Subcutaneous Panniculitis - Like T-cell Lymphoma: A Tertiary Care Institute Experience
Subcutaneous Panniculitis - Like T-cell Lymphoma: A Tertiary Care Institute Experience
Dr. Jigar Shah 1*, Dr. Atul Kothari2, Dr. Sonal Jain*3, Dr. Urvashi Shah4
1. Dr. Jigar Shah, Associate Professor, Ram Krishna Medical College Hospital and Research Centre, Bhopal, Madhya Pradesh.
2. Dr. Atul Kothari, Assistant Professor, Sri Aurobindo Medical College and PG Institute, Indore.
3. Dr. Sonal Jain, Assistant Professor, Ruxmaniben Deepchand Gardi Medical College, Ujjain, Madhya Pradesh.
4. Dr Urvashi Shah, Tutor, Department Of Community Medicine, Gmers Medical College and Hospital, Gotri, Vadodara,Gujarat.
*Correspondence to: Dr. Sonal Jain, Assistant Professor, Ruxmaniben Deepchand Gardi Medical College, Ujjain, Madhya Pradesh.
Copyright.
© 2025 Dr. Sonal Jain This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 18 July 2025
Published: 23 July 2025
Subcutaneous Panniculitis - Like T-cell Lymphoma: A Tertiary Care Institute Experience
Introduction
Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) is a rare form of primary cutaneous lymphoma characterized by cytotoxic alpha-beta T cells, which closely resemble panniculitis. In the latest (5th edition) World Health Organization (WHO) Classification of Tumours of Hematopoietic and Lymphoid Tissues, Subcutaneous Panniculitis-like T-cell Lymphoma (SPTCL) is recognized as a distinct subtype of non-Hodgkin lymphoma [1]. Subcutaneous panniculitis-like T-cell lymphoma (SPTCL) is a rare condition that can occur in individuals of all ages but is most commonly diagnosed in young adults and middle-aged individuals, with a median age of onset around 36 years. SPTCL primarily affects the subcutaneous fat, presenting as solitary or multiple erythematous nodules and plaques, often localized to the lower extremities but potentially more widespread. Systemic symptoms may include fever, chills, malaise, weight loss, and hepatosplenomegaly. A significant association exists between SPTCL and hemophagocytic lymphohistiocytosis (HLH), a severe hyperinflammatory condition reported in approximately 30-40% of cases. Histologically, SPTCL is marked by CD8+ T-cells expressing αβ T-cell receptors (TCRs) that infiltrate subcutaneous adipose tissue, encircling adipocytes in a characteristic pattern and causing adipocyte necrosis. The immunophenotype typically includes CD8+, CD56-, CD30-, granzyme B+, TIA-1+, and beta F1+ markers. Molecular clonality assays are essential to differentiate SPTCL from γδ T-cell lymphomas, which have a more aggressive course and are considered distinct entities. Autoimmune conditions are present in about 20% of cases, with lupus erythematosus panniculitis (LEP) often considered in the differential diagnosis due to overlapping clinical and histopathological features. Histologically, neoplastic CD8+ T cells that express beta F1 typically surround and disrupt individual adipocyte membranes. While most cases have a favorable prognosis and follow an indolent course, 15% to 20% may develop hemophagocytic syndrome (HPS) [2-4]. The present study is to determine the relative frequency of Subcutaneous Panniculitis-Like T-cell Lymphoma, its correlation with Immunohistochemistry (IHC) and clinical Diagnosis.
Material and Methods
This 5 year study was carried out in Department of General Pathology, RKDF Medical College Hospital and Research Centre, Bhopal, India. The histopathological material (slides and blocks) were accrued from Department of General Pathology. This study included biopsy material of patients registered within RKDF and from those referred from outside. The clinical information and laboratory parameters were obtained from clinical workstation and charts from medical records department or from the biopsy request forms for outside referral cases. All cases of primary cutaneous lymphomas diagnosed in our department were included in our study. Cases on which further studies were not possible due to preservation or storage artifacts in the histopathological tissue material or paraffin embedded block and these cases were not included for special studies (For Notch-1 and Fox p1). However their clinic-pathological profiles were included in the study. Following parameters were recorded from each patient: Morphological subtype, demographic details, Complete blood counts including hemoglobin level, total leukocyte count (TLC), differential count (DC) and platelet count (PLC); serum lactate dehydrogenase (LDH) levels and erythrocyte sedimentation rate(ESR), Special studies including bone marrow aspiration, trephine biopsy and TCR rearrangement studies (if done). Data entry and all statistical analysis were done using Epi-info software. Descriptive statistics such as frequency and percentage were used. Categorical variables were analysed using χ2 test with Yates continued correction and Fischer’s exact test. A p value of < 0.05 was considered statistically significant.
Results
A total of 115 cutaneous lymphomas were diagnosed, out of 11320 skin biopsies, during 5 years in the Department of General Pathology, RKDF Medical College Hospital and Research Centre. The frequency of cutaneous lymphoma in our study was 1.01 per 100 biopsy specimen. This lymphoma comprised 9.8% (115 of 1164) of all extra nodal Non-Hodgkin’s lymphomas in the same duration. Of these 89% (102 of 115 cases) were cutaneous T cell lymphoma (CTCL), 6% (7 of 115 cases) were cutaneous B cell lymphoma and 0.9% (1 of 115 cases) were Blastic plasmacytoid dendritic cell neoplasm. Of these, Mycosis fungoides comprised 44 % (51 cases) followed by SPTCL 21% (24 cases) and CD 30 positive lymphoproliferative disorder 17.4% (20 cases).
Table - 1: NUMBER AND FREQUENCY OF SUBTYPES OF CUTANEOUS LYMPHOMAS IN OUR STUDY (N=115).
|
Sr. no |
Types |
Number |
Frequency |
|
1 |
Mycosis Fungoides (MF) |
51 |
44.3% |
|
|
Folliculotropic MF (FMF) |
02 |
1.70% |
|
|
Pagetoid reticulosis (PR) |
01 |
0.86% |
|
2 |
Sézary Syndrome (SS) |
01 |
0.86% |
|
3 |
Cutaneous CD 30 positive lymphoproliferative disorder |
20 |
17.3% |
|
|
Primary Cutaneous ALCL (PCALCL) |
13 |
11.3% |
|
|
Lymphomatoid Papulosis (LyP) |
07 |
6.08% |
|
4 |
Subcutaneous Panniculitis like T-cell lymphoma (SPTCL) |
24 |
20.9% |
|
5 |
Extra nodal NK/T cell lymphoma, nasal type |
01 |
0.86% |
|
6 |
Cutaneous Gamma Delta T cell lymphoma (CGDTCL) |
04 |
3.47% |
|
7 |
Cutaneous CD4 positive small/medium sized pleomorphic T cell lymphoma (PCSMTCL) |
01 |
0.86% |
|
8 |
Primary cutaneous marginal zone B cell lymphoma (PCMZBL) |
02 |
1.73% |
|
9 |
Primary cutaneous follicle center lymphoma |
01 |
0.86% |
|
10 |
Primary cutaneous diffuse large B cell lymphoma, other |
04 |
3.47% |
|
11 |
Blastic plasmacytoid dendritic cell neoplasm |
01 |
0.86% |
|
12 |
Other cases |
05 |
4.34% |
Table - 2: INTERNATIONAL PROGNOSTIC INDEX {IPI} OF CUTANEOUS LYMPHOMAS (EXCLUDING MF/SS).
