Endoscopic Resection of a Large Ampullary Adenoma
Endoscopic Resection of a Large Ampullary Adenoma
Ethan Barkley, BS1*, Corey Mealer, BS1, Marissa Jansen, BS1, Manjakkollai P. Veerabagu, MD2, Leila Kutob, MD3
1. Medical Student, College of Medicine; Medical University of South Carolina
2. Affiliate Associate Professor; Medical University of South Carolina
3. AnMed Health System Department of Pathology
*Correspondence to: Ethan Barkley, BS, Medical Student, College of Medicine; Medical University of South Carolina.
Copyright
© 2025 Ethan Barkley, BS, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 14 July 2025
Published: 23 July 2025
Abstract:
An ampullary adenoma is a glandular dysplastic lesion arising from the duodenal papilla and is a pre-cancerous lesion. It is either sporadic or associated with familial adenomatous polyposis syndrome. Treatment options for ampullary adenoma include a pancreaticoduodenectomy, surgical local excision, or endoscopic ampullectomy. An endoscopic approach has been shown to be an effective treatment for these lesions while avoiding the risks that come with more invasive surgical procedures. In this report, we discuss a case of a large ampullary adenoma with low grade dysplasia treated successfully with endoscopic ampullectomy.
Key words: Ampullary adenoma, endoscopic ampullectomy.
Endoscopic Resection of a Large Ampullary Adenoma
Case
The patient is a 78-year-old female with prior history of breast cancer and remote history of intracranial aneurysmal clipping and cholecystectomy. She presented with elevated liver enzymes, common bile duct dilation on CT scan imaging and elevated cancer antigen 19-9, which had been normal initially. Subsequent testing showed progressive CA 19-9 elevation as high as 152 units/ml, after the patient was referred to an oncologist. Additionally, she could not undergo MRI or MRCP due to incompatibility of MR technology with aneurysm clips in her brain. In the setting of consistent CA 19-9 elevation, the patient underwent EGD, EUS, and ERCP procedures. EGD revealed a very large ampullary adenoma (Fig 1). EUS showed dilated 1.7cm common bile duct with a hyperechoic mass involving the ampulla. During ERCP successful sphincterotomy, brush biopsy of the distal bile duct and cold forceps biopsy of the ampulla were performed and a 10mm x 80mm fully covered self-expanding metal wall flex stent (Boston Scientific) was placed with good biliary drainage.
Tissue track samples resulted in the diagnosis of ampullary adenoma without evidence of high-grade dysplasia (Figures 4,5), and cytology from the brush biopsy showing atypical glandular cells. CA 19-9 levels decreased from 152 to 25 units/ml one month post-procedure. Due to a diagnosis of ampullary adenoma, the patient and her family were counseled on ampullectomy to prevent ampullary cancer versus watchful waiting. They elected to pursue ampullectomy. The procedure was performed under general anesthesia. A side viewing duodenoscope was advanced to the second part of the duodenum. The previously placed biliary stent was retrieved using a snare. The scope was then repositioned to visualize a large 3.5 cm ampullary adenoma (Fig 1). Cholangioscopy (spyglass Boston Scientific) also revealed possible skip lesions versus changes from the prior stent in the bile duct. Submucosal injection was then performed and the lesion lifted from the wall showing that it was not fixed to the side of the duodenum. A 27 mm Captivator snare was used to perform ampullectomy which was retrieved using a Roth net. A residual lesion on the margin was removed using a 15 Captivator, also retrieved using a Roth net. After ampullectomy, cannulation of the pancreatic duct was achieved using Jagwire Revolution (Boston Scientific), after which a 5 cm French single pigtail pancreatic stent was placed. The bile duct was also cannulated, after which double-pigtail 7 cm 10 French plastic biliary stent was placed with good biliary drainage (Figures 2,3). Margins were ablated using Argon plasma coagulation to prevent adenoma recurrence. Histopathology showed intestinal type ampullary adenoma with low grade dysplasia and no invasive adenocarcinoma (Figures 6,7). One month later, ERCP was conducted during which both the double and single pigtail stents were removed, and further interrogation of the bile duct with cholangioscopy revealed no skip lesions suggesting that the observed small changes were due to the previous metal stenting. A 60mm x 10mm self-expanding metal biliary wall stent (Boston Scientific) was placed with good biliary drainage. The patient tolerated both procedures well without any adverse event.
