Neglected Case of Primary Hyperparathyroidism
Neglected Case of Primary Hyperparathyroidism
Samah Abdelhamid Bala Elnour*1, Ali Nagi2, Mohamed Daffalla Awadalla Gismalla3, Dr.Khalid Mohamed Nafie4
1. Consultant General Surgeon, Saudi Arabia.
4. MBBS, MMSCP, MPATH, Histopathologist.
*Correspondence to: Samah Abdelhamid Bala Elnour, Consultant General Surgeon, Saudi Arabia.
Copyright.
© 2025 Samah Abdelhamid Bala Elnour This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 29 July 2025
Published: 08 Aug 2025
Neglected Case of Primary Hyperparathyroidism
Background
The clinical presentation of primary hyperparathyroidism (pHPT) varied noticeably from a severe disease to an asymptomatic condition in Western countries. However, the disease is completely different in Eastern countries. Bone and joint-related signs and symptoms, such as bone pain and multiple fractures, are common in these patients. The principal effect of parathyroