Triple Negative Breast Cancer Behavior – A 7 Years Single Cancer Center - Hospital Based Study
Triple Negative Breast Cancer Behavior – A 7 Years Single Cancer Center - Hospital Based Study
HebatAllah Mahmoud Bakri Ahmed1*, Samir Shehata Mohamed Eid 2, Amal Rayan Ibrahim 3, Aya Saber khalifa Mohamed4
1,2,3,4. Clinical Oncology Departement, Assiut university hospitals, Egypt.
*Correspondence to: HebatAllah Mahmoud Bakri Ahmed, Clinical Oncology Departement, Assiut university hospitals, Egypt.
Copyright.
© 2025 HebatAllah Mahmoud Bakri Ahmed This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 19 July 2025
Published: 28 July 2025
DOI: https://doi.org/10.5281/zenodo.16737010
Abstract
Back ground and aim : In Egypt, Triple negative breast cancer (TNBC) accounts for 11.05% of breast cancer cases which rises to 25% thankful to the presidential campaign for screening and early detection . This retrospective study aims to conduct an epidemiological analysis of the clinical behavior and outcomes of TNBC patients treated at the Clinical Oncology Department of Asyut University Hospital from 2015 to 2022.
Methods : Data were collected from medical records of 127 Patients >18 years diagnosed with TNBC aiming to identify the patterns, prognostic factors, and therapeutic responses in different stages , in addition to their survival data and correlation with different factors. Primary Endpoints were Incidence and behavior of TNBC including stage at presentation, recurrence patterns, and metastatic sites , treatment received . Disease-Free Survival (DFS) , Overall Survival (OS) , and Progression-Free Survival (PFS) were secondary end points .
Statistical Analysis was conducted using SPSS version 26.0. Kaplan-Meier method was used for estimating survival data and log-rank test was used to compare survival curves. Cox proportional hazards model was constructed to assess the influence of each factor on PFS and OS. Statistical significance was set at p < 0.05.Logistic regression analysis was performed to predict event occurrence (recurrence or progression) using odds ratios (ORs).
Results : The majority being female (95.3%) with mean age was 51.2 ± 13.5 years , with the highest proportion of patients (79.5%) aged >40. Of 95 patients non metastatic cohort 32.3% had early-stage disease, 42.5% were diagnosed with locally advanced disease. Neo adjuvant CTH was administered to 68.4% of non-metastatic patients, with (pCR) rate of 53.8%.
About (32) 25.2% presented with metastatic disease, multiple sites of Mets represented in 59.4%.Of note most common site was lung by (53.3%) and least was bone by (26.7%) . Taxol-containing chemotherapy regimen was the most common protocol used in both settings 82.1% and 65.63% . At the data cut off September 2023 there was 80.0% of non-metastatic patients were alive versus 53.1% of metastatic patients.
For non metastatic cohort , Median DFS time is 52 months but a notable sharp decline is observed at 36 months to (66.3%), indicating importance of meticulous FU is needed in the first 3 years from diagnosis due to higher rate of recurrence especially in those with Locally advanced stage (p=0.038*), larger tumor size (T3-T4, p=0.024*), node-positive status (p=0.029*), lymph vascular invasion positive (p=0.018*), and Taxol-free chemotherapy regimens (p=0.021*). Cox regression analysis revealed that locally advanced stage (HR=1.92, p=0.026), node-positive status (HR=2.45, p=0.004), LVI presence (HR=2.96, p=0.001), and BRCA negatively (HR=1.86, p=0.036) remained independent prognostic factors for DFS.
For metastatic cohort : The median PFS is 32 months with a significant drop occurs at 36 months (40.8%) only alive. In univariate analysis, several factors significantly predicted worse PFS: age ≥40 years (HR=1.68, p=0.034), ECOG =2 (HR=2.34, p=0.006), multiple metastatic sites (HR=1.96, p=0.015) especially non regional lymph nodes metastases (HR=1.82, p=0.031) then liver (HR=2.16, p=0.007). However, when these factors were examined together in multivariate analysis, only poor performance status (HR=1.96, p=0.044) and lung metastases (HR=1.88, p=0.036) remained independently significant ( Table 3 )
Non-metastatic cohort have a significantly longer Overall median survival time 52 months compared to metastatic patients 36 months . Factors associated with dismal OS include age ≥40 years (HR=2.15, p=0.004), locally advanced stage (HR=1.85, p=0.016) and LVI (HR=1.89, p=0.012), positive nodal status (HR=2.24, p=0.002), metastatic disease at presentation (HR=3.42, p<0.001), multiple metastatic sites (HR=2.18, p=0.004), and brain metastasis (HR=2.85, p<0.001) carries shorter OS, which remained independent statistical significance in multivariate analysis.