|
IPI
|
SPTCL N=22 |
ALCL N=10 |
LyP N=2 |
CGDTCL N=3 |
CMZBCL N=2 |
CDLBCL, other N=2 |
|
Low Low Intermediate High Intermediate High |
9% 77% 14% --- |
30% 60% 10% --- |
--- 100% --- --- |
--- 100% --- --- |
--- --- 100% --- |
--- --- 100% --- |
The median age of presentation was 39 years (range 3 to 88 years). More than 35% of the cases were seen in 4th – 6th decade. There was a male preponderance with male to female ratio of 1.19. Pediatric (<18 years) cutaneous lymphomas constituted 15.6% (18 of 115 cases). Of these the most common was mycosis fungoides (44.4%) followed by SPTCL (33.3%) and ALCL (11.1%). There was a female preponderance in pediatric lymphoma with male to female ratio of 0.8:1. Regarding frequency of subtypes of cutaneous lymphomas according to Table - 1, Mycosis fungoides was the most common subtype of all cutaneous lymphomas comprising 44% (51 of 115 cases), followed by SPTCL which comprised 21% (24 of 115 cases). The next common subtype was cutaneous CD 30 positive lymphoproliferative disorder (including Lymphomatoid papulosis and Anaplastic large cell lymphoma) comprising 17.4% (20 of 115 cases). The most common sites of presentation in the present study were trunks and extremities (Non photo exposed areas) accounting for 56% (64 of 114 cases) followed by trunks, extremities, head and neck region (Non photo exposed areas and photo exposed areas) 36% (40 of 114 cases) and scalp, head and neck region (Photo exposed areas, 8%). The frequencies of sites of involvement have been shown in Figure - 1. According to Table - 2 regarding clinical presentation and laboratory findings of the study population, macules and patches were seen in 29% (34 of 114 cases), followed by nodules 28% (33 of 114 cases) and plaque 26% (30 of 114 cases). Fever was present in 18.4% (21 of 114 cases) and edema (pedal, periorbital and generalized edema) was present in 8.7% (10 of 114 cases). Hepatomegaly was seen in 17.5% (20 of 114 cases). Splenomegaly was seen in 18.4% (21 of 114 cases). Marrow involvement at presentation was seen in 24% (10 of 41 cases). Anemia was a common manifestation and seen in 33.7% (27 of 80 cases). Leucopenia was seen in 19.2% (15 of 78 cases). Thrombocytopenia was seen in 18.6% (14 of 75 cases). Serum LDH levels were elevated in 18.8% (13 of 69 cases).
Figure - 1: FREQUENCY OF SUBCATEGORY OF VARIOUS CUTANEOUS LYMPHOMAS.
Subcutaneous panniculitis like T-cell lymphoma comprised 21% (24 of 115 cases) of all cutaneous lymphomas. The median age of presentation was 29 years (range 3-56 years) and was predominantly seen in females with a male to female ratio of 1:4.7. Most patients presented with multiple subcutaneous nodules of varying sizes. Lower extremities were the most common site of involvement followed by upper extremities and trunk (17 of 22 cases). Marrow involvement was present in 17% (2 of 12 cases). B symptoms were seen in 75% (15 of 20 cases). Hepatomegaly was seen in 47% (11 of 23 cases) and splenomegaly in 43% (10 of 23 cases). Hepatosplenomegaly was seen in 34% (8 of 23 cases). Anemia was seen in 43% (10 of 23 cases). Leucopenia was seen in 39% (9 of 23 cases). Thrombocytopenia and pancytopenia were seen in 26% (6 of 23 cases) and 13% (3 of 23 cases) respectively. Bone marrow was involved in 15% (3 of 20 cases). Serum LDH level was elevated in 78% (18 of 23 cases) (Normal value 225-460 Units/litre). Serum LDH of more than 1000 units was found in 11 patients, with an average of 2443 units/litre. Angiocentricity and angioinvasion were found in two patients among these 11 patients. Bone marrow was involved in two cases with raised LDH levels. All the cases were CD3+ and CD20- with an average MIB1 labeling index of 40-50%. CD8 was positive in 69% (13 of 19 cases). CD56 was focally positive in 5% (1 of 19 cases). Cytotoxic granule associated protein granzyme B was positive in 88% (7 of 8 cases).
The treatment and follow-up data was available for 19 patients of SPTCL. Majority (89%, 17 of 19 cases) received systemic multiagent chemotherapy (CHOP/CVP/DHAP). The median follow-up period of 20 patients was 19 months (Range 1-80 months) during which 15 patients developed new lesions and/or deteriorated symptomatically (75%), 4 remained stable (20%) and 1 patient improved (symptomatically/clinically) (5%). Out of 11 patients with raised LDH (more than 1000 units/litre), 10 patients received chemotherapy (data of one patient was not available). Almost all patients of SPTCL showed deterioration in the form of either developed new lesions and/or worsening of symptoms.
Discussion
A total of 115 cutaneous lymphomas were diagnosed, out of 11,320 skin biopsies, during 5 years (01/01/2020 to 31/12/2024) in the Department of General Pathology, RKDF, Bhopal. This comprised 2.1% (115 of 5467) of all lymphomas diagnosed during 5 years of study which is slightly higher than documented in western literature (<1%) [5, 6]. The frequency of cutaneous lymphoma in our study was 1.01 per 100 biopsy specimen, this is in contrast to 0.7 and 1.0 in Indian [7] and western literature [8]. Cutaneous lymphomas comprised 9.8% of all extra nodal Non-Hodgkin’s lymphomas which is similar to documented western literature (10%) [6].
