Cytokeratin 19 Expression in Different Brain Cysts Subtypes
Cytokeratin 19 Expression in Different Brain Cysts Subtypes
Hamza Abu-Farsakh 1, Sbeih, Ibrahim2; Altheeb, Ruba3; Sbeih, Asil4, Abu-Farsakh, Hussam5*
1) Jordan University. Medical school
2) Chairman of Neurosurgery Department, Farah Hospital, Amman, Jordan
3) Intern at Royal Jordanian Medical Services, Amman, Jordan
4) Consultant neurosurgery in Private practice, Amman, Jordan
5) Consultant Pathologist, First Medical lab, Amman, Jordan
*Correspondence to: Hussam Abu Farsakh, Consultant Pathologist, First Medical lab, Amman, Jordan.
ORCID ID 0000-0002-5463-4979
Copyright.
© 2025 Hussam Abu Farsakh This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 30 June 2025
Published: 11 July 2025
DOI: https://doi.org/10.5281/zenodo.16312025
Abstract
Background: Intracranial brain cysts may have a congenital origin and possibly an ectodermal origin. The exact origin of some of these cysts is still obscure. Cytokeratin 19 is a 40 kDa protein specifically expressed in the periderm, a transiently superficial layer enveloping the developing epidermis.
Material and methods: Cytokeratin 19 staining was performed on all cystic brain lesions diagnosed in our lab, totaling 105 brain cystic lesions. These included 28 epidermoid cysts (tumor), 16 colloid cysts, 6 arachnoid cysts, 7 Rathke's cleft cysts, 2 neuroglial cysts, 44 craniopharyngiomas, one neurenteric cyst, one dermoid cyst.
Results: Cytokeratin 19 was expressed in 28 out of 28 epidermoid cysts, 16 out of 16 colloid cysts, 6 out of 6 arachnoid cysts, 7 out of 7 Rathke's cleft cysts, 2 out of 2 neuroglial cysts, and 44 out of 44 craniopharyngiomas, as well as one neurenteric cyst and one dermoid cyst.
Conclusions: Cytokeratin 19 is expressed in almost all brain cysts examined. However, it does not differentiate one brain cyst type from another. This expression is probably because cytokeratin 19 is expressed in embryological epithelial cells of the epidermis, which may be the origin of most brain cysts. This finding point that almost all brain cysts are congenital in origin.
Cytokeratin 19 Expression in Different Brain Cysts Subtypes
Introduction
Intracranial cysts are common findings in the general population, particularly during childhood. (1) These cysts exhibit diverse histopathology presentations; they vary in location and symptoms. (2) Studying the origin and immunobiological features of brain cysts may help understand their pathogenesis and lead to advancement in treatment methods.
Cytokeratin 19 (K19, CK19) is a type 1 acidic cytoplasmic intermediate filament (IF) protein found in epithelial cells and, like other keratins, it plays a crucial role in preserving their structural integrity. (3,4). K19 structure contains 399 amino acids made up of 3 domains: a head, a central rod, and a unique tail, The uniqueness of a K19 tail lies in the fact that it’s a short helical C-terminal domain, unlike all the other keratins which typically possess a longer tail. K19 is the smallest IF protein at 40 kDa due to its notably short carboxyl-terminal tail (13-residue long). (4–7)
Cytokeratin 19, among other lightweight intermediate filaments (IFs), is typically found in simple non-stratified epithelia, including the lining of the intestines, kidney ducts, secretory gland cells, mesothelium, gallbladder, hepatic and pancreatic ducts, endometrium, fallopian tubes, breast, bladder, lungs, thyroid epithelium, and ovarian epithelium. (6,8)
The periderm is a specialized layer that emerges in the ectoderm around the fourth week of development. This layer exists temporarily during embryonic development and is shed before birth. It serves as a non-adhesive surface, preventing epithelial cell-to-cell adhesions that could lead to birth defects and fusions. (9)
In the early stages of embryological development, K19-positive embryological cells in the ectoderm can be identified as early as 6-8 days. However, their presence gradually diminishes as development progresses, eventually becoming undetectable in fetal skin by approximately weeks 18-20 of fetal life. (10)
Numerous studies have suggested that K19 could serve as an indicator of pluripotent stem cells (7,11). One study supporting this theory specifically focused on labeling the bulge area of the hair root sheath (7), a well-known location for housing stem cells (12). Stem cells are characterized by their extended cell cycle and infrequent division. Due to their longevity, pluripotent stem cells also retain H3 thymidine for an extended time. In a study, a comparison between foreskin samples obtained from a 2-day-old donor and a 42-year-old donor revealed a significant decrease in K19-retaining cells in the older donor, supporting the notion that these cells may indeed be stem cells (7). The observed decline of these cells over time aligns with the idea that as individuals age, the presence of stem cells naturally decreases. This finding explains why wounds and burns tend to take longer to heal in older individuals, as the diminished population of these cells may contribute to a slower regenerative response. (13,14)
Material and Methods
All cases diagnosed as brain cysts in our lab from 2005 to 2023 were reviewed. The types of cysts that were identified are as follows: 28 epidermoid cysts (tumor), 16 colloid cysts, 6 arachnoid cysts, 7 Rathke’s cleft cysts, 44 craniopharyngioma, 2 neuroglial cysts, one neurenteric cyst, and one dermoid cyst.