Conclusion and Recommendation :
TNBC can be a curable disease especially if diagnosed with earlier stages, giving taxanes in their neo/adj setting with meticulous follow up especially in first 3 years remains highly important as increasing rates of recurrence and distant metastasis , in addition to encouraging usage of maintenance options available as PARPi or IO if available . Predicting factors associated with poor response and dismal OS in metastatic setting helps in identifying risks and modifying treatment plan to improve the outcome.
Keywords: Triple negative breast cancer, survival , recurrence , Follow up , early detection.
Triple Negative Breast Cancer Behavior – A 7 Years Single Cancer Center - Hospital Based Study
Introduction
Breast cancer (BC) is the most common malignancy in women, with 2.3 million new cases and 685,000 deaths reported globally in 2020 1. The incidence is significantly higher in developed countries compared to developing nations. It remains a major cause of cancer-related mortality worldwide, ranking as the fifth leading cause of cancer death. In Egypt, breast cancer accounts for 16.4% of all cancer-related deaths 2
TNBC is a distinct and aggressive subtype of breast cancer characterized by the absence of ER (Estrogen Receptor ), PR (Progesterone Receptor ), and human epidermal growth factor receptor 2 (HER2) 3. This subtype is associated with poor prognosis, higher rates of recurrence, and limited targeted therapeutic options. This study aims to conduct an epidemiological analysis of the clinical behavior and outcomes of TNBC in patients treated at the Clinical Oncology Department of Asyut University Hospital from 2015 to 2022. The study seeks to evaluate TNBC incidence, patient demographics, disease characteristics, treatment approaches, and survival outcomes, providing insights into the patterns, prognostic factors, and therapeutic responses of TNBC in this specific population.
Patients and Methods
This study is a retrospective cohort study conducted at the Clinical Oncology Department, Asyut University Hospital. Data were collected from medical records of patients diagnosed with (TNBC) between 2015 and 2022.
Inclusion Criteria
• Patients aged ≥ 18 years.
• Both male and female patients.
• Histo pathologically confirmed early, locally advanced., metastatic TNBC.
Exclusion Criteria
• Patients diagnosed with other types of malignancies.
• Patients with tumors expressing HER2, ER, or PR.
Primary Endpoints
1. Incidence of TNBC at the Clinical Oncology Department, Asyut University Hospital from 2015-2022.
2. Behavior of TNBC, including stage at presentation, recurrence patterns, and metastatic sites.
3. Types of treatment received, including chemotherapy, radiotherapy, immunotherapy, and targeted therapies.
Secondary Endpoints
1. Disease-Free Survival (DFS): Defined as the time from the date of diagnosis to the first documented cancer recurrence, second malignancy, or death from any cause.
2. Overall Survival (OS): Defined as the time from the date of diagnosis until death from any cause.
3. Progression-Free Survival (PFS): Defined as the time from the date of diagnosis until disease progression or death.
Treatment Modalities & Follow-up Data: Recurrence status, metastatic progression, treatment response, survival status at last follow-up.
Statistical Analysis
• The statistical analysis was conducted using SPSS version 26.0.
• Descriptive statistics were presented as mean ± standard deviation (SD) for continuous variables and numbers (percentages) for categorical variables.
• Survival analysis:
- Kaplan-Meier method was used for estimating DFS, OS, and PFS.
- Median survival times were calculated as the time at which 50% of patients had experienced the event (death for OS, progression for PFS).
- Log-rank test was used to compare survival curves.
• Regression Analysis:
- Cox proportional hazards model was constructed to assess the influence of each factor on PFS and OS.
- Hazard ratios (HR), confidence intervals (CI), and p-values were reported to determine statistical significance.
• Chi-square and Fisher's exact test were used for categorical variable comparison (e.g., response rates between different treatment groups).
• Logistic regression analysis was performed to predict event occurrence (recurrence or progression) using odds ratios (ORs).
• Statistical significance was set at p < 0.05.
Results
The majority being female (95.3%) with mean age was 51.2 ± 13.5 years , with the highest proportion of patients (79.5%) aged >40. ( Table 1 )clarifies difference of baseline characteristics between 2 cohorts . of 95 patients non metastatic cohort 32.3% had early-stage disease, 42.5% were diagnosed with locally advanced disease .Neo adjuvant CTH was administered to 68.4% of non-metastatic patients, with (pCR) rate of 53.8%.
