Congenital Diaphragmatic Hernia, A Case Report
Congenital Diaphragmatic Hernia, A Case Report
Bustillos Rodríguez Alvaro1, Luna Logroño Alejandra2, Manzano Moscoso Daniel3
1. Resident Physician, Neonatology Unit, Hospital de Especialidades de las Fuerzas Armadas N°1, Quito Ecuador;
2. Specialist Pediatrician, Neonatology Unit, Hospital de Especialidades de las Fuerzas Armadas N°1, Quito Ecuador;
3. Specialist Pediatric Surgeon, Hospital de Especialidades de las Fuerzas Armadas N°1, Quito Ecuador.
*Correspondence to: Bustillos Rodríguez Alvaro. Resident Physician, Neonatology Unit, Hospital de Especialidades de las Fuerzas Armadas N°1, Quito Ecuador.
Copyright
© 2025: Bustillos Rodríguez Alvaro. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 16 June 2025
Published: 01 July 2025
Abstract
Congenital diaphragmatic hernia is a defect in the diaphragm in which abdominal organs are found in the chest. It occurs in 2.4 out of every 10,000 births. Prenatal diagnosis is made in 50-60% of cases by ultrasound. At birth, signs include respiratory distress, cyanosis, and scaphoid abdomen; diagnosis at birth is made by chest X-ray. It is often associated with congenital heart defects and gastrointestinal or neurological anomalies. Treatment includes hemodynamic stabilization, immediate oro-intubation, mechanical ventilation management, and associated pulmonary hypertension management. Corrective surgery is performed when clinical conditions allow. We present the case of a term newborn who presented with left-sided diaphragmatic hernia, received treatment and hemodynamic stabilization, and finally underwent surgery, where additional intestinal malrotation was found. After the procedure, the patient showed favorable evolution upon medical discharge.
Congenital Diaphragmatic Hernia, A Case Report
Introduction
Congenital diaphragmatic hernia is defined as a discontinuity in the development of the diaphragm, which leads to the presence of abdominal structures in the thorax, and results in pulmonary hypoplasia.(1) Its prevalence is approximately 2.4 cases per 10,000 births, with a male-to-female predominance ratio of 1:0.69. (2) (3) There are multiple genetic and environmental factors for its development, among these are mentioned: Turner, Fryns syndromes, trisomy 13, 18 and 21, autosomal recessive, autosomal dominant, and X-linked inheritance patterns, although exposure to drugs such as thalidomide, anticonvulsants, mycophenolate, and vitamin A deficiency are also mentioned.(4) The pathogenesis is related to defects in the pleuroperitoneal folds, which would cause failures in diaphragmatic closure; however, it is currently postulated that there are also failures in fetal lung development as a first step towards the failure of diaphragmatic closure.(4)
Classification
According to their location, diaphragmatic hernias are classified as posterolateral or Bochdaleck (70-75%) with left predominance, anterior or Morgagni hernias (23-28%), and central hernias (2-7%). (5)
Prenatal Diagnosis
Approximately 50-60% of cases are diagnosed during the prenatal period. If suspected, the mother should be referred to a specialized center, and a detailed ultrasound is recommended, which identifies abdominal organs in the thoracic cavity, the presence of polyhydramnios, abnormal cardiac axis, or mediastinal displacement. (6) Fetal magnetic resonance imaging is useful for evaluating the size of the defect and lung involvement, and genetic alterations could also be investigated. With these results, a decision can be made between expectant management, fetal intervention, or termination of pregnancy. (7) (8)
Postnatal Diagnosis
At birth, patients often present with respiratory distress, cyanosis, decreased breath sounds on the affected side, and scaphoid abdomen. (9) Diagnosis is determined by a chest X-ray, which shows intestinal loops passing through the diaphragm, pulmonary hypoplasia, and deviation of mediastinal structures. (10)
The most commonly associated major congenital anomaly in these patients is congenital heart disease (ventricular septal defects 24%, atrial septal defects 21%, single ventricle defects 12%, coarctation of the aorta 9%, tetralogy of Fallot 5%); for this reason, echocardiography is useful. (11) Other non-cardiac anomalies include: gastrointestinal, neural tube defects, central nervous system malformations, genitourinary defects, esophageal atresia, cryptorchidism. Patients with diaphragmatic hernia have intestinal malrotation and a predisposition to developing volvulus. The rates of this potentially devastating complication vary from 3-9%. (12)
Treatment
Initial management should be by a certified multidisciplinary team. Prenatal diagnosis allows anticipating the measures to be taken. When respiratory distress presents, immediate endotracheal oro-intubation is necessary, and bag-mask ventilation should be avoided to prevent more air from entering the stomach. Placement of a naso or orogastric tube is recommended to decompress the stomach. (5)(13)
Initial stabilization: A central or peripheral venous access should be placed to administer fluids and medications. (5) Achieve oxygenation goals between 85-95%, blood pressure within appropriate percentiles for gestational age, and treat hydroelectrolytic and acid-base disorders. (10)
Mechanical ventilation: There is no ideal ventilation mode to apply in newborns with this pathology. (5) The maximum inspiratory pressure (PIP) should be adjusted to less than 25 cm H2O, permissive hypercapnia (PaCO2 55 mmHg), and preductal oxygenation of 85- 90% to minimize barotrauma and avoid hypoxemia and acidosis. (10)(14)
Additional pulmonary hypertension occurs due to damage to the pulmonary vasculature. Several drugs may be useful in treatment, such as: nitric oxide, sildenafil, milrinone, bosentan, and prostaglandins. (15)
For surgical resolution, the patient's hemodynamic stability must be considered when deciding when to perform the procedure. (16) Surgical correction involves relocating displaced organs and closing the defect. It is generally performed by an open procedure. Most defects are primarily repaired (60-70%), while in more severe cases (and larger defects), a patch, which can be absorbable or non-absorbable (polytetrafluoroethylene), is used. (17)
Case Presentation
Male newborn, 36.4 weeks by last menstrual period, Apgar 5 -7T, weight 2,570 grams, ORH+. No significant maternal history, normal prenatal ultrasounds. Born in a second- level hospital, presenting with acute respiratory distress, requiring advanced neonatal resuscitation with positive pressure ventilation and orotracheal intubation. Transferred to our hospital where the presence of left diaphragmatic hernia associated with respiratory insufficiency, shock, perinatal asphyxia, and seizures was determined. Admitted to the neonatal intensive care unit in the following conditions: Hypotonic, flaccid, hyporeactive pupils with diminished brainstem reflexes, oro-intubated, shock score 4 points, and on vasoactive drugs. Chest X-ray showed the presence of intestinal loops in the left hemithorax, signs of left pulmonary hypoplasia (figure 1);
Figure 1 AP Chest X-ray
Opacification of left hemithorax and contralateral cardiac displacement
Source: Hospital de Especialidades de las Fuerzas Armadas N°1
Values of tissue hypoxia (table 1), metabolic acidosis (table 2), and an infectious profile with leukocytosis and neutrophilia (table 3) were observed. Received treatment with invasive mechanical ventilation support and broad-spectrum antibiotic therapy. But due to worsening clinical condition and marked leukocytosis, meropenem (30 mg/kg/dose) intravenously every 8 hours was started, as well as vancomycin (20 mg/kg/dose) intravenously daily for 10 days. Anticonvulsants were started due to the presentation of subtle seizures with lip-smacking, based on levetiracetam (25 mg/kg/day) intravenously every 12 hours for 27 days. Received vasoactive doses due to arterial pressures below the 50th percentile and moderate to severe shock score, with dopamine (14 ug/kg/min) for 10 days, norepinephrine (0.3 ug/kg/min) for 12 days. Complementary studies included a normal transfontanellar ultrasound. An electroencephalogram reported rare epileptiform paroxysms with spike morphology in the anterior regions of the left hemisphere. A brain magnetic resonance imaging showed hyperacute ischemic lesions in border-zone territories of frontoparietal, occipitotemporal, and thalamic regions, as a sequela of severe or sustained perinatal and/or postnatal hypoxic disease. The echocardiogram performed at 15 days of life reported: pulmonary pressures with mild pulmonary hypertension. Received sildenafil for pulmonary hypertension (1mg/kg/dose) orally for 5 days.