Cytokeratin 19 (Dako: 1/100) staining was performed on all cases, with one representative paraffin block of formalin-fixed sections selected for each.
Results
Cytokeratin 19 was expressed in 28 out of 28 epidermoid cysts [Figure 1A], 16 out of 16 colloid cysts [Figure 1B], 6 out of 6 arachnoid cysts, 7 out of 7 Rathke’s cleft cysts, 2 out of 2 neuroglial cysts [Figure 1C]and 44 out of 44 craniopharyngiomas [Figure 1D], one neuroenteric cyst and one dermoid cyst.
Figure 1 (A) positive K19 staining of an epidermoid cyst (B) positive K19 staining of a colloid cyst (C) positive K19 staining of a neuroglial cyst (D) positive K19 staining of craniopharyngioma
Discussion
Brain cysts have been theorized to have embryological and possibly ectodermal origins. In this study, we provide further support for this theory through these cysts' strong positivity of cytokeratin 19.
In this review, we will discuss the value and significance of cytokeratin 19 in each type of brain cyst.
Craniopharyngiomas are benign primary brain cysts typically found in childhood. Despite their benign nature, these cysts often adhere to surrounding structures, including the hypothalamus, infundibular stalk, and basal blood vessels. (15) The adamantinomatous subtype of craniopharyngiomas exhibits genetic and histological similarities to tumors found in the jaw, such as calcifying odontogenic cysts and ameloblastomas. This resemblance suggests a potential origin from odontogenic epithelium. (16,17)
It is theorized that craniopharyngiomas arise from remnants of either the craniopharyngeal duct or Rathke's pouch, both of which originate from the stomadeum, a structure derived from the ectodermal surface. (18) During embryonic development, Rathke's pouch and the inferior infundibulum come into contact around the fourth week and subsequently fuse by the eighth week, forming the craniopharyngeal canal. (19,20) This embryological theory finds support in the discovery of epithelial cells between the roof of the pharynx and the third ventricle floor, primarily along the anterior part of the infundibulum and the superior anterior part of the adenohypophysis. These are sites previously occupied by Rathke's pouch and the craniopharyngeal canal. (19) Positive immunostaining for K19 provides further support for this embryological origin. K19 persists in the periderm and basal layer until 18-20 weeks of gestation when it completely disappears from the skin. (21) This observation could suggest an embryonal epithelial origin for craniopharyngioma.
While it is believed that craniopharyngiomas and Rathke's cleft cysts both originate from Rathke's pouch, they are typically easily distinguishable from each other. However, in some cases, the cysts might present with epithelial squamous or columnar neoplastic cells, without the characteristic features such as nodules of craniopharyngiomas or the unambiguous cells of Rathke's cleft cysts. (22)
In 2002, a study investigated cytokeratin 19 expression in these two cysts among other cytokeratins. Both cysts tested positive for K19, but craniopharyngiomas had >50% of their cells positively stained, while Rathke's cleft cyst cells were only 25%-50% positively stained. This is an important observation, as the origin of Rathke's cleft cyst is a subject of debate, it's endodermal or ectodermal in origin, but it's generally accepted to be congenital. (22–24)
Epidermoid cysts are rare, accounting for 1-2% of all intracranial tumors. These congenital tumors exhibit a very slow growth pattern, similar to that of epidermal cells. (25) It is hypothesized that they develop from residual epidermal cells abandoned after the neural groove's closure and the ectoderm's separation from the neural tube, which happens between 3 and 4 weeks of development. (26)
Arachnoid cysts are the most prevalent type of intracranial cysts. (27) The origin of arachnoid cysts has been debated for quite some time. Various studies have proposed different theories. They may be secondary due to acquired factors such as trauma, infections, or hemorrhage. Or primary congenital in origin. (28,29) Our discovery of CK19 within these cysts suggests the congenital origin of those cysts.