Of these all cutaneous lymphomas, 89% were cutaneous T cell lymphoma (CTCL), 6% (7 of 115 cases) were cutaneous B cell lymphoma and 0.9% was Blastic plasmacytoid dendritic cell neoplasm. The frequency of cutaneous T cell lymphoma was almost similar to published one Indian study (94%)[7], however study done by George et al and a documented frequency in Willemze review article were 64% and 77% respectively [8, 9]. The frequency of cutaneous B cell lymphoma was almost similar to previous Indian study (5.67%) [7] and Far East study (4%) [10]. The documented frequency in western literature was higher (up to 35%) [8, 11, 12], which is statistically significant (p=0.0004). This may be due to high prevalence of Borrelia burgdorferi infection in European countries [13], which is associated with cutaneous marginal zone lymphoma. The frequency of Blastic dendritic cell neoplasm is almost similar throughout the world, however very few cases [14] have been documented with occasional case series [15]. Of these, Mycosis fungoides comprised 44 % and is commonest of all cutaneous as well as cutaneous T cell lymphomas. This distribution is similar to Western/Far East literature [8, 10] and few Indian studies (p=0.2641) [9, 16]. However a study done by Doshi, et al. showed higher percentage of Mycosis fungoides (73%) (p=0.0001) [7]. Studies done in Germany and Japan showed lower frequency as low as 30% due to unidentified reasons [17].
Subcutaneous panniculitis like T-cell lymphoma accounted for 21% of all our primary cutaneous lymphomas. This subtype accounts for 0.5-1% in most of the Western countries [8, 17] and 10-15% in other Asian countries [18,10]. This was statistically significant (p=0.0000). A previous Indian study also showed a frequency of 15% [9, 19]. Younger age group was affected in our study with median age of 29 years and was predominantly seen in the female population with a male to female ratio of 1:4.7. The female dominance and the similar age group had already described in literature [5, 8, 16]. Most of our patients presented with subcutaneous nodules with predilection to extremities (lower limb > upper limb) and trunk, similar to what has been documented in the literature by Kong et al and Hoque, et al. [20, 21]. Bone marrow involvement was slightly higher (17%) than mentioned in previous studies [20, 21]. Organomegaly including hepatomegaly and splenomegaly was seen in 48% and 43% of our cases respectively. Thrombocytopenia and pancytopenia were seen in 26% and 13% respectively. Serum LDH level was elevated in 90%. These were almost similar to what has been described in a previous Indian study by Burad, et al. and Lee, et al. Specifically organomegaly and lymphadenopathy were described in few studies with hemophagocytosis [8, 20, 21]. There was no statistical significance between clinical presentation and laboratory parameters with prognosis of this tumor.
86% of the patients presented with Ann Arbor stage I/II disease suggesting localized disease and belonged to low and low intermediate risk category. This was comparable with a previous Indian study by Burad, et al. in which 83% of patients presented with localized cutaneous disease and were stage I/II [16]. All our cases had typical morphology what has been described in literature including rimming of fat spaces, hemophagocytosis and bean bag cells [5, 16, 20, 21]. Four of our patients showed granulomas with multinucleated giant cells which has been highlighted by Willemze, et al. and Kong, et al. [8, 20]. The typical immunophenotype in our cases was CD3+ (100%), CD8+ rimming the fat spaces (69%), Granzyme B (88%) and CD4 (predominantly background lymphocytes) (11%), similar to what has been documented in literature [5, 8, 22]. CD56 was positive focally and weakly positive in one case, which showed deterioration of symptoms during follow up. CD56 positivity is very rare [22, 23]. Average follow up period was 19 months in our study (range 1-80 months). Majority of the patients were treated with multiagent chemotherapy (89%). One patient showed improvement with reduction of lesions and symptomatic improvement. One patient each showed angiocentricity and angiodestruction in their biopsy. These patients showed deteriorated of symptoms as described in literature [8, 20]. EBV-LMP1 was not done in these cases. Both were advised for TCR rearrangement study. Because of economic constraints TCR study was not done. In our study there was no statistically significant difference in prognosis related to organomegaly, hemophagocytosis, pancytopenia or angiodestruction. These parameters were prognostically important as documented in literature [5, 21]. This might be due to limited follow up of our cases. Also the presence of granulomas did not show any significant correlation with prognosis or response to therapy. The significant of which is uncertain.