About (32) 25.2% presented with metastatic disease multiple sites of Mets represented in 59.4% versus 40.6% for single sites . Of note most common site was lung by (53.3%) and least was bone by (26.7%),with equal percentage of brain and non regional LNs (46.7%). Taxol-containing chemotherapy regimen was the most common protocol used in both settings 82.1% and 65.63% .
At the data cut off September 2023 there was 80.0% of non-metastatic patients were alive versus 53.1% of metastatic patients .
Notably for non metastatic cohort : median DFS time is 52 months (95% CI: 25.253 - 78.747) a high survival probability in the early months , but a notable sharp decline is observed at 36 months to (66.3%), indicating importance of meticulous FU is needed in the first 3 years from diagnosis due to higher rate of recurrence especially in those with Locally advanced stage (p=0.038*), larger tumor size (T3-T4, p=0.024*), node-positive status (p=0.029*), lymph vascular invasion positive (p=0.018*), and Taxol-free chemotherapy regimens (p=0.021*) . Cox regression analysis revealed that locally advanced stage (HR=1.92, p=0.026), node-positive status (HR=2.45, p=0.004), LVI presence (HR=2.96, p=0.001), and BRCA negatively (HR=1.86, p=0.036) remained independent prognostic factors for DFS (Table 2)
For metastatic cohort : The median PFS is 32 months Figure 1 (95% CI: 15.702 - 48.298) with a significant drop occurs at 36 months (40.8%) only alive . with no improvement beyond this point, highlighting importance of incorporation of newer lines as early as possible to improve quality of life and treatment response .
In univariate analysis, several factors significantly predicted worse PFS: age ≥40 years (HR=1.68, p=0.034), ECOG =2 (HR=2.34, p=0.006), multiple metastatic sites (HR=1.96, p=0.015) especially non regional lymph nodes metastases (HR=1.82, p=0.031) then liver (HR=2.16, p=0.007). However, when these factors were examined together in multivariate analysis, only poor performance status (HR=1.96, p=0.044) and lung metastases (HR=1.88, p=0.036) remained independently significant ( Table 3 )
Regarding OS , non-metastatic cohort have a significantly longer Overall median survival time 52 months (95% CI = 25.02–78.98) compared to metastatic patients 36 months ( 95% CI = 24.89–47.25) Figure 2 . Factors associated with dismal OS include age ≥40 years (HR=2.15, p=0.004), locally advanced stage (HR=1.85, p=0.016) and LVI (HR=1.89, p=0.012), positive nodal status (HR=2.24, p=0.002), metastatic disease at presentation (HR=3.42, p<0.001), multiple metastatic sites (HR=2.18, p=0.004), and brain metastasis (HR=2.85, p<0.001) carries shorter OS , which remained independent statistical significance in multivariate analysis ( Table 4 )
Table 1 : Base line characteristics
|
Characteristic
|
Sub category |
Non metastatic cohort (n= 95) |
Metastatic cohort (n=32) |
|
Stage at presentation |
Early Stage |
41 (32.3 % ) |
|
|
Locally Advanced
|
54 (42.5 % ) |
|
|
|
Age |
<40 years |
22 (23.2%) |
4 (12.5%) |
|
≥40 years |
73 (76.8%) |
28 (87.5%) |
|
|
Family History
|
Positive |
17 (17.9%) |
9 (28.1%) |
|
Negative |
78 (82.1%) |
23 (71.9%) |
|
|
Menopausal Status |
Pre-menopausal |
35 (36.8%) |
7 (21.9%) |
|
Post-menopausal |
60 (63.2%) |
25 (78.1%) |
|
|
|
0
|
33 (34.7%) |
5 (15.6%) |
|
1
|
40 (42.1%) |
14 (43.8%) |
|
|
2
|
22 (23.2%) |
13 (40.6%) |
|
|
Type of Surgery
