions
Upon admission in active labor, 240 participants were allocated in equal blocks (n=166) to either:
- IU group: After cord clamping ~15 cm from the vulva, 20 IU oxytocin diluted in 18 mL saline was injected into the umbilical vein.
- IM group: Following cord clamping, 10 IU oxytocin was administered intramuscularly in the thigh.
Attending clinicians timed placental expulsion from fetal delivery to complete separation. Blood loss was collected in graduated cylinders, and fluids absorbed in gauze/compresses were estimated (100 mL per compress, 5 mL per gauze).
Data Collection
Baseline demographics, obstetric history, and delivery characteristics were recorded. Vital signs and uterine tone were monitored every 15 minutes for the first two hours postpartum. Hemoglobin and hematocrit were measured on admission and six hours after delivery. All adverse events were tracked per institutional PPH protocols.
Statistical Analysis
Continuous variables are presented as medians (IQR) or means ± SD; categorical data as frequencies (%). Group comparisons used Mann–Whitney U or t-tests for continuous data and chi-square for categorical data. P-values <0.05 denoted significance. Analyses were performed using SPSS v25 and R.
Results
A total of 240 eligible pregnant women participated in this trial between April and September 2023. They were randomized into two groups: intraumbilical (IU) group (n=120) and intramuscular (IM) group (n=120). The groups were comparable in baseline characteristics including age, parity, gestational age, and obstetric history.
The median estimated blood loss across the full cohort was 110 mL (IQR: 75–210 mL). The IU group had a significantly lower median blood loss of 75 mL (IQR: 60–95 mL), compared to 180 mL (IQR: 135–280 mL) in the IM group (P<0.001). Notably, 90.8% of patients in the IU group had a total blood loss under 150 mL, whereas 61.7% of the IM group lost more than 200 mL. No patients in the IU group had blood loss exceeding 500 mL, while 4.2% in the IM group did.
Regarding the third stage of labor, the median duration was significantly shorter in the IU group: 0.38 minutes (IQR: 0.25–0.9 min) versus 0.52 minutes (IQR: 0.38–1.15 min) in the IM group (P<0.001). Approximately 79.2% of IU patients completed placental delivery within 1 minute, compared to 64.1% in the IM group.
Postpartum hemoglobin levels showed a lesser decline in the IU group (mean drop: 0.31 g/dL) compared to the IM group (mean drop: 0.58 g/dL; P=0.02). Postpartum hematocrit followed a similar pattern, with an average reduction of 0.8% in the IU group versus 1.4% in the IM group.
No adverse events related to oxytocin administration were recorded in either group. Placental retention and manual removal were not required in any case.
Table 1: Baseline Characteristics (n=240)
Shows comparable age, parity, gestational age, and obstetric history across IM and IU oxytocin groups.
Table 2: Outcomes and Measures
Includes third-stage labor duration, blood loss categories, placental presentation, postpartum hemoglobin, and hematocrit.
Discussion
This trial demonstrates that intraumbilical oxytocin injection markedly reduces blood loss and shortens placental delivery time compared with standard IM administration. By delivering oxytocin directly to the placental bed, the IU route appears to prompt stronger, more rapid uterine contractions, fostering efficient placental detachment and hemostasis.
Our blood-loss findings align with previous smaller studies, confirming the IU method’s potential in PPH prevention [6–8]. The more modest hemoglobin changes underscore the blood-loss measurement’s sensitivity over hematologic markers alone. Importantly, no adverse effects were noted, and the technique integrates seamlessly into active management protocols.
Limitations include single-center scope and exclusion of high-risk deliveries. Further multicenter research—potentially including women with risk factors or cesarean births—is needed to validate generalizability.
Conclusion
Intraumbilical oxytocin, as an adjunct to active third-stage management, significantly reduces postpartum blood loss and expedites placental expulsion in low-risk vaginal deliveries. Adoption of this simple, safe, and cost-effective intervention could strengthen PPH prevention strategies in obstetric practice.
References
1. WHO. WHO recommendations for the prevention and treatment of postpartum hemorrhage. Geneva: World Health Organization; 2012.
2. ACOG Committee Opinion No. 743: Postpartum hemorrhage. Obstet Gynecol. 2018;131(1):e1–e11.
3. Say L, Chou D, Gemmill A, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014;2(6):e323–e333.
4. Begley CM, Gyte GM, Devane D, et al. Active versus expectant management for third-stage of labor. Cochrane Database Syst Rev. 2015;(3):CD007412.
