EUS Guided Drainage in the Treatment of Pelvic Abscesses: A Case Series
EUS Guided Drainage in the Treatment of Pelvic Abscesses: A Case Series
Varshita Goduguchinta, DO1; Mohamed Ebrahim, MD2; Raahi Patel, DO3; Navkiran Randhawa, DO4; Ahamed Khalyfa, DO5; Mahnoor Inamullah, MD6; Rahil Desai, DO7; Kamran Ayub, MD, MRCP, FASGE8*
1, 3,7. Franciscan Health- Olympia Fields, IL;
2 Ascension Saint Joseph Hospital - Chicago, IL;
4 Medical College of Georgia - Department of Gastroenterology;
5 The University of Iowa - Department of Gastroenterology;
6 Southwest Gastroenterology, a Division of GI Partners of Illinois;
8 Silver Cross Hospital, New Lenox, IL ;
*Correspondence to: Dr. Kamran Ayub, MD, MRCP, FASGE; Silver Cross Hospital, New Lenox, IL.
Copyright
© 2025 Dr. Kamran Ayub, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 23 June 2025
Published: 30 June 2025
DOI: https://doi.org/10.5281/zenodo.16755016
Abstract:
Background: Pelvic abscesses, particularly those in deep supralevator or perirectal locations, pose significant therapeutic challenges when traditional drainage approaches are contraindicated or technically unfeasible. This study aims to evaluate the safety and efficacy of endoscopic ultrasound (EUS)-guided transrectal drainage of pelvic abscesses using
lumen-apposing metal stents (LAMS), with or without coaxial plastic stents.
Methods: This retrospective case series includes six patients who underwent EUS-guided drainage across three hospitals from May 2020 to May 2024. All patients had symptomatic pelvic abscesses not amenable to image-guided percutaneous drainage, confirmed by cross-sectional imaging.
Interventions were performed using a linear-array echoendoscope and the electrocautery-enhanced Hot AXIOS™ LAMS system. A 7 Fr × 3 cm double pigtail plastic stent was placed coaxially in four patients to enhance drainage. Data collected included patient demographics, abscess characteristics, procedural details, clinical outcomes, and adverse events.
Results: Technical success was achieved in all six cases (100%). The average initial abscess size was 5.65 × 4.62 cm (range: 4.0–8.2 cm). Clinical resolution, defined by symptom improvement and radiologic confirmation of cavity collapse (<2 cm), occurred in all patients. Median stent indwelling time was four weeks. No major complications (bleeding, stent migration, perforation, or infection) were observed. Minor self-limited pelvic pain was reported in two patients. No patients required surgical intervention or experienced abscess recurrence during follow-up.
Conclusions: EUS-guided transrectal drainage using LAMS, with optional coaxial plastic stenting, is a safe and effective alternative for managing pelvic abscesses in patients not suitable for percutaneous drainage. These findings support its growing role in multidisciplinary abscess management and highlight its utility in high-risk or anatomically challenging cases.
EUS Guided Drainage in the Treatment of Pelvic Abscesses: A Case Series
Introduction
Pelvic abscesses are common infections arising from the anal glands and range in severity from superficial perianal infections to deep pelvic abscesses, such as intersphincteric, ischiorectal, and supralevator collections. Prompt and effective drainage is the cornerstone of management, as delayed or inadequate intervention may result in complications including fistula formation, chronic infection, or systemic sepsis1.
While surgical incision and drainage (I&D) and image-guided percutaneous drainage remain the mainstays of treatment, certain clinical scenarios can limit the feasibility or safety of these conventional approaches. Anatomical challenges, such as deep supralevator location, lack of percutaneous access windows, or proximity to critical structures, as well as patient-related factors like severe comorbidities, immunosuppression, or prior pelvic surgeries, may preclude safe or effective drainage by traditional means. Endoscopic ultrasound (EUS)-guided drainage has emerged as a minimally invasive alternative in select cases, allowing for direct visualization and targeted access to abscess cavities through the rectal or colonic wall. EUS offers precise localization of deep-seated collections and the ability to place internal drains under real-time imaging guidance, minimizing procedural risk and avoiding the need for general anesthesia or external drains. Although widely adopted in the drainage of pancreatic fluid collections, its role in managing pelvic abscesses, particularly rectal or perirectal, has gained increasing attention 1, 2.