|
BCS |
67 (70.5%) |
- |
|
Mastectomy |
28 (29.5%) |
- |
|
|
BRCA Status |
Positive |
13 (13.7%) |
6 (18.8%) |
|
Negative |
60 (63.2%) |
17 (53.1%) |
|
|
Not Available |
22 (23.1%) |
9 (28.1%) |
Table 2 : univariate and multivariate analysis for DFS
|
Variable |
Univariate Analysis (HR, 95% CI) |
P-value |
Multivariate Analysis (HR, 95% CI) |
P-value |
|
<40 years (Ref) |
Reference |
- |
Reference |
- |
|
≥40 years |
1.82 (1.12 - 2.96) |
0.042* |
1.64 (0.98 - 2.74) |
0.058 |
|
Early Stage (Ref) |
Reference |
- |
Reference |
- |
|
Locally Advanced |
2.53 (1.48 - 4.32) |
0.001* |
1.92 (1.08 - 3.41) |
0.026* |
|
Node Negative (Ref) |
Reference |
- |
Reference |
- |
|
Node Positive |
3.18 (1.82 - 5.56) |
<0.001* |
2.45 (1.34 - 4.48) |
0.004* |
|
T1-T2 (Ref) |
Reference |
- |
Reference |
- |
|
T3-T4 |
2.86 (1.64 - 4.98) |
0.001* |
1.76 (0.94 - 3.28) |
0.076 |
|
LVI Absent (Ref) |
Reference |
- |
Reference |
- |
|
LVI Present |
3.84 (2.12 - 6.96) |
<0.001* |
2.96 (1.56 - 5.62) |
0.001* |
|
PNI Absent (Ref) |
Reference |
- |
Reference |
- |
|
PNI Present |
2.96 (1.42 – 6.18) |
0.004* |
1.68 (0.76 – 3.72) |
0.198 |
|
BRCA Positive (Ref) |
Reference |
- |
Reference |
- |
|
BRCA Negative |
2.14 (1.24 - 3.68) |
0.006* |
1.86 (1.04 - 3.32) |
0.036* |
Table 3; PFS cox regression
|
Variable |
Univariate Analysis (HR, 95% CI) |
P-value |
Multivariate Analysis (HR, 95% CI) |
P-value |
|
<40 years (Ref) |
Reference |
- |
Reference |
- |
|
≥40 years |
1.68 (1.04 - 2.72) |
0.034* |
1.42 (0.84 - 2.40) |
0.186 |
|
ECOG 0-1 (Ref) |
Reference |
- |
Reference |
- |
|
ECOG 2 |
2.34 (1.28 - 4.26) |
0.006* |
1.96 (1.02 - 3.76) |
0.044* |
|
Single metastatic site (Ref) |
Reference |
- |
Reference |
- |
|
Multiple metastatic sites |
1.96 (1.14 - 3.38) |
0.015* |
1.72 (0.96 - 3.08) |
0.068 |
|
Non regional lymph nodes mets |
1.82 (1.06 - 3.12) |
0.031* |
1.64 (0.92 - 2.92) |
0.094 |
|
Liver mets |
2.16 (1.24 - 3.76) |
0.007* |
1.88 (1.04 - 3.40) |
0.036* |
Figure1:PFS Kaplan Meier curve
Figure 2: OS Kaplan Miere curve
Table 4: os cox regression
|
Variable |
Univariate Analysis (HR, 95% CI) |
P-value |
Multivariate Analysis (HR, 95% CI) |
P-value |
|
<40 years (Reference) |
- |
- |
- |
- |
|
- ≥ 40 years |
3.15 (1.52-4.21) |
0.002* |
1.21 (1.12-3.62) |
0.010* |
|
Early stage (Reference) |
- |
- |
- |
- |
|
Locally Advanced stage |
1.85 (1.12-3.06) |
0.016* |
1.76 (1.04-2.97) |
0.035* |
|
Metastatic stage |
3.42 (1.98-5.91) |
<0.001* |
3.15 (1.78-5.57) |
<0.001* |
|
LVI Absent (Reference) |
- |
- |
- |
- |
|
LVI Present |
1.89 (1.15-3.11) |
0.012* |
1.75 (1.04-2.94) |
0.035* |
|
Node Negative (Reference) |
- |
- |
- |
- |
|
Node Positive |
2.24 (1.35-3.72) |
0.002* |
2.06 (1.21-3.51) |
0.008* |
|
Single site of mets (Reference) |
- |
- |
- |
- |
|
Multiple sites |
2.18 (1.28-3.71) |
0.004* |
1.95 (1.12-3.39) |
0.018* |
|
Absent brain mets (Reference) |
- |
- |
- |
- |
|
positive brain mets |
2.85 (1.65-4.92) |
<0.001* |
2.45 (1.38-4.35) |
0.002* |
Discussion
Our study included 127 TNBC patients, with female predominance (95.3%) and a small proportion of male patients (4.7%) as in table (1) consistent with global findings, where TNBC is commonly diagnosed in women. However, a study analyzing TNBC cases from the National Cancer Database reported a small but notable presence of male TNBC patients, accounting for 6% of all TNBC cases 4 which was higher than our data.