Neuroglial cysts are very rare cysts recognized as congenital benign neuroepithelial cysts. (30) It is widely believed that segments of the neural tube that correspond to tela choroidea are displaced towards the subarachnoid space. This may explain their presence in diverse locations and the variability in their structure. (31)
Colloid cysts once believed to originate from the ectoderm, are now recognized as having an endodermal origin. (32) A study conducted in 1989 found that colloid cysts had an intense reaction to CK 19, unlike the choroid plexus, which initially led to the belief that they were ectodermal in origin. (33) However, recent studies have strongly suggested an endodermal origin. An extensive recent immunohistochemistry study of colloid cysts and the choroid plexus reveals more similarities between the cysts' staining and that of the respiratory tract and certain endodermal differentiated cysts. While this points towards an endodermal origin, some similarities with certain ectodermal differentiated cysts were also observed. (34)
Neurenteric intracranial cysts are extremely rare congenital benign cysts of endodermal origin. The exact cause of these cysts is not fully understood. (35) According to an accepted theory, a failure in the separation of the notochord and foregut results in the incorporation of primitive endodermal cells into the notochord. These displaced clusters of alimentary tissue eventually develop into cysts. However, this theory fails to account for the varied locations where these cysts can originate, particularly within the supratentorial region. (35,36)
Dermoid cysts are benign, slow-growing neoplasms, that are established as a congenital anomaly that forms due to ectopic ectodermal tissue within the brain at the time of the neural groove closure. (37)
In Summary: Cytokeratin 19 is present in primitive epithelial cells. The presence of K19 staining in brain cysts mentioned above does not necessarily support or rule out ectodermal or endodermal origin. However, it indicates that these cysts are of embryologic origin, rather than acquired cysts. Additional immunohistological investigations are suggested to offer a clearer understanding of the source of these cysts.
References
1. Pereira RG, de Freitas Ribeiro BN, de Lima Hollanda RT, de Almeida LB, Simeão TB, Marchiori E. Non-neoplastic intracranial cystic lesions: Not everything is an arachnoid cyst. Radiol Bras. 2021;54(1):49–55.
2. Osborn AG, Preece MT. Intracranial cysts: Radiologic-pathologic correlation and imaging approach. Vol. 239, Radiology. 2006. p. 650–64.
3. Mujyambere B, Jayaraj R, Suja S. Cytokeratin 19 (CK19) as a marker for Epithelial Differentiation and Malignant Transformation: Its Clinical relevance in Diagnosis, Prognosis and Treatment response monitoring. 2018.
4. Fradette J, Germain L, Seshaiah P, Coulombe PA. The Type I Keratin 19 Possesses Distinct and Context-dependent Assembly Properties* [Internet]. 1998. Available from: http://rsb.info.nih.gov/
5. Bader BL, Magin TM, Hatzfeld M, Franke WW. Amino acid sequence and gene organization of cytokeratin no. 19, an exceptional tail-less intermediate filament protein. Vol. 5, The EMBO Journal. 1986.
6. Stasiak PC, Purkis PE, Leigh IM, Lane EB. Keratin 19: Predicted Amino Acid Se uence and Broad Tissue Distribution Suggest it Evolve 1 from Keratinocyte Keratins. 1989.
7. Michel M, Torok N, Godbout MJ, Lussier M, Gaudreau P, Royal A, et al. Keratin 19 as a biochemical marker of skin stem cells in vivo and in vitro: keratin 19 expressing cells are differentially localized in function of anatomic sites, and their number varies with donor age and culture stage. J Cell Sci. 1996 May 1;109(5):1017–28.