In this case series, we describe six patients with rectal abscesses who underwent successful EUS-guided drainage after traditional surgical and interventional radiology (IR) options were deemed unsuitable or high-risk. These cases highlight the growing utility of EUS in the multidisciplinary management of complex pelvic infections, supporting its use as a safe and effective alternative in appropriately selected patients.
Methods
All six consecutive patients included in this retrospective case series, performed across three hospitals from May 2020 to May 2024, were carefully selected for EUS-guided transrectal drainage based on a comprehensive pre-procedural evaluation. Each patient presented with symptomatic pelvic abscesses that were deemed unsuitable by interventional radiology for percutaneous drainage due to their deep pelvic location and proximity to critical vascular or visceral structures. Selection criteria included confirmation of abscess presence on cross-sectional imaging (CT or MRI) with failure of antibiotic therapy alone. Pre-procedural imaging was meticulously reviewed to assess abscess dimensions, wall characteristics, and spatial relationship to the rectal wall, with particular attention to maintaining a safe distance from major vascular structures. The interventions were performed in a dedicated interventional endoscopy suite equipped with fluoroscopic capabilities and advanced life support systems. All procedures were conducted under propofol sedation administered by a dedicated anesthesiology team. A single advanced endoscopist with extensive experience in therapeutic EUS performed all cases using a linear-array echoendoscope (GF-UCT-180; Olympus Medical Systems, Tokyo, Japan) with Doppler functionality. The Hot AXIOS™ LAMS system (10-15 mm diameter, Boston Scientific) was selected as the primary drainage device due to its integrated electrocautery-enhanced delivery system, which facilitates direct access without requiring separate needle puncture, and lumen opposing design.
The standardized procedural approach began with careful endoscopic inspection of the rectum to exclude contraindications such as severe proctitis or obstructing lesions. Under continuous EUS guidance, the abscess cavity was identified and its relationship to the rectal wall was confirmed. Doppler interrogation was systematically performed to map regional vasculature and identify the safest puncture trajectory. The hot AXIOS delivery system was then advanced under direct endoscopic and endosonographic visualization, with the electrocautery tip used to create a controlled transmural tract into the abscess cavity. Deployment of the LAMS followed a standardized sequence: initial release of the distal flange within the abscess cavity under EUS guidance, followed by deployment of the proximal flange in the rectal lumen under direct endoscopic visualization, creating a stable 10-15 mm diameter conduit for drainage (Table 1).
Following LAMS deployment, a 7 Fr x 3 cm double pigtail plastic stent (Cook Medical, Indiana, USA) was routinely placed through the lumen of each AXIOS stent using a combination of guidewire and catheter techniques in four of the patients. This coaxial stenting strategy was employed to prevent stent occlusion from viscous purulent material and to minimize the risk of stent migration. Immediate post-procedural assessment included endoscopic confirmation of purulent drainage and fluoroscopy of proper stent positioning when indicated. Patients were monitored in a step-down unit for 24 hours, depending on their clinical condition. A structured follow-up protocol was implemented for all patients, beginning with outpatient clinical evaluation at 1 week post-procedure. Subsequent management was guided by a combination of symptom assessment and scheduled imaging follow-up, with CT or MRI performed at 2–4 week intervals to monitor abscess resolution. Endoscopic evaluation with possible hydrogen peroxide lavage was performed in cases with persistent collections. Definitive stent removal was only undertaken after both clinical resolution (absence of fever, pain, or leukocytosis) and radiologic confirmation of abscess resolution (cavity collapse to <2 cm). The median indwelling time for stents was 4 weeks (range 3-6 weeks), with removal performed using standard endoscopic techniques under propofol sedation. All procedures incorporated multiple safety measures, including real-time Doppler assessment, continuous hemodynamic monitoring, and immediate availability of surgical support. Complications were monitored with specific attention to bleeding, perforation, stent migration, and infection.