The mean age was 51.2 years as in table (1) similar to data derived from Ahmed et al. 5 in a large Chinese cohort and a Tunisian database study reinforcing the trend that TNBC primarily affects middle-aged women in Africa 6 and an Egyptian study by Zakaria et al. 2018 7 and Makar et al 2019 8 documented that 46% TNBC cases occurred in women younger than 50 years, with a higher prevalence of aggressive tumor characteristics. Of note , our patients presented with non metastatic either as locally advanced disease , early stage by 42% , 32 % respectively and 25% only metastatic , thanks to presidential campaign in Egypt which started 2019 for early diagnosis and detection though the contrary seen for example in an Iranian study reported the frequent late-stage presentation 9
In Table 1 : 70.5% underwent breast-conserving surgery (BCS), while 29.5% had a mastectomy between 2015- 2022 , with rising rates in later years after 2018 for BCS due to more surgical convincing about it especially after the comparable published international results to MRM with more satisfied quality of life for female patients .Ours was similar to 2 studies in AFRICA a Tunisian one by Darouich et al. 10 and Egyptian study by Gado et al. 11 who found a 68% BCS rate.
The median DFS time was 52 months and show a high survival probability in the early months ( 97.8% at 12 months and 93.8% at 18 months) ,but , a notable sharp decline is observed at 36 months to (66.3%) , with 5 years survival only to 44% which was similar to a study by Stewart et al. reported a similar DFS drop beyond 36 months 12 . All these pushing the bar for highlighting the higher incidence of late recurrences , the importance of meticulous Follow up especially in the first 3 years and encouraging the introduction of novel agents as maintenance aims for improvement of survival .
A Univariate Cox regression used for identification of factors associated with worse DFS as seen in table(2). It was found 7 factors : age ≥ 40 years (HR=1.82, p=0.042), locally advanced stage (HR=2.53, p=0.001), nodal positivity which was present in 58.9% in our patients carries (HR=3.18, p<0.001) , larger tumor size (T3-T4) (HR=2.86, p=0.001), LVI which was detected in more than two thirds of our patients (HR=3.84, p<0.001), PNI was 4.2% of cases (HR=2.96, p=0.004), and BRCA negative (HR=2.14, p=0.006). While in the multivariate analysis, 4 out of 7 remained independent predictors : node-positive , LVI , locally advanced stage , BRCA negativity .
Thankful to Morante et al. who developed a prognostic model for distant recurrence-free survival (DRFS) in TNBC patients . Align with ours , T3-T4 tumors (HR = 2.92, 95% CI: 1.39 - 6.17), positive lymph node status (HR = 2.78, 95% CI: 1.74 - 4.45), and age ≥ 40 years (HR = 2.66, 95% CI: 1.46 - 4.88) significantly contributed to a higher risk of recurrence 13. Similarly, Koca et al. 2021 14 analyzed identifying tumor size ≥ 2 cm (HR = 7.82, p = 0.001), presence of axillary lymph node metastases (HR = 8.93, p = 0.003), as independent predictors of disease recurrence and Soares et al. further confirmed the negative impact of LVI (HR = 2.8, p = 0.017) and lack of pathological complete response (HR = 0.3, p = 0.011) on DFS. 15
Un like a meta analysis done by Tutt et al who found that BRCA1/2 mutations were associated with worse DFS (HR=2.04, p=0.032). 16 , BRCA mutation in our cohort was associated with improved DFS may be explained by giving carboplatin in TNBC as SOC in our institutional guidelines as the role of platinum in BRCA carriers explained by Basourakos SP et al(2017) The mechanism of platinum agents lies in its covalent binding with DNA which leads to DNA damage. As the DNA damage accumulates and the damage burden exceeds the limit of DNA repair, cancer cell could not maintain normal mitosis and ultimately undergo apoptosis . 17
The use of taxanes-based regimens has been shown to improve survival, 10 , 15 due to better response rates especially in early-stage TNBC. So , it was incorporated in the guidelines and used in 80% of our non metastatic cohort either as neoadjuvant or adjuvant giving 81.4% with stable disease , and 18.7% experience progressive disease with no CR at the end of treatment .
Mortality was higher in the metastatic group (46.9%) compared to non metastatic especially in Patients with multiple metastases , of 25 patients who diagnosed with met.TNBC , multiple sites rather than single site identified in 59.4% and 40.6% respectively .Lung was site of predilection (53.3%) and least was bone by (26.7%),with equal percentage of brain and non regional LNs (46.7 % ). This aligns with findings from Gu et al who reported that multiple metastatic sites contribute to poor response to systemic therapy and higher tumor burden, leading to rapid disease progression. (18)
Regarding PFS in figure (1) , our patients showed median PFS of 32 months (95% CI: 15.7 - 48.3) consistent with a study by Jomaa and Nagy reported a 15 months in TNBC patients ( 19). In our cohort, the PFS rate remained at 100% up to 12 months then a gradual decline was observed at 18 months and 24 months by(86.5%) and (81.7%) respectively , denoting an increasing risk of progression over time reaches to the only half of the patients at 36 months (40.8%), This pattern aligns with findings from a study on TNBC patients, which reported a 3-year progression -free survival rate of 63.2% (20)
Several factors significantly predicted worse PFS by the univariate analysis shown in table (3), age ≥40 years (HR=1.68, p=0.034), ECOG =2 (HR=2.34, p=0.006), multiple metastatic sites (HR=1.96, p=0.015), lymph nodes metastases (HR=1.82, p=0.031), and liver metastases (HR=2.16, p=0.007). However, in multivariate analysis, only poor performance status and liver metastases remained independently significant . This finding is consistent with previous studies that associate poor ECOG >/= 2 and older ages as more vulnerable as they are expected to have more comorbidities , decreased chemotherapy tolerance and overall reduced progressive survival outcomes (21) .
Regarding organ-specific metastases, LN and liver involvement seen significantly influenced PFS. As Liver metastases are known to be a poor prognostic due to aggressive tumor biology and limited response to systemic treatments and LN metastases, despite being initially responsive to chemotherapy, are prone to rapid recurrence, reducing long-term disease control
The overall survival was in non-metastatic individuals exhibited a significantly difference by 20 months 52 months versus 32 months for metastatic setting being statistically significant (p=0.001) . These findings align with a study published in 2024 by Kesireddy M, indicating that metastatic TNBC is associated with poor median overall survival, typically ranging from 8 to 23 months 21
The risk factors for worse survival shown in table(4) were also the age ≥40 years (HR=2.15, p=0.004), locally advanced stage (HR=1.85, p=0.016) presence of either LVI (HR=1.89, p=0.012), or positive nodal status (HR=2.24, p=0.002), and metastatic disease at presentation (HR=3.42, p<0.001), multiple metastatic sites (HR=2.18, p=0.004), and brain metastasis (HR=2.85, p<0.001) . All these factors maintained independent statistical significance in the multivariate analysis, suggesting they each contribute uniquely to survival outcomes. Wen et al. and Costa et al also demonstrated that patients with locally advanced stage , that lymph node positivity (HR = 2.06, p=0.008) and lymphovascular invasion (HR = 1.75, p=0.035) were independent predictors of worse outcomes. (22 ),(23).
Conclusion and Recommendations
Based on the findings of this study, the following recommendations are proposed to enhance the management and outcomes of TNBC patients. :
• BRCA1/2 testing should be routinely offered to TNBC patients, particularly younger patients and those with strong family history, as BRCA-positive TNBC have options for PARPi treatment.
• ICIs, particularly pembrolizumab, should be integrated whenever possible into neoadjuvant regimens for stage II-III TNBC, as they have demonstrated improved event-free survival (EFS).
• In m TNBC, first-line treatment should be individualized based on PD-L1 status, BRCA mutations, and disease burden
• Regular monitoring and early detection of metastases in 1st 3 years through imaging and clinical assessments should be performed to improve survival outcomes.
• Post-treatment surveillance should follow standardized guidelines, with clinical follow-ups every 3–6 months for the first three years, then every 6–12 months for the next two years
Limitations: The deficiency of electronic data system and dependance on paper sheets hinder more proper data collection , Availability of blocks for BRCA assessment to all cases
Future Directions: Molecular subtyping of TNBC is needed to identify biomarkers predicting treatment response and resistance. Liquid biopsy and circulating tumor DNA (ctDNA) analysis should be explored for real-time monitoring of disease recurrence and treatment response.