Figure 1 - Ampullary adenoma visualized on EGD
Figure 2 – Cannulation
Figure 3 - Stenting
Figure 4 - Initial ampullary biopsy, 40x magnification
Figure 5 - Initial ampullary biopsy, 100x magnification
Figure 6 – Ampullary adenoma tissue analysis, 40x magnification
Figure 7 – Ampullary adenoma tissue analysis, 100x magnification
Discussion
Ampullary adenoma is a glandular dysplastic lesion arising from the duodenal papilla with pre-malignant potential1. Their disease progression is similar to the colon adenoma to adenocarcinoma pathway. They can be sporadic or associated with pathological processes such as familial adenomatous polyposis (FAP). Though they carry malignant potential, their prognosis is usually more favorable to other peri-ampullary tumors of epithelial origin such as cholangiocarcinoma, or adenocarcinoma of the pancreatic head2. The diagnosis of ampullary adenoma is made using endoscopy with tissue sampling to differentiate from ampullary adenocarcinoma. Should histology indicate adenocarcinoma, some studies have suggested that differentiation between pancreatobiliary or intestinal origins has some prognostic value, with the latter exhibiting a more favorable prognosis3. Once the adenoma has been diagnosed, features that require further imaging using MRI or CT or endoscopic ultrasound include lesions of greater than 1 cm, presence of high grade dysplasia, ulceration, irregular margins, or spontaneous bleeding.
Treatment options for ampullary adenoma include a pancreaticoduodenectomy (Whipple procedure), surgical local excision, or endoscopic ampullectomy4. Though surgical resection often provides for complete resection, it is associated with much higher rates of complication than endoscopy ampullectomy5. Both the American and European society for gastrointestinal endoscopy recognize the growing role of endoscopy (ampullectomy) as the first line treatment in select cases5,6. In addition to removal of the lesion it also relieves biliary obstruction. EUS allows for the assessment of infiltration of the periampullary wall and if there is any biliary or pancreatic duct extensions and presence of metastatic lymph nodes. Additionally, an important step after ampullectomy is prophylactic pancreatic stent placement to reduce the risk of pancreatitis. The decision to perform sphincterotomy and biliary stent placement is also supported by a growing body of research supporting their use to ensure adequate biliary drainage and prevent acute cholangitis and biliary sticture4. The most serious complications of endoscopic ampullectomy are perforation, delayed bleeding, and pancreatitis7. Long term outcomes for endoscopic resection of ampullary adenoma are generally favorable and success rates are comparable to more invasive surgical resection8.
In this report, we present an interesting case of successful endoscopic removal of a very large ampullary adenoma (ampullectomy) without any adverse events and avoided higher risk more invasive surgical options.
References
1) Chini, P., Dragonov, P.V., Diagnosis and management of ampullary adenoma: the expanding role of endoscopy. World Journal of Gastrointestinal Endoscopy. 2011;3(12):241-247.
2) Martin, J.A., Haber, G.B. Ampullary adenoma: clinical manifestations, diagnosis, and treatment. Gastrointestinal Endoscopy Clinics. 2003;13(4):649-669.
3) Schirmacher, P., Büchler, M. W. Ampullary adenocarcinoma - differentiation matters. BMC Cancer. 2008;8:251.
4) Espinel, J., Pinedo, E., Ojeda, V., Guerra del Rio, M. Endoscopic management of adenomatous ampullary lesions. World Journal of Methodology. 2015;5(3):127-135.
5) Vanbiervliet, G., Strijker, M., Arvanitakis, M., Aelveot, A., Arnelo, U., Beyna, T., Busch, O., Deprez, P.H., Kunovsky, L., Larghi, A., Manes, G., Moss, A., Napolean, B., Nayar, M., Perez-Cuadrado-Lopez, E., Seewald, S., Barthet, M., van Hooft, J.E. Endoscopic management of ampullary tumors: European Society of Gastrointestinal endoscopy (ESGE) guideline. Endoscopy. 2021;53(4):429-448.