Results
A total of six patients (n = 6) underwent endoscopic ultrasound (EUS)-guided transrectal drainage for deep pelvic abscesses that were not amenable to percutaneous drainage as deemed by interventional radiology. The cohort consisted of four females and two males, with a mean age of 70.2 years (range: 46 to 92 years), illustrating that this minimally invasive technique was applied across a wide age range, including elderly individuals who typically present higher procedural risks. The decision to proceed with EUS-guided drainage was driven by the deep pelvic location of the abscesses and their proximity to critical anatomical structures, such as the bladder, pelvic vessels, and rectum, which rendered percutaneous drainage either unsafe or technically not feasible.
The initial abscess sizes, as measured on cross-sectional imaging (CT or MRI), varied from 4.0 × 3.2 cm to 8.2 × 6.1 cm, with a mean size of 5.65 × 4.62 cm. The smallest abscess was observed in a 92-year-old female, while the largest abscess was seen in an 86-year-old male patient. Each abscess exhibited differing fluid viscosity and amounts of internal debris, which guided the selection of drainage techniques and adjunctive measures. All patients underwent successful transrectal deployment of a lumen-apposing metal stent (LAMS) under EUS guidance. A 15 mm Axios stent was used in five patients, while a 10 mm Axios stent was selected for the 92-year-old female with the smallest abscess cavity to better accommodate the dimensions of the abscess and minimize the risk of local tissue trauma.
In addition to the Axios stent, a 7 Fr × 3 cm double pigtail plastic stent was placed coaxially through the LAMS in four patients (Image 1). The decision to place a coaxial stent was influenced by the endoscopist’s assessment of the abscess characteristics at the time of drainage, particularly the viscosity of the fluid and the risk of lumen occlusion by debris. In the remaining two patients, brisk drainage was observed upon initial stent placement, and the endoscopist determined that additional coaxial support was not required. The number of endoscopic procedures per patient ranged from two to four, reflecting the individualized nature of management depending on abscess size, response to initial drainage, and the presence of residual debris. All patients underwent at least one follow-up hydrogen peroxide lavage, typically performed one week following the initial drainage. Four patients underwent a total of three procedures (stent placement, lavage, and removal). The 86-year-old male patient, who had the largest abscess at 8.2 × 6.1 cm, required four procedures: stent placement, two lavages at one- and three-week post-procedure, and stent removal. This patient’s more complex abscess required additional interventions to achieve adequate clearance of residual material.
The duration of stent indwelling varied from three to six weeks (median: four weeks), depending on the speed of clinical and radiologic resolution. For example, the 68-year-old female with a 4.6× 4.4 cm abscess had her stent removed at three weeks following near-complete resolution, whereas the patient with the largest abscess required six weeks before stent removal was appropriate. Radiologic follow-up confirmed excellent outcomes across the cohort. Five patients showed residual cavities measuring less than 1 cm, and the patient with the largest initial abscess had a residual cavity measuring less than 2 cm. No cases of abscess recurrence were documented during the follow-up period, demonstrating the effectiveness of this approach for durable abscess resolution.
From a safety standpoint, the procedures were well tolerated in all cases. There were no major complications, including no instances of bleeding, perforation, stent migration, or secondary infection necessitating hospital readmission or antibiotic escalation. Two patients reported minor post-procedural discomfort localized to the pelvis: a 50-year-old female experienced pain for three days following the procedure, and a 79-year-old male reported discomfort lasting four days. In both cases, symptoms resolved spontaneously without requiring additional analgesia or medical intervention. No patient experienced unplanned hospitalization, no surgical rescue procedures were necessary, and no escalation of antibiotic therapy beyond periprocedural prophylaxis was required.