Table 1 Asphyxia Blood Profile
|
CPK MB: |
283.6 U/L |
|
CK: |
3 364.8 U/L, |
|
LDH: |
2 677.2 U/L |
|
Troponin I: |
1.48 ng/ml. |
Elevated Markers
Source: HE-1 Laboratory
Table 2 Venous Blood Gas
|
PH |
7,15 |
|
PCO2 |
29 mmHg |
|
PO2 |
25 mmHg |
|
HCO3 |
10,1 mmol/L |
|
EBASE |
-17,4 mmol/L |
|
SATO2 |
37,8 % |
|
LACTATE |
>17 mmol/L |
Source: HE-1 Laboratory
Table 3 Complete Blood Count - Renal Function
|
Leukocytes |
26,37 10³/µL |
|
Neutrophils |
79,2% |
|
Hemoglobin |
12,6 g/dL |
|
Hematocrit |
37,9 % |
|
Platelets |
99,000 10³/µL |
|
Urea |
38,27 mg/dL |
|
Creatinine |
1,5 mg/dL |
|
C reactive protein (PCR) |
0,17 mg/dL |
Source: HE-1 Laboratory
Due to hemodynamic instability, he underwent surgery on day 9 of life. Surgical findings were: left diaphragmatic defect of 5 cm in diameter, loops of small intestine, transverse colon, descending colon, cecum, appendix, ascending colon, sigmoid colon, inside the thoracic cavity, hypoplastic left lung with minimal expansion, associated complete intestinal malrotation. A left subcostal abdominal incision of 8cm was performed, location of the diaphragmatic defect, rearrangement of intestinal loops in the abdominal cavity, primary diaphragmatic raphe with continuous 3/0 prolene suture, without the need for mesh or thoracic tube placement. (figure 2 and 3)
Figure 2 Diaphragmatic hernia intraoperative view
Congenital defect with intestinal content
Source: Dra. Luna Alejandra. Neonatology Service HE-1
Figure 3 Abdominal Content in Thorax
Associated intestinal malrotation
Source: Dra. Luna Alejandra. Neonatology Service HE-1
After the surgical procedure, an adequate clinical evolution was observed, where he received high-frequency ventilation support for 3 days, and then non-invasive ventilation for 1 day. Control chest X-ray showed correction of the defect and an improvement in left lung expansion. (Figure 4). He remained in the unit for 28 days, where he received comprehensive management and was discharged medically, oxygen-dependent by nasal cannula at low flows.
Figure 4 Postoperative Chest X-ray
Correction of left diaphragmatic defect.
Source: Hospital de Especialidades de las Fuerzas Armadas N°11
Discussion
A clear origin of congenital diaphragmatic hernia is unknown, but it results from the association of multiple factors. It occurs due to incomplete closure of the pleuroperitoneal folds, which generates defects in the diaphragm and allows the displacement of abdominal organs into the thoracic cavity. This alteration interferes with the normal development of the lungs, with an estimated prevalence of 2.5 cases per 10,000 births. (3)
In recent decades, the survival rate of these patients has improved considerably thanks to advances in perinatal and surgical management. Putnam et al. reported in 2016 that, in an international cohort of 3665 neonates, survival to discharge ranged from 70% to 84%. However, associated morbidity remains significant, with gastrointestinal alterations in 61% of cases, pulmonary in 30%, neurological in 20%, and presence of multiple morbidities in 34%. Among the most relevant prognostic factors, defect size stands out as the main predictor of complications. (18)
On the other hand, studies such as that by Burgos et al., conducted in Sweden between 1987 and 2013, report mortality rates ranging from 36% to 44% in patients born with congenital diaphragmatic hernia. Furthermore, they introduce the concept of "hidden mortality," which includes cases not born due to pregnancy terminations or intrauterine deaths, raising the total estimated mortality to 45% in this population. This underscores the need for early and effective diagnostic and therapeutic strategies. (19)
Regarding surgical technique, the placement of prosthetic patches has been associated with a higher risk of complications. In a systematic review by Heiwegen et al., with 2910 patients between 1991 and August 2020, it was observed that the use of patches significantly increases the probability of recurrence (2.8 times), as well as chylothorax (2.5 times) and intestinal obstruction (double the risk), compared to repair without a patch. (17) Therefore, when anatomical conditions allow, primary raphe is preferred, as described in the case here.