6) Chathadi, K.V., Khashab, M.A., Acosta, R.D., Chandrasekhara, V., Eloubeidi, M.A., Faulx, A.L., Fonkalsrud, L., Lightdale, J.R., Saltzman J.R., Shaukat, A.S., Wang, A., Cash, B.D., DeWitt, J.M. The role of endoscopy in ampullary and duodenal adenomas. Gastrointestinal Endoscopy. 2015;82(5):773-781.
7) Bassan, M., Bourke, M. Endoscopic ampulletomy: a practical guide. Journal of Interventional Gastroenterology. 2012;2(1):23-30.
8) Lee, J., Oh, D., Seo, D., Song, T.J., Park, D.H., Lee, S.K., Hong, S. Long term outcomes of ampullary adenoma according to resected margin status after endoscopic papillectomy. Gut and Liver. 2024;18(4):747-755.
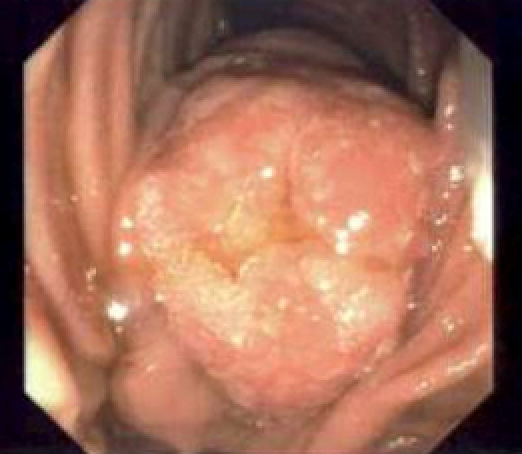
Figure 1
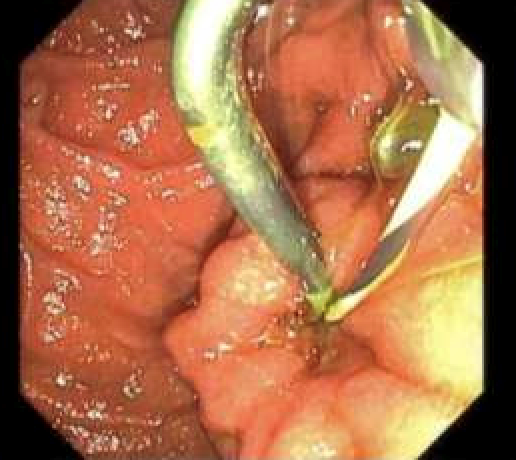
Figure 2