Figure 1: Endoscopic view showing successful deployment of a LAMS (Axios) into a rectal abscess cavity, with a coaxially placed 7 Fr × 3 cm double pigtail plastic stent to support ongoing drainage and minimize occlusion
|
Table 1: Demographics, Abscess Parameters, and Clinical Course in Patients Undergoing EUS-Guided Transrectal Drainage Using Lumen-Apposing Metal Stents |
||||||
|
Patient Demographics (Age/Sex) |
Drainage |
Initial Rectal Abscess size |
Post Drainage Cavity size |
Drainage Time |
Number of Procedures (stent placement; lavage(s); removal) |
Complications |
|
68 yo F |
Axios 15 mm with 7 Fr x 3 cm double pigtail stent |
4.6 x 4.4 cm |
< 1 cm cavity |
3 weeks |
2 |
None |
|
46 yo F |
Axios 15 mm with 7 Fr x 3 cm double pigtail stent |
5.2 x 4.9 cm |
< 1 cm cavity |
4 weeks |
3 |
None |
|
86 yo M |
Axios 15 mm 7 Fr x 3 cm double pigtail stent |
8.2 x 6.1 cm |
< 2 cm cavity |
6 weeks - had 2 lavages (1weeks and 3 weeks later) |
4 |
None |
|
50 yo F |
Axios 15 mm |
6.2 x 4.9 cm |
< 1 cm cavity |
4 weeks |
3 |
Post procedure pain x 3 days; self-resolved |
|
92 yo F |
Axios 10 mm |
4.0 x 3.2 cm |
< 1 cm cavity |
3 weeks |
2 |
None |
|
79 yo M |
Axios 15 mm 7 Fr x 3 cm double pigtail stent |
5.7 x 4.2 cm |
< 1 cm cavity |
4 weeks |
3 |
Post procedure pain x 4 days; self-resolved |
Discussion
Pelvic abscesses may develop from a variety of causes, including gastrointestinal perforation, inflammatory bowel disease, and postoperative complications from colorectal, gynecological, or urological surgeries. Among these, acute diverticulitis is reported as one of the most common causes, particularly when associated with localized perforation or walled-off peritonitis3. When complicated by rupture or sepsis, pelvic abscesses can become life-threatening, and conservative management with antibiotics alone is often insufficient. Image-guided or surgical drainage is usually necessary for definitive management.
Endoscopic ultrasound (EUS) has become an increasingly valuable modality in the minimally invasive management of rectal and perirectal abscesses, particularly those that are deep-seated, anatomically inaccessible, or not amenable to surgical or percutaneous drainage. By employing a linear-array echoendoscope, EUS provides direct visualization of pelvic anatomy and real-time needle guidance, allowing for precise transrectal or transanal access to abscess cavities with reduced risk to surrounding structures1. This case series supports the use of minimally invasive EUS-guided transrectal drainage as an effective and well-tolerated intervention for deep pelvic abscesses that are not amenable to percutaneous drainage. All patients in our series achieved radiologic resolution without recurrence, and clinical recovery was complete. Even in patients with large or complex abscesses, including the one with an 8.2 × 6.1 cm cavity requiring multiple lavages, the EUS-guided approach was sufficient to avoid surgical intervention. The consistency of favorable outcomes across varying abscess sizes suggests that this technique may also be effective in multilocular or partially septated collections, likely due to internal communication between compartments.
Multiple studies support the safety and efficacy of EUS-guided drainage in this context. Varadarajulu et al. demonstrated in a 25-patient case series that EUS-guided transrectal drainage of pelvic abscesses is a highly effective and safe minimally invasive approach, achieving a 96% clinical success rate without complications or recurrences4. Ratone et al expanded on this by comparing EUS-guided drainage to other minimally invasive approaches and found significantly shorter hospital stays and fewer complications associated with EUS in a retrospective study involving 7 patients 2.
A systematic review by Ak?nc? et al. reinforced these findings, reporting pooled technical and clinical success rates of 100% and 93.9%, respectively, in patients with pelvic fluid collections, including abscesses, treated with EUS guidance1. Another recent multicenter study by Chow et al. also highlighted the role of lumen-apposing metal stents (LAMS) in facilitating EUS-guided drainage of pelvic abscesses, particularly in complex or recurrent cases, with favorable safety and efficacy outcomes5. In our cohort, placement of LAMS was successful in all cases, and in most patients, coaxial plastic stents were placed through the Axios stent to optimize drainage and minimize occlusion. Importantly, no additional flushing catheters or nasocystic irrigation systems were used, consistent with evidence suggesting that such adjuncts may not be necessary for successful outcomes6,7. Concerns about potential stent occlusion from fecal matter or pus have led some practitioners to advocate for adjunctive irrigation or larger stent diameters4. However, based on our findings and consistent with other reports6,7, effective cavity resolution can often be achieved with proper lavage technique and selective use of coaxial plastic stents.
Despite these promising results, the widespread use of EUS-guided drainage remains limited by the availability of expertise, specialized equipment, and procedural standardization. The technique is typically confined to tertiary care centers with experienced interventional endosonographers. As shown in our case series, EUS-guided drainage offers a safe and effective alternative in the six patients who are not candidates for conventional drainage methods.