Subcutaneous panniculitis like T-cell lymphoma accounted for 21% of all our primary cutaneous lymphomas, significantly higher than reported in western literature and almost similar to previous Indian and Far East Asian studies. This was statistically significant (p=0.0000) Bone marrow involvement in subcutaneous panniculitis like T cell lymphoma was slightly higher (17%) than documented in other Indian and Far East Asian studies. There was no prognostically significant difference if there was associated hemophagocytosis syndrome, hepatosplenomegaly or CD56 positivity as has been reported by other studies.
Conclusion
Cutaneous lymphomas though it forms only around 10% of the total extra nodal lymphomas are challenging for their versatile presentation. Classification of these lymphomas has undergone a sea change since the 1980s with new entities being added on with each WHO edition that comes into print. Also knowledge of entities like Indolent CD8 positive T cell lymphoma of the ear and CD8 positive aggressive epidermotropic cytotoxic T cell lymphoma will help in differentiating it from Mycosis fungoides, Pityriaris Lichenoides Chronica, Vitiligo by clinico-pathological correlation only and not by histology or clinical features alone.
Diagnosis of each entity represents a challenge as it is different from its nodal counterpart or does not have a nodal counterpart. Most of the cutaneous T cell lymphomas have good prognosis with respect to their nodal counterparts. FOXP1 and Notch 1 can be used in the future for prognostication of these lymphomas and new markers like Beta F1 and Gamma delta and EBER done on a routine basis will also help in making diagnosis easier.
References
1. Chen C, Yin J, Duan M, Wang W, Zhao D, Wei C, Jia C, Zhang W, Zhou D and Zhang Y (2024) Chemo-free salvage treatment outperforms traditional chemotherapy in advanced lines of relapsed/refractory subcutaneous panniculitis-like T-cell lymphoma. Front. Immunol. 15:1476875. doi: 10.3389/fimmu.2024.1476875
2. Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IBdO, Berti E, et al. The 5th edition of the world health organization classification of haematolymphoid tumours: lymphoid neoplasms. Leukemia. (2022) 36(7):1720–48. doi: 10.1038/s41375-022-01620-2
3. 2. Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. (2016) 127(20):2375–90. doi: 10.1182/blood-2016-01-643569
4. 3. Willemze R, Jaffe ES, Burg G, Cerroni L, Berti E, Swerdlow SH, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. (2005) 105(10):3768–85. doi: 10.1182/blood-2004-09-3502
5. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW, editors. WHO classification of tumors of haematopoietic and lymphoid tissues. Lyon: IARC; 2008.
6. Zucca E, Roggero E, Bertoni F, Cavalli F. Primary extranodal non-Hodgkin’s lymphomas. Part 1: Gastrointestinal, cutaneous and genitourinary lymphomas. Ann Oncol Off J Eur Soc Med Oncol ESMO. 1997 Aug;8(8):727–37.
7. Doshi B, Khopkar U. Retrospective study of spectrum of cutaneous lymphoma presenting to dermatology. Indian J Dermatol Venereol Leprol. 2011;77(4):512.
8. Willemze R, Jaffe ES, Burg G, Cerroni L, Berti E, Swerdlow SH, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005 May 15;105(10):3768–85.
9. George R, Bhuvana S, Nair S, Lakshmanan J. Clinicopathological profile of cutaneous lymphomas--a 10 year retrospective study from south India. Indian J Cancer. 1999 Dec;36(2-4):109–19.
10. Lee M-W, the Korean Dermatopathology Research Group. Characteristics of cutaneous lymphomas in Korea. Clin Exp Dermatol. 2003 Nov 1;28(6):639–46.
11. Fink-Puches R, Zenahlik P, Bäck B, Smolle J, Kerl H, Cerroni L. Primary cutaneous lymphomas: applicability of current classification schemes (European Organization for Research and Treatment of Cancer, World Health Organization) based on clinicopathologic features observed in a large group of patients. Blood. 2002 Feb 1;99(3):800–5.