Ethics approval and consent to participate:
This study was authorized by the Assuit university
Authors' contributions:
Dr. HebatAllah Mahmoud Bakri contributed the trial design , supervised the study and follow up , served as the principal investigator and drafted the manuscript . Dr. Samir Shehata and Amal Rayan provided ongoing supervision and follow-up. Dr Aya Saber conducted the study, collected the data, and drafted the manuscript. All authors reviewed and approved the final manuscript.
Competing interests
No competing financial interests exist.
References
[1] Arnold M, Morgan E, Rumgay H, et al. Current and future burden of breast cancer: Global statistics for 2020 and 2040. The Breast. 2022;66:15-23.
[2] Ibrahim AS, Khaled HM, Mikhail NN, et al. Cancer incidence in Egypt: results of the national population?based cancer registry program. Journal of cancer epidemiology. 2014;2014(1):437971.
[3] Sun X-B, Liu W-W, Wang B, et al. Correlations between serum lipid and Ki-67 levels in different breast cancer molecular subcategories. Oncology Letters. 2022;25(2):53.
[4] McNamara KM, Oguro S, Omata F, et al. The presence and impact of estrogen metabolism on the biology of triple-negative breast cancer. Breast cancer research and treatment. 2017;161:213-27.
[5] Ahmed R, Din HU, Afzal S, et al. Clinicopathological Characteristics Of Triple Negative Breast Cancer: Triple Negative Breast Cancer. Pakistan Armed Forces Medical Journal. 2017;67(5):838-42.
[6] Darouich S, El Amine El Hadj O, Betaieb I, et al. Triple negative breast cancer: A clinico-epidemiological and histopronostic study of 90 cases. La Tunisie medicale. 2017;95(1):37-44.
[7] El-Kinaai N, Zakaria A-S, Nassar HR, et al. Triple negative breast cancer, clinicopathologic study of Egyptian patients, NCI experience. The Medical Journal of Cairo University. 2018;86(September):2747-53.
[8] Makar WS. Clinicopathological characteristics and survival of triple-negative breast cancer patients: a single Institution Study from Egypt. Research in Oncology. 2019;15(1):31-4.
[9] Abdollahi A, Etemadi M. Pathological characteristics of triple-negative breast cancer at main referral teaching hospital, April 2014 to April 2015, Tehran, Iran. International journal of hematology-oncology and stem cell research. 2016;10(4):200.
[10] Darouich S, El Amine El Hadj O, Betaieb I, et al. Triple negative breast cancer: A clinico-epidemiological and histopronostic study of 90 cases. La Tunisie medicale. 2017;95(1):37-44.
[11] Gado N, Ibrahim D, Atef D, et al. Clinical characteristics of triple negative breast cancer in Egyptian women: a hospital-based experience. Cancer Ther Oncol Int J. 2016;4:426.
[12] Stewart RL, Updike KL, Factor RE, et al. A Multigene Assay Determines Risk of Recurrence in Patients with Triple-Negative Breast Cancer. Cancer research. 2019;79(13):3466-78.
[13 ] Morante Z, Ferreyra Y, Pinto JA, et al. A prognostic model for distant recurrence-free survival in triple-negative breast cancer (TNBC) and the outcomes of initiation of adjuvant chemotherapy in the risk of relapse. Journal of Clinical Oncology. 2022.
[14 ] Koca B, Yildirim M, Kuru B. Prognostic factors affecting disease-free survival in triple-negative breast cancer and impact of Ki-67. Indian Journal of Surgery. 2022;84(Suppl 3):708-13
[15] Soares RF, Garcia AR, Monteiro AR, et al. Prognostic factors for early relapse in non-metastatic triple negative breast cancer — real world data. Reports of Practical Oncology and Radiotherapy. 2021;26:563 - 72.
[16] Tutt AN, Garber JE, Kaufman B, et al. Adjuvant olaparib for patients with BRCA1-or BRCA2-mutated breast cancer. New England Journal of Medicine. 2021;384(25):2394-405.
[17] Basourakos SP, Li L, Aparicio AM, Corn PG, Kim J, Thompson TC. Combination Platinum-based and DNA Damage Response-targeting Cancer Therapy: Evolution and Future Directions. Curr Med Chem (2017) 24:1586–606.
[18] Gu Y, Wu G, Zou X, et al. Prognostic Value of Site-Specific Metastases and Surgery in De Novo Stage IV Triple-Negative Breast Cancer: A Population-Based Analysis. Medical Science Monitor : International Medical Journal of Experimental and Clinical Research. 2020;26:e920432-1 - e-20.
