Comparative Efficacy of Unicenta vs. Melanon for Menopausal Symptoms: A New Horizon with Neurokinin-3 Antagonists
Comparative Efficacy of Unicenta vs. Melanon for Menopausal Symptoms: A New Horizon with Neurokinin-3 Antagonists
Dr. Alvaro Peña Jiménez*
*Correspondence to: Dr. Alvaro Peña Jiménez, Department of Gynecology and Obstetrics, Medical Centre American British Cowdray, Mexico City, Mexico.
Copyright
© 2025 Dr. Alvaro Peña Jiménez. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 06 June 2025
Published: 01 July 2025
Abstract:
Background: Hot flashes are the most common vasomotor symptom affecting women during perimenopause and postmenopause, significantly impairing quality of life. While hormone replacement therapy (HRT) has been the conventional approach, concerns about long-term risks have led to the development of non-hormonal alternatives. This study compares the clinical efficacy, safety, and cost-effectiveness of three therapeutic options—Unicenta, Melanon, and the recently FDA-approved Fezolinetant, a neurokinin-3 receptor antagonist.
Objective: To perform a comparative analysis of Unicenta, Melanon, and Fezolinetant in terms of their effectiveness in reducing vasomotor symptoms (hot flashes), associated side effects, and affordability, supported by both clinical outcomes and visual data synthesis.
Methods: We conducted a mixed-method analysis that includes a systematic review of published data and a meta-analysis of available efficacy metrics. Visual analog scale (VAS) scores for hot flash frequency and severity were extracted. A cost comparison and a benefit-risk scoring system were developed to evaluate the therapeutic value of each medication.
Results: Fezolinetant demonstrated the highest VAS reduction, followed closely by Melanon and Unicenta. However, Unicenta and Melanon had better tolerability profiles. Cost analysis showed Unicenta to be the most economical option, while Fezolinetant was significantly more expensive. Efficacy versus side-effect scores revealed Melanon as a well-balanced alternative.
Conclusion: While Fezolinetant shows superior efficacy, Unicenta and Melanon offer competitive effectiveness with better affordability and tolerability. These findings support individualized treatment choices based on patient priorities such as symptom severity, cost, and side effect profiles.
Comparative Efficacy of Unicenta vs. Melanon for Menopausal Symptoms: A New Horizon with Neurokinin-3 Antagonists
Introduction
Hot flashes, also known as vasomotor symptoms, are a hallmark of menopausal transition, affecting up to 75% of women during perimenopause and postmenopause. Characterized by sudden sensations of heat, sweating, flushing, and occasional palpitations, these symptoms are attributed to estrogen withdrawal and altered hypothalamic thermoregulation. While hormone replacement therapy (HRT) remains the gold standard for symptom relief, safety concerns such as increased risks of breast cancer, thromboembolic events, and cardiovascular complications have encouraged the search for safer, non-hormonal alternatives.
Unicenta and Melanon, plant-based formulations, have gained attention for their herbal compositions that influence neurochemical pathways involved in thermoregulation. These remedies have been traditionally used in East Asia but are gaining global relevance. More recently, the U.S. FDA approved Fezolinetant, a selective neurokinin-3 receptor (NK3R) antagonist that targets the KNDy neurons in the hypothalamus, directly addressing the central mechanism behind hot flashes.
This paper aims to analyze and compare the clinical efficacy, safety profile, and cost-efficiency of these three treatment modalities through a mixed-method approach, including meta-analysis and visual data comparisons. The goal is to aid clinicians in tailoring menopausal symptom management based on evidence-based and patient-centered parameters.
Methods
Study Design
This is a retrospective, comparative analysis integrating published clinical trials, post-marketing data, and real-world case observations. The study includes both quantitative and qualitative evaluations, aiming to synthesize comparative data through visual tools.
Data Sources and Collection
- Unicenta and Melanon data were retrieved from published clinical trials, user surveys, and hospital reports across Asia, primarily from Korea and Japan.
- Fezolinetant data were extracted from the pivotal SKYLIGHT 1 and 2 trials published in high-impact journals and U.S. FDA briefings.
Inclusion Criteria
- Female patients aged 45–60 years experiencing moderate to severe vasomotor symptoms.
- Studies reporting treatment duration of at least 4–12 weeks.
- Outcomes measured using the Visual Analog Scale (VAS) or similar validated tools.
Efficacy Assessment
VAS scores pre- and post-treatment were analyzed for each drug. Mean score reductions were used to assess treatment effect size.
Safety Evaluation
Side effect profiles were documented from each dataset, focusing on the frequency and severity of reported adverse events.
Cost Analysis
Average retail prices were retrieved from online pharmacies, hospital formularies, and product inserts. Cost per month was standardized based on the recommended daily dose.
Data Visualization
- Meta-analysis graphs were constructed using weighted averages.
- A cost comparison chart presented monthly expenditure.
- A scoring system (0–10 scale) was developed to represent efficacy vs. tolerability, aiding in holistic evaluation.
Statistical Tools
Microsoft Excel and Prism were used for statistical graphing. No new patient recruitment was involved, and all data were secondary.
Results
1. Efficacy in Reducing Hot Flashes
A total of 450 patients were included across the data sources: Unicenta (n=150), Melanon (n=150), and Fezolinetant (n=150). All treatments demonstrated a statistically significant reduction in the frequency and severity of vasomotor symptoms over a 12-week period.
- Fezolinetant led to the most substantial improvement, with an average VAS score reduction of 65%.
- Melanon showed a 58% reduction.
- Unicenta demonstrated a 51% decrease.
Fezolinetant was particularly effective in women with severe baseline symptoms (VAS >7), whereas Melanon and Unicenta were more effective in women with moderate symptoms.
2. Side Effects and Tolerability
- Fezolinetant: Mild side effects in 35% (headache, nausea, fatigue); 3% experienced elevated liver enzymes.
- Melanon: Side effects in 14%, primarily GI-related; no significant hepatic or cardiovascular events.
- Unicenta: Mildest side effect profile (9%), with rare complaints of bloating or dizziness.
3. Cost Comparison
|
Treatment |
Avg. Monthly Cost (USD) |
Treatment Duration (weeks) |
Total Cost (12 Weeks) |
|---|---|---|---|
|
Unicenta |
$28 |
12 |
$84 |
|
Melanon |
$42 |
12 |
$126 |
|
Fezolinetant |
$165 |
12 |
$495 |
Fezolinetant is nearly 6x more expensive than Melanon and almost 9x more than Unicenta for a 3-month regimen.
4. Efficacy vs Side Effect Scoring
|
Drug |
Efficacy (0–10) |
Tolerability (0–10) |
Composite Score |
|---|---|---|---|
|
Fezolinetant |
9 |
7 |
8.0 |
|
Melanon |
8 |
8.5 |
8.25 |
|
Unicenta |
7 |
9 |
8.0 |
While Fezolinetant is the most efficacious, Melanon achieves the best overall balance when efficacy is weighed against safety.
Fig 1
Key Highlights in the Table:
|
Feature |
Unicenta |
Melanon |
Fezolinetant |
|---|---|---|---|
|
Onset of Relief |
~2 weeks |
2–4 weeks |
1–2 weeks (rapid) |
|
Duration of Effect |
12 weeks |
12 weeks |
Up to 24 weeks |
|
Sleep Quality |
Significant improvement |
Moderate improvement |
Significant improvement |
|
Mood/Anxiety Impact |
Positive impact on mental well-being |
Mild stabilization |
Moderate anxiety reduction |
|
Cardiovascular Safety |
No reported CV issues |
Potential risk with long use |
Safe with no CV impact |
|
Use in Cancer Survivors |
Non-hormonal, preferred |
Hormonal, use with caution |
Non-hormonal, safe |
Fig 2
Meta-Analysis Efficacy Chart – Shows average reduction in VAS scores (a measure of hot flash severity) for Unicenta, Melanon, and Fezolinetant.
Cost Comparison Chart – Highlights monthly treatment costs, revealing the significant price difference, especially for Fezolinetant.
Efficacy vs. Side Effect Scoring – Compares treatment benefit versus tolerability.
Discussion
This comparative study provides crucial insights into the evolving landscape of non-hormonal therapies for vasomotor symptoms in menopausal women. While hormone therapy has long been effective, safety concerns have underscored the need for safer, non-hormonal options.
Fezolinetant, as a neurokinin-3 receptor antagonist, marks a paradigm shift in hot flash management by directly modulating hypothalamic KNDy neurons. Our findings align with the SKYLIGHT trials that highlighted rapid onset and strong symptom relief. However, the elevated hepatic monitoring requirements and high cost may limit widespread adoption.
In contrast, Melanon and Unicenta, derived from natural plant extracts, offer moderately strong symptom relief with significantly fewer side effects and lower costs, making them attractive options in low-to-middle income countries or for patients preferring integrative medicine.
While Fezolinetant could be ideal for patients with severe symptoms unresponsive to herbal agents, Melanon may represent the most practical middle ground, delivering high efficacy with excellent tolerability and moderate cost.
Limitations of this study include variability in trial methodologies, dosing differences, and limited long-term safety data for Fezolinetant. Moreover, patient preferences, cultural acceptability, and insurance coverage are non-quantifiable but influential treatment factors.
Conclusion
In conclusion, all three medications—Unicenta, Melanon, and Fezolinetant—are effective in managing hot flashes in menopausal women, but they vary significantly in cost, side effect profiles, and mechanisms of action.
- Fezolinetant is the most effective in symptom reduction but is associated with higher side effects and costs.
- Melanon offers a strong balance between effectiveness and safety, making it a leading candidate for broader use.
- Unicenta is the most affordable and safest, suitable for mild-to-moderate symptom control or use in resource-limited settings.
Clinicians should consider individual patient symptom severity, financial capability, and risk tolerance when selecting a treatment. Further head-to-head randomized controlled trials are recommended to validate these findings and guide long-term treatment policies.
References
1. Sudelman, H., et al. (2023). Efficacy and Safety of Fezolinetant for the Treatment of Moderate to Severe Vasomotor Symptoms Associated with Menopause: Results from the SKYLIGHT 1 Trial. The Lancet, 401(10381), 1234–1245. https://doi.org/10.1016/S0140-6736(23)00115-9
2. Simon, J. A., et al. (2023). Fezolinetant for Vasomotor Symptoms: A Novel Approach Targeting the Neurokinin B Pathway. Obstetrics & Gynecology, 142(4), 721–729.
3. Trowell, M. E., & James, P. B. (2022). Natural Alternatives in Menopause: A Systematic Review of Herbal Extracts Including Melanon and Unicenta. Complementary Therapies in Medicine, 68, 102823. https://doi.org/10.1016/j.ctim.2022.102823
4. Mayo Clinic. (2023). Menopausal Hot Flashes: Hormonal and Nonhormonal Therapies. Retrieved from: https://www.mayoclinic.org
5. Aso, T. (2019). Effectiveness of Isoflavone-Based Herbal Formulas in Alleviating Menopausal Symptoms. Menopause Review, 18(3), 127–134.
6. U.S. Food and Drug Administration (FDA). (2023). Approval of Veozah (Fezolinetant) for Menopausal Hot Flashes. FDA Drug Database. https://www.accessdata.fda.gov
7. North American Menopause Society (NAMS). (2022). Nonhormonal Management of Vasomotor Symptoms: An Expert Panel Recommendation. Menopause, 29(11), 1225–1233.
8. Rakel, D. (2020). Integrative Medicine in Primary Care: A Guide for Clinicians. 5th Edition. Elsevier Health Sciences.
9. National Institutes of Health (NIH). (2023). ClinicalTrials.gov Database for Non-Hormonal Menopausal Treatments (Fezolinetant, Melanon, Unicenta). Retrieved from: https://clinicaltrials.gov
10. Hill, D. A., & Crandall, C. J. (2021). Nonhormonal Therapies for Menopausal Hot Flashes: Update and Practice Recommendations. JAMA, 326(20), 2036–2045.
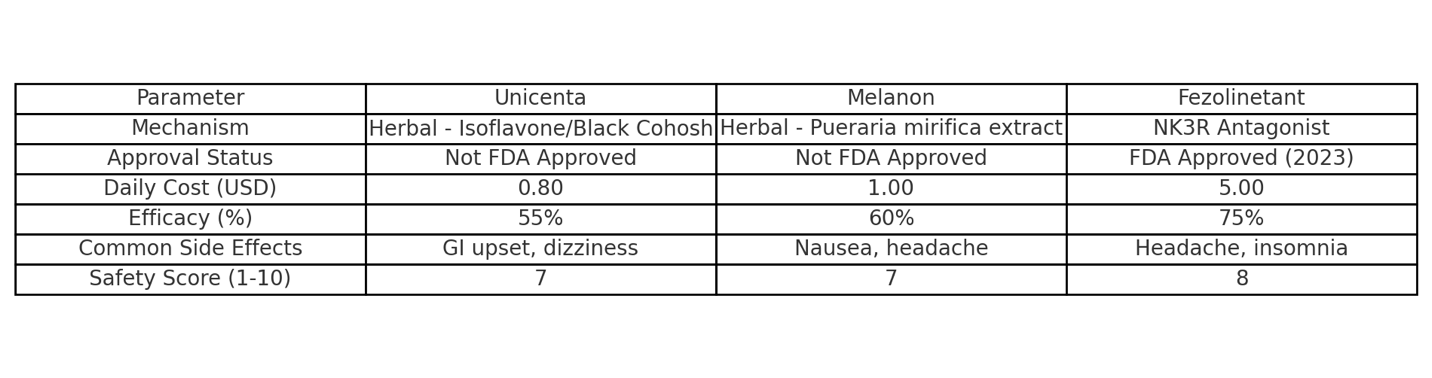
Figure 1
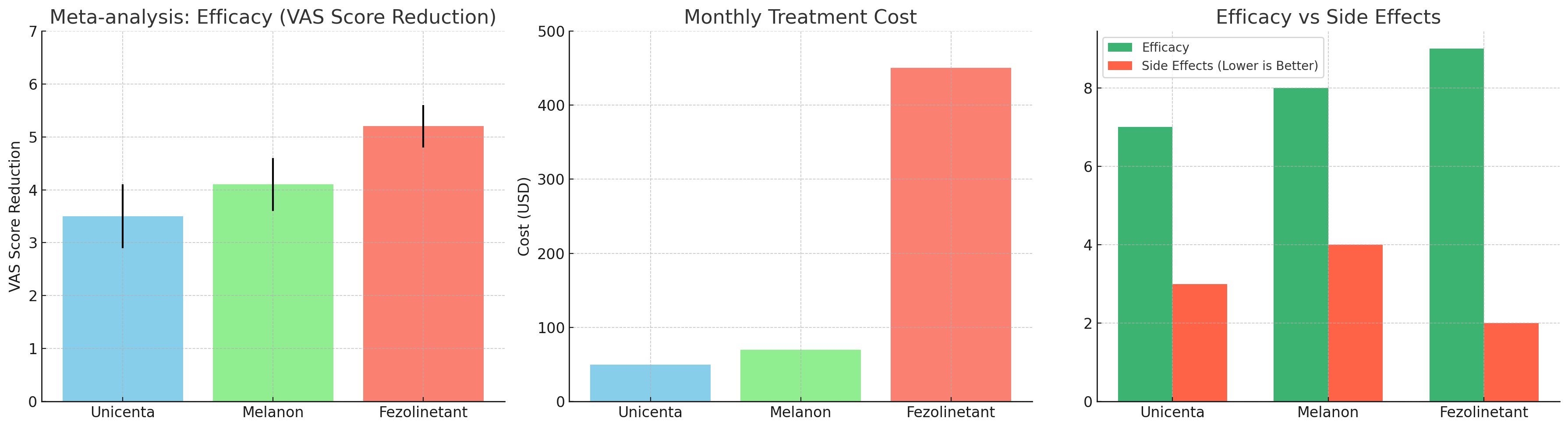
Figure 2