Conclusion
Our case series demonstrates that EUS-guided transrectal drainage of pelvic abscesses using Axios stents, with or without coaxial plastic stents, is a safe and effective alternative to percutaneous or surgical approaches, particularly when standard drainage routes are not feasible. All procedures were successfully completed under propofol sedation with fluoroscopic assistance, and no major complications or abscess recurrences occurred. These findings add to the growing body of evidence supporting EUS-guided pelvic abscess drainage as a first-line therapeutic option in select patients.
References
[1] Ak?nc? D, Ergun O, Topel Ç, Çiftçi T, Akhan O. Pelvic abscess drainage: outcome with factors affecting the clinical success. Diagn Interv Radiol. 2018;24(3):146-152. doi:10.5152/dir.2018.16500
[2] Ratone, Jean-Philippe; Bertrand, Julie; Godat, Sébastien1; Bernard, Jean-Paul; Heyries, Laurent. Transrectal drainage of pelvic collections: Experience of a single center. Endoscopic Ultrasound 5(2):p 108-110, Mar–Apr 2016. | DOI: 10.4103/2303-9027.180474
[3] Kriwanek S, Armbruster C, Beckerhinn P, Dittrich K. Prognostic factors for survival in colonic perforation. Int J Colorectal Dis. 1994;9(3):158-162. doi:10.1007/BF00290194
[4] Varadarajulu S, Drelichman ER. Effectiveness of EUS in drainage of pelvic abscesses in 25 consecutive patients (with video). Gastrointest Endosc. 2009;70(6):1121-1127. doi:10.1016/j.gie.2009.08.034
[5] Chow KW, Cumpian NA, Makar R, Zargar P, Oza F, Suliman I, Eysselein V, Reicher S. Lumen-Apposing Metal Stents in the Management of Complex Pelvic Abscesses. Diagnostics. 2024; 14(24):2854. https://doi.org/10.3390/diagnostics14242854
[6] Puri R, Eloubeidi MA, Sud R, Kumar M, Jain P. Endoscopic ultrasound-guided drainage of pelvic abscess without fluoroscopy guidance. J Gastroenterol Hepatol. 2010;25(8):1416-1419. doi:10.1111/j.1440-1746.2010.06328.x
[7] Giovannini M, Bories E, Moutardier V, et al. Drainage of deep pelvic abscesses using therapeutic echo endoscopy. Endoscopy. 2003;35(6):511-514. doi:10.1055/s-2003-39673.
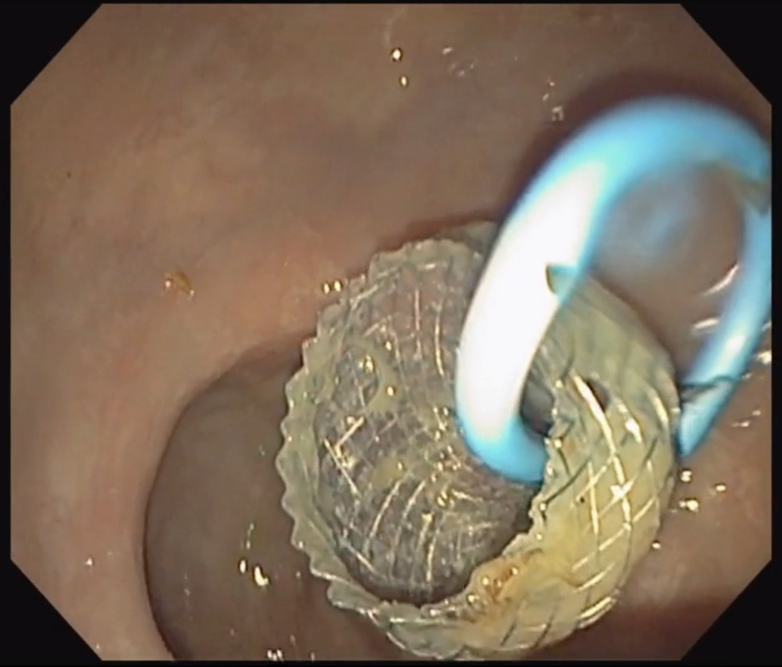
Figure 1