It is unclear whether immediate surgical repair increases mortality or decreases hospital days. This is demonstrated by Yunes et al. in their meta-analysis conducted in 2017, with four systematic reviews that, together, include 38 primary studies. The certainty of the evidence is very low. Given the limited evidence, it is usually decided to operate on patients later, once sustained hemodynamic stability has been achieved. (20)
This position is reinforced by the recommendations of the CDH EURO 2015 consortium, whose clinical practice guideline determines that surgical repair of the diaphragmatic defect should be performed after clinical stabilization, defined as follows: mean arterial pressure normal for age, preductal saturation levels of 85–95% with FiO2 below 50%, lactate below 3 mmol/l, urine output greater than 1 ml/kg/h. However, the evidence on this is limited. (21)
In the described case, the patient initially required support measures to achieve adequate hemodynamic stabilization. Vasoactive agents were administered to achieve arterial pressures within normal ranges for his age. In the context of neonatal sepsis, empirical broad-spectrum antibiotic treatment was initiated, according to current clinical recommendations. Respiratory management involved invasive mechanical ventilation, and as clinical improvement was evidenced, ventilatory parameters were progressively reduced. Likewise, targeted treatment for pulmonary hypertension was implemented, and anticonvulsant medication was started due to the presence of seizures, which subsided with the established management. These factors, although common in patients with congenital diaphragmatic hernia, represent significant challenges on the path to hemodynamic stabilization. Due to a favorable response to treatment, sustained hemodynamic stability was achieved, evidenced by the absence of vasoactive support requirements, low ventilatory parameters, absence of seizures, and a negative infectious profile, with no clinical signs of active sepsis. Considering sustained hemodynamic stability and according to available evidence supporting surgical intervention once patient stabilization is achieved, correction of the diaphragmatic defect was performed on the ninth day of life. The surgery was successfully carried out by primary raphe of the defect using prolene suture material, without the need for prosthetic patches. This approach aligns with evidence supporting delayed intervention in stable patients and possibly contributed to the patient's favorable outcome. In the following days, oral feeding was started with adequate tolerance and good evolution. Finally, he was discharged with supplemental oxygen support via low- flow nasal cannula, in favorable general condition. This case highlights the importance of an individualized approach, guided by both the patient's clinical condition and available evidence. Multidisciplinary management, close monitoring, and surgical planning based on hemodynamic stability criteria appear to be key to optimizing outcomes in neonates with congenital diaphragmatic hernia.
Conclusion
Congenital diaphragmatic hernia remains a complex anomaly that requires a comprehensive clinical approach and decision-making based on both patient evolution and available scientific evidence. The presented case highlights the relevance of achieving sustained hemodynamic stability before proceeding with surgical intervention, a strategy that aligns with current recommendations and can contribute to improved clinical outcomes. Furthermore, the possibility of performing a primary repair without patches, as in this case, is associated with a lower incidence of subsequent complications, reinforcing the importance of individualized surgical evaluation. This case illustrates that, even in challenging clinical scenarios, timely coordination between neonatal, surgical, and intensive care teams allows for optimizing results. The experience gained, combined with current knowledge, suggests that the successful treatment of congenital diaphragmatic hernia does not depend exclusively on surgical intervention, but on a carefully adapted clinical plan for each patient.
References
1. Kosi?ski P, Wielgo? M. Congenital diaphragmatic hernia: pathogenesis, prenatal diagnosis and management — literature review. Ginekol Pol. 2017 Jan 31;88(1):24–30.
2. Feng Z, Wei Y, Wang Y, Liu C, Qu D, Li J, et al. Development of a prediction nomogram for 1-month mortality in neonates with congenital diaphragmatic hernia. BMC Surg. 2024 Jun 27;24(1):198.
3. McGivern MR, Best KE, Rankin J, Wellesley D, Greenlees R, Addor MC, et al. Epidemiology of congenital diaphragmatic hernia in Europe: a register-based study. Arch Dis Child Fetal Neonatal Ed. 2015 Mar;100(2):F137–44.
4. Köse EE. Congenital diaphragmatic hernia. The Turkish Journal of Thoracic and Cardiovascular Surgery. 2024 Feb 5;32(Supplementum 1):89–97.
5. Chandrasekharan PK, Rawat M, Madappa R, Rothstein DH, Lakshminrusimha S. Congenital Diaphragmatic hernia – a review. Matern Health Neonatol Perinatol. 2017 Dec 11;3(1):6.
6. Chatterjee D, Ing RJ, Gien J. Update on Congenital Diaphragmatic Hernia. Anesth Analg. 2020 Sep 17;131(3):808–21.
7. Bendixen C, Brosens E, Chung WK. Genetic Diagnostic Strategies and Counseling for Families Affected by Congenital Diaphragmatic Hernia. European Journal of Pediatric Surgery. 2021 Dec 15;31(06):472–81.
8. Cordier AG, Russo FM, Deprest J, Benachi A. Prenatal diagnosis, imaging, and prognosis in Congenital Diaphragmatic Hernia. Semin Perinatol. 2020 Feb;44(1):51163.
9. Yeon Lee S, Jackson JE, Lakshiminrusimha S, Brown Y Diana L Farmer EG. Trastornos anatómicos del tórax y las vías respiratorias. 2025.
10. Lizano Villarreal MJ, Arroyo Solís D, Gamboa Hernández F. Hernia diafragmática congénita: una actualización en el abordaje diagnóstico y terapéutico. Revista Medica Sinergia. 2023 Apr 1;8(4):e974.