8. Fradette J, Germain L, Seshaiah P, Coulombe PA. The Type I Keratin 19 Possesses Distinct and Context-dependent Assembly Properties* [Internet]. 1998. Available from: http://rsb.info.nih.gov/
9. Moreci RS, Lechler T. Epidermal structure and differentiation. Vol. 30, Current Biology. Cell Press; 2020. p. R144–9.
10. Birgitte Lane E, Bartek J, Purkis PE, Leigh IM. Keratin Antigens in Differentiating Skin.
11. Bartek J, Bartkov J, Rejthar A, Kovai~ik ~ J, Luka~ Z, Vojti~ek B. Differential expression of epithelial tissues revealed antibodies keratin 19 in normal human by monospecific monoclonal. Vol. 18, Histochemical Journal. 1986.
12. Joulai Veijouye S, Yari A, Heidari F, Sajedi N, Ghoroghi Moghani F, Nobakht M. Bulge Region as a Putative Hair Follicle Stem Cells Niche: A Brief Review [Internet]. Vol. 46, Iran J Public Health. 2017. Available from: http://ijph.tums.ac.ir
13. Grove GL. Age-Related Differences in Healing of Superficial Skin Wounds in Humans. Vol. 272, Arch Dermatol Res. 1982.
14. Germain L, Rouabhia M, Guignard R, Carrier L, Bouvard V, Auger FA. Improvement of human keratinocyte isolation and culture using thermolysin. Burns. 1993.
15. Yachnis AT. Craniopharyngioma: Embryology, Pathology, and Molecular Aspects. Embryology, Pathology, and Molecular Aspects. In: Craniopharyngiomas: Comprehensive Diagnosis, Treatment and Outcome. Elsevier Inc.; 2015. p. 95–105.
16. Sekine S, Takata T, Shibata T, Mori M, Morishita Y, Noguchi M, et al. Expression of enamel proteins and LEF1 in adamantinomatous craniopharyngioma: evidence for its odontogenic epithelial differentiation. Vol. 45, Histopathology. 2004.
17. Song-tao Q, Xiao-rong Y, Jun P, Yong-jian D, Jin L, Guang-long H, et al. Does the calcification of adamantinomatous craniopharyngioma resemble the calcium deposition of osteogenesis/odontogenesis? Histopathology. 2014 Feb;64(3):336–47.
18. Nourbakhsh A, Brown B, Vannemreddy P, Lian T, Nanda A, Guthikonda B. Extracranial infrasellar ectopic craniopharyngioma: A case report and review of the literature. Skull Base. 2010;20(6):475–80.
19. Prabhu VC, Brown HG. The pathogenesis of craniopharyngiomas. Vol. 21, Child’s Nervous System. 2005. p. 622–7.
20. Hanna E, Rock L, Sas A, Weissman J. Sphenoclival Rathke’s Cleft Cysts: Embryology, Clinical Appearance and Management.
21. Birgitte Lane E, Bartek J, Purkis PE, Leigh IM. Keratin Antigens in Differentiating Skin.
22. Xin W, Rubin MA, Mckeever PE. Differential Expression of Cytokeratins 8 and 20 Distinguishes Craniopharyngioma From Rathke Cleft Cyst. Vol. 126, Arch Pathol Lab Med. 2002.
23. Kalfas F, Scudieri C. Endodermal cysts of the central nervous system: Review of the literature and a case report. Asian J Neurosurg. 2020 Dec;15(04):989–96.
24. A Mackenzie IR, Gilbert JJ, A Mackenzie J J Gilbert IR. Cysts of the neuraxis of endodermal origin. Vol. 199, Neurosurgery, and Psychiatry.
25. Patibandla M, Yerramneni V, Mudumba V, Manisha N, Addagada G. Brainstem epidermoid cyst: An update. Asian J Neurosurg. 2016 Sep;11(03):194–200.
26. Berger MS, Wilson CB. Epidermoid cysts of the posterior fossa. Vol. 62, J Neurosurg. 1985.
27. Pereira RG, de Freitas Ribeiro BN, de Lima Hollanda RT, de Almeida LB, Simeão TB, Marchiori E. Non-neoplastic intracranial cystic lesions: Not everything is an arachnoid cyst. Radiol Bras. 2021;54(1):49–55.
28. Pascual-Castroviejo I, Roche MC, Martinez Bermejo A, Arcas J, Garcia Blazquez M. Primary intracranial arachnoidal cysts A study of 67 childhood cases. Vol. 7, Child’s Nerv Syst. 1991.