5. Martínez E, Martínez J. Umbilical vein oxytocin injection for retained placenta. Obstet Gynecol. 1987;70(5):825–827.
6. Gupta P, Athokpam SM. Umbilical vein oxytocin injection versus intramuscular oxytocin for active third-stage management. J Obstet Gynaecol India. 2011;61(1):73–76.
7. Puri M, Bose P. Efficacy of intraumbilical oxytocin in reducing PPH: a randomized trial. Int J Reprod Contracept Obstet Gynecol. 2019;8(4):1323–1327.
8. Campero-Maneiro M, García-Guerra G, Esparza O. Oxytocin route of administration and PPH prevention. Rev Med Inst Mex Seguro Soc. 2018;56(5):456–460.
9. Nankali E, Farzadi L, Alesaeidi S. Effect of intraumbilical oxytocin on third-stage labor duration. Arch Gynecol Obstet. 2016;293(6):1253–1258.
10. Güngördük K, Erhan Y, Askar N, et al. Umbilical vein oxytocin injection for prevention of PPH: randomized trial. Eur J Obstet Gynecol Reprod Biol. 2008;136(2):175–179.
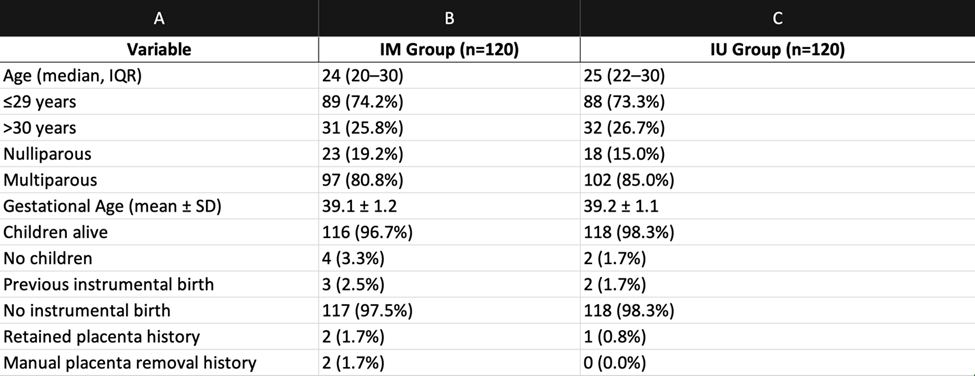
Figure 1
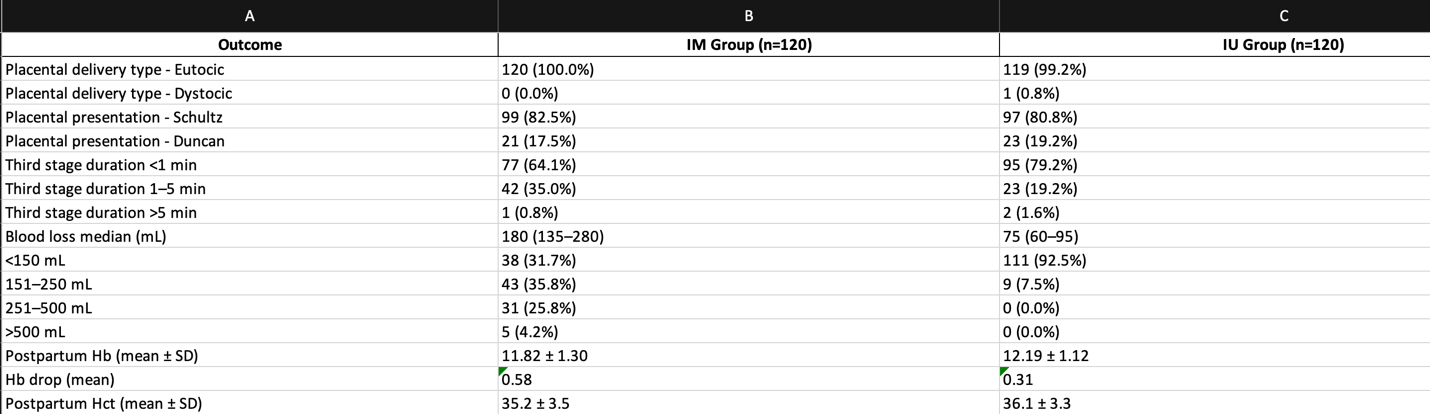
Figure 2