11. Hedrick H, Adzick S. Congenital diaphragmatic hernia (CDH) in the neonate: Clinical features and diagnosis. Uptodate [Internet]. 2024 [cited 2025 Jan 24]; Available from: https://www.uptodate.com/contents/congenital-diaphragmatic- hernia-cdh-in-the-neonate-clinical-features-and-diagnosis?search=hernia%20dia%20fragm%C3%A1tica%20cong%C3%A9nita& source=search_result&selectedTitle=3%7E60&usage_type=default&display_rank=3
12. Hedrick H, Adzick S. Congenital diaphragmatic hernia (CDH) in the neonate: Management and outcome. Uptodate. 2024;
13. Weiner GM., Zaichkin Jeanette. Libro de texto para la Reanimación Neonatal editor, Gary M. Weiner ; editores asociados, Jeanette Zaichkin. American Academy of Pediatrics; 2022.
14. Cordier AG, Saada J, Mokhtari M, Benachi A. Hernia diafragmática congénita. EMC - Ginecología-Obstetricia. 2017 Jun;53(2):1–12.
15. Yang MJ, Russell KW, Yoder BA, Fenton SJ. Congenital diaphragmatic hernia: a narrative review of controversies in neonatal management. Transl Pediatr. 2021 May;10(5):1432–47.
16. Puligandla PS, Skarsgard ED, Offringa M, Adatia I, Baird R, Bailey JAM, et al. Diagnosis and management of congenital diaphragmatic hernia: a clinical practice guideline. Can Med Assoc J. 2018 Jan 29;190(4):E103–12.
17. Heiwegen K, de Blaauw I, Botden SMBI. A systematic review and meta-analysis of surgical morbidity of primary versus patch repaired congenital diaphragmatic hernia patients. Sci Rep. 2021 Jun 16;11(1):12661.
18. Putnam LR, Harting MT, Tsao K, Morini F, Yoder BA, Luco M, et al. Congenital Diaphragmatic Hernia Defect Size and Infant Morbidity at Discharge. Pediatrics. 2016 Nov 27;138(5):e20162043.
19. Burgos CM, Frenckner B. Addressing the hidden mortality in CDH: A population- based study. J Pediatr Surg. 2017 Apr;52(4):522–5.
20. Yunes A, Luco M, Pattillo JC. Early versus late surgical correction in congenital diaphragmatic hernia. Medwave. 2017 Nov 17;17(09):e7081–e7081.
21. Snoek KG, Reiss IKM, Greenough A, Capolupo I, Urlesberger B, Wessel L, et al. Standardized Postnatal Management of Infants with Congenital Diaphragmatic Hernia in Europe: The CDH EURO Consortium Consensus - 2015 Update. Neonatology. 2016;110(1):66–74.
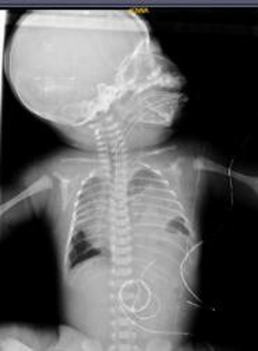
Figure 1
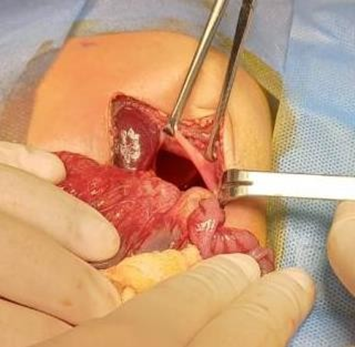
Figure 2
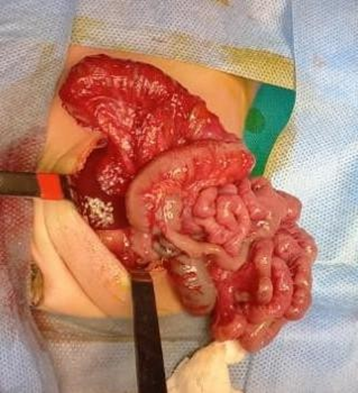
Figure 3
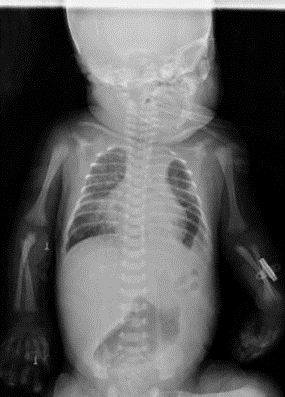
Figure 4