29. Robinson RG. Congenital Cysts of the Brain: Arachnoid Malformations. Vol. 4, Progr. neurol. Surg. Karger; 1971.
30. Robles LA, Paez JM, Ayala D, Boleaga-Duran B. Intracranial glioependymal (neuroglial) cysts: a systematic review. Vol. 160, Acta Neurochirurgica. Springer; 2018. p. 1439–49.
31. Friede RL, Yasargil MG. Supratentorial intracerebral epithelial (ependymal) cysts: Review, case reports, and fine structure. J Neurol Neurosurg Psychiatry. 1977;40(2):127–37.
32. Hasegawa M, Nouri M, Nagahisa S, Hayashi T, Adachi K, Hirose Y, et al. Neuroepithelial cyst of the fourth ventricle. Child’s Nervous System. 2015 Jan 1;31(1):155–9.
33. Kondziolka D, Bilbao JM. An immunohistochemical study of neuroepithelial (colloid) cysts. Vol. 71, J Neurosurg. 1989.
34. Lach B, Scheithauer BW, Gregor A, Wick MR. Colloid cyst of the third ventricle. J Neurosurg. 1993 Jan;78(1):101–11.
35. Preece SMT, Osborn AG, Chin SS, Preece MT, Smirniotopoulos JG. Pathology Spectrum Intracranial Neurenteric Cysts: Imaging and Intracranial Neurenteric Cysts: Imaging and Pathology Spectrum [Internet]. Vol. 27, AJNR Am J Neuroradiol. 2023. Available from: http://www.ajnr.org/content/27/6/
36. Harris CP, Dias MS, Brockmeyer DL, Townsend JJ, Willis BK, Apfelbaum RI. Neurenteric cysts of the posterior fossa: recognition, management, and embryogenesis. Neurosurgery. 1991 Dec;29(6):893–7; discussion 897-8.
37. Jyoti Das C, Tahir M, Debnath J, Shyam Pangtey G. Ruptured intracranial dermoid. BMJ Case Rep. 2009;2009:bcr2006109835b..
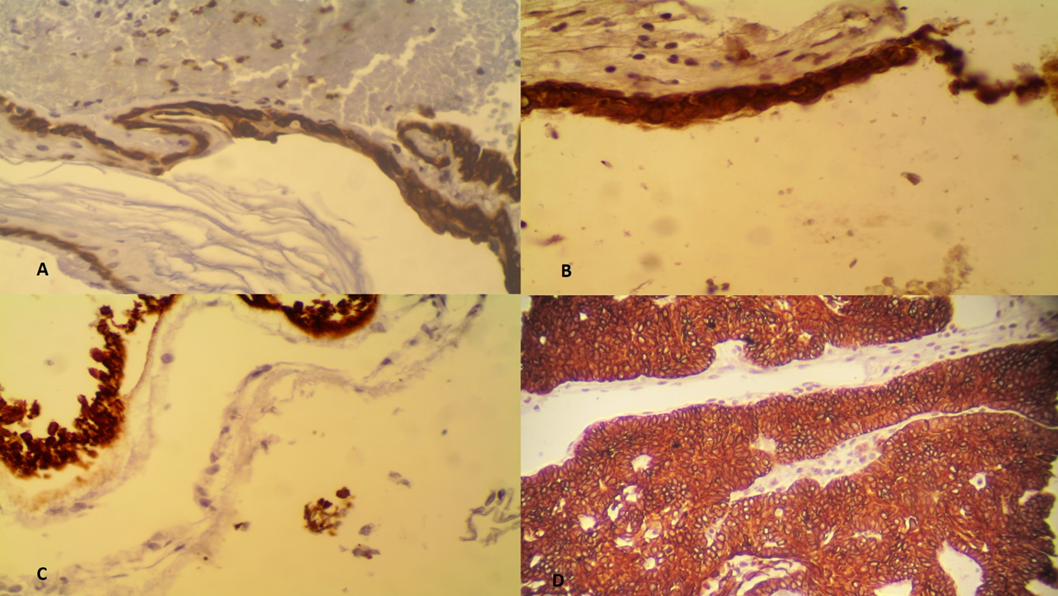
Figure 1