12. Espinet B, García-Herrera A, Gallardo F, Baró C, Salgado R, Servitje O, et al. FOXP1 molecular cytogenetics and protein expression analyses in primary cutaneous large B cell lymphoma, leg-type. Histol Histopathol. 2011 Feb;26(2):213–21.
13. Cerroni L, Zöchling N, Pütz B, Kerl H. Infection by Borrelia burgdorferi and cutaneous B-cell lymphoma. J Cutan Pathol. 1997 Sep 1;24(8):457–61.
14. Peter CVD, Chakrapani A, Shah S, Shah A, Srivastava A. Blastic Plasmacytoid Dendritic Cell Neoplasm Presenting as Fever with Diffuse Cutaneous Nodules. Indian J Dermatol. 2012;57(1):45–7.
15. Herling M, Teitell MA, Shen RR, Medeiros LJ, Jones D. TCL1 expression in plasmacytoid dendritic cells (DC2s) and the related CD4+ CD56+ blastic tumors of skin. Blood. 2003 Jun 15;101(12):5007–9.
16. Burad DK, Therese MM, Nair S. Peripheral T-cell lymphoma: frequency and distribution in a tertiary referral center in South India. Indian J Pathol Microbiol. 2012 Dec;55(4):429–32.
17. Hallermann C, Niermann C, Fischer R-J, Schulze H-J. Survival data for 299 patients with primary cutaneous lymphomas: a monocentre study. Acta Derm Venereol. 2011 Sep;91(5):521–5.
18. Chang S-E, Jee M-S, Kim KJ, Choi J-H, Sung K-J, Moon K-C, et al. Relative frequency of the different types of cutaneous T cell and natural killer cell lymphomas in Korea based on the proposed WHO classification and the EORTC classification. J Dermatol. 2003 Jan;30(1):42–7.
19. Bakhshi S, Das P, Puri K, Singhal M, Ramam M, Sharma A, et al. Subcutaneous panniculitis-like T-cell lymphoma: A clinicopathologic study of 5 cases. Indian J Pathol Microbiol. 2011;54(2):318.
20. Kong YM*, Dai BM+, Kong J, Zhou X, Lu HM*, Shen LM*, et al. Subcutaneous Panniculitis-like T-cell Lymphoma: A Clinicopathologic, Immunophenotypic, and Molecular Study of 22 Asian Cases According to WHO-EORTC Classification. J Surg Pathol Oct 2008. 2008;32(10):1495–502.
21. Hoque S r., Child F j., Whittaker S j., Ferreira S, Orchard G, Jenner K, et al. Subcutaneous panniculitis-like T-cell lymphoma: a clinicopathological, immunophenotypic and molecular analysis of six patients. Br J Dermatol. 2003 Mar 1;148(3):516–25.
22. Dabbs D. Immunohistology of Non Hodgkin Lymphoma. Diagnostic Immunohistochemistry. 2006: 138-143.
23. Go RS, Wester SM. Immunophenotypic and molecular features, clinical outcomes, treatments, and prognostic factors associated with subcutaneous panniculitis-like T-cell lymphoma. Cancer. 2004 Sep 15;101 (6):1404–13.
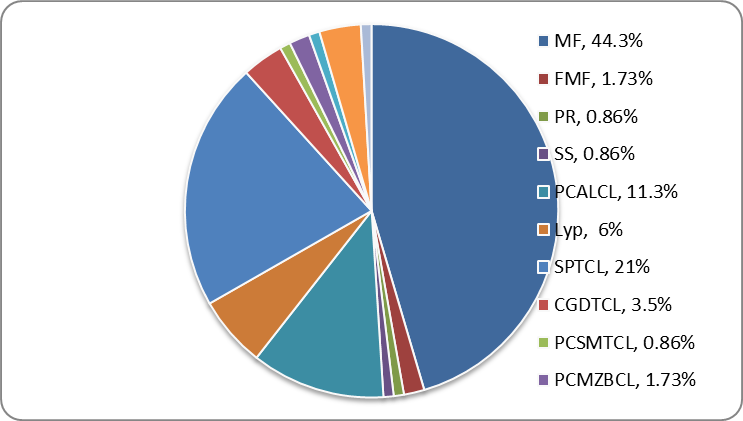
Figure 1