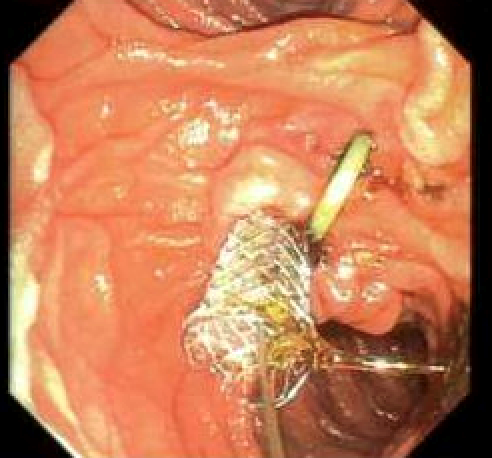
Figure 3
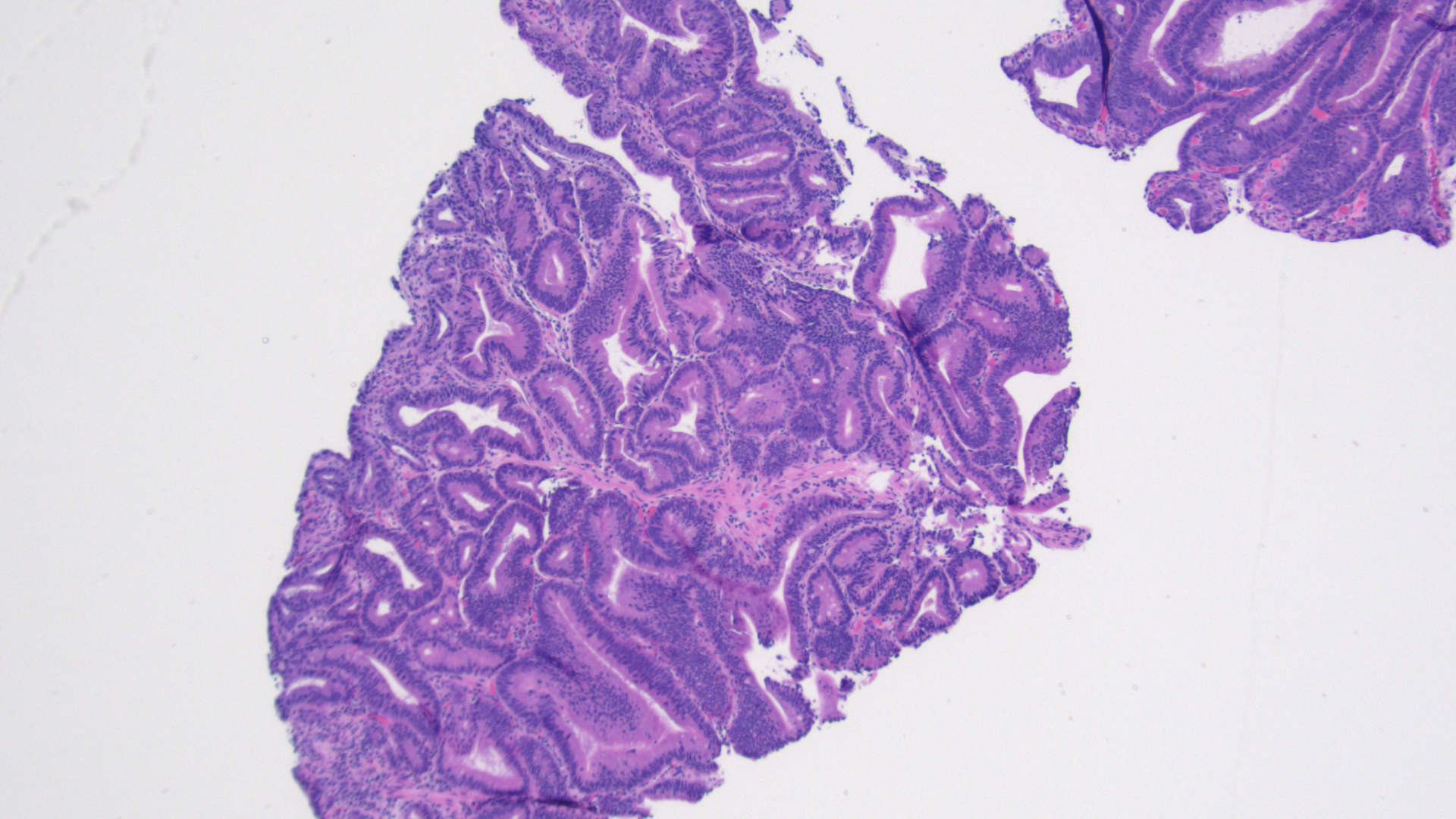
Figure 4
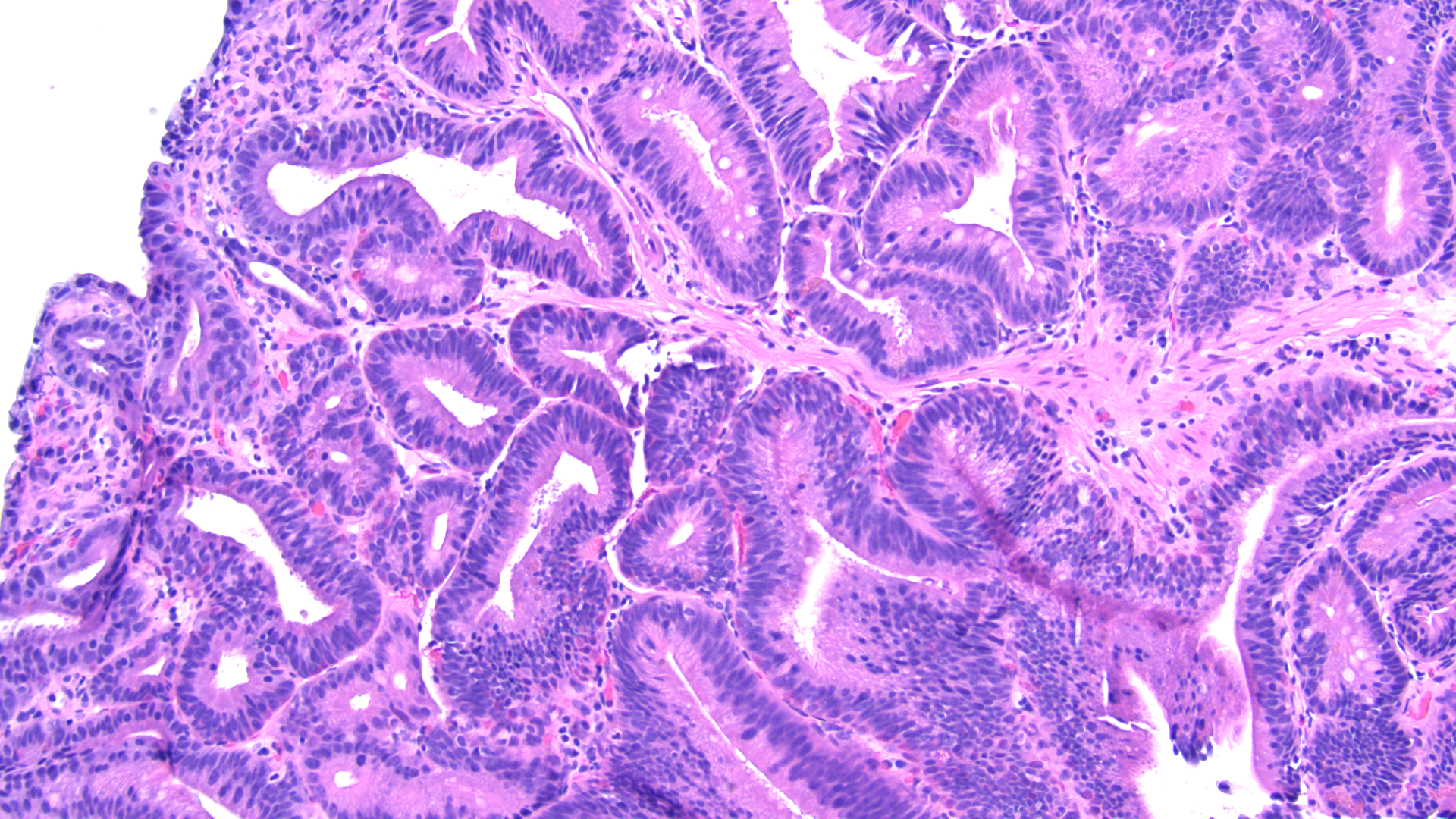
Figure 5
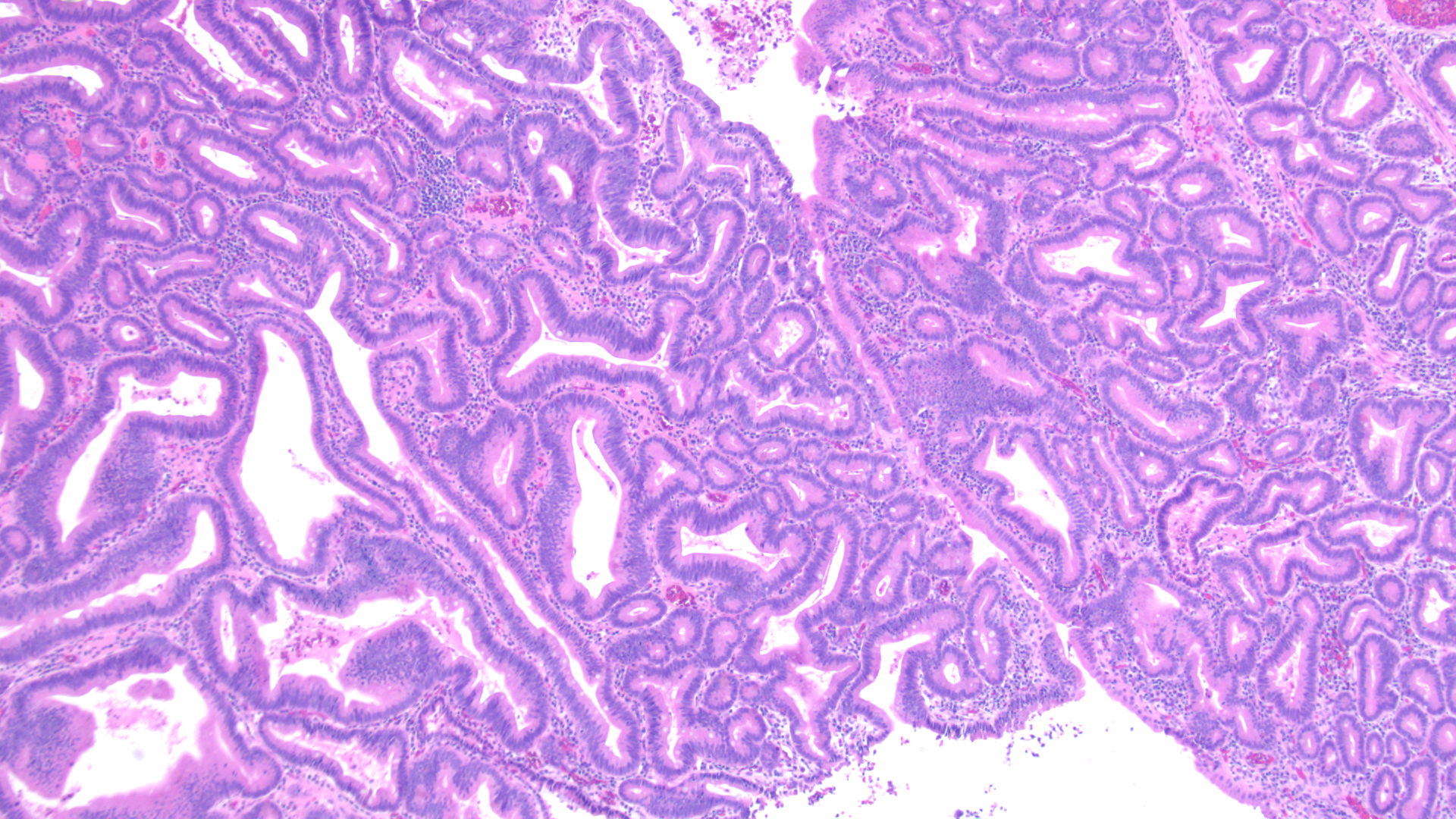
Figure 6
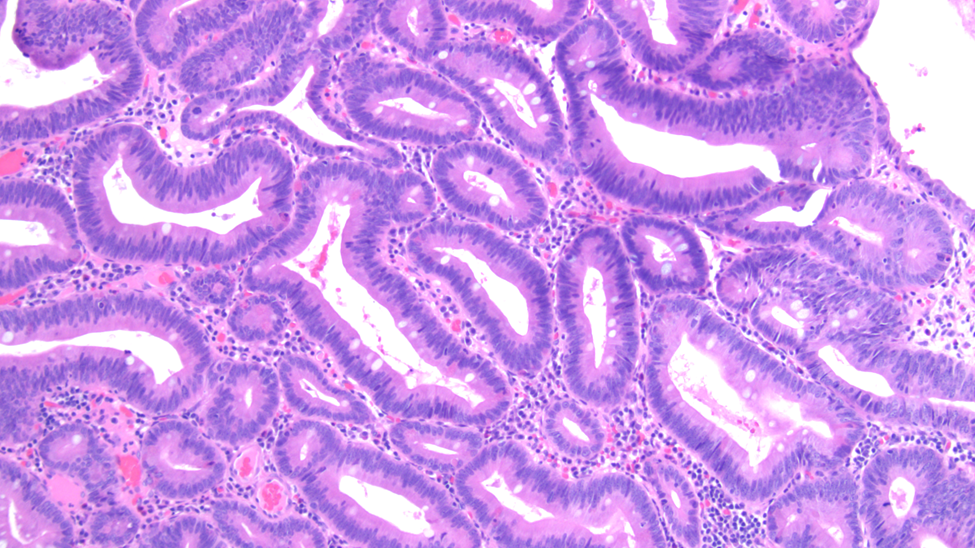
Figure 7