[19] Jomaa MK, Nagy AA. Survival outcomes in Egyptian patients with triple-negative breast cancer: Single institute experience. American Society of Clinical Oncology. 2015;5(2):48-56.
[20] Tavassoli FA. Pathology and genetics. Tumours of the breast and female genital organs. Peritoneal tumours. 2003:197-202.
[21] Kesireddy M, Elsayed L, Shostrom VK, et al. Overall Survival and Prognostic Factors in Metastatic Triple-Negative Breast Cancer: A National Cancer Database Analysis. Cancers. 2024;16(10).
[22 ] Wen S, Manuel L, Doolan M, et al. Effect of Clinical and Treatment Factors on Survival Outcomes of Triple Negative Breast Cancer Patients. Breast Cancer : Targets and Therapy. 2020;12:27 - 35.
[23] Costa REARd, Oliveira FTRd, Araújo ALN, et al. Prognostic factors in triple-negative breast cancer: a retrospective cohort. Revista da Associacao Medica Brasileira. 2021;67 7:950-7.
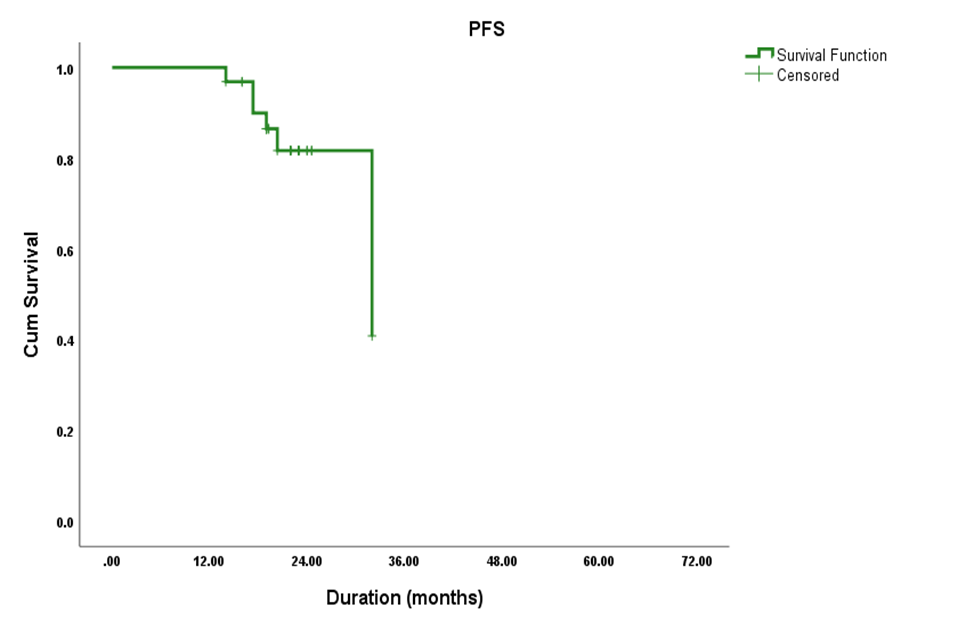
Figure 1
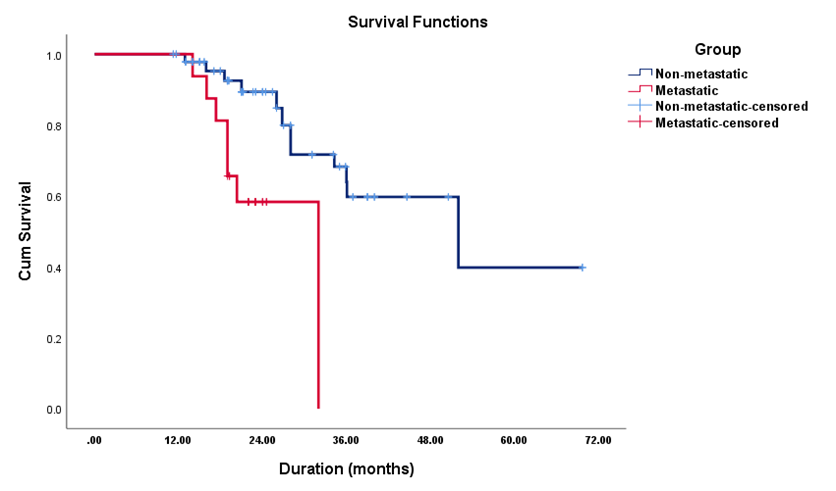
Figure 2

 ECOG status
ECOG status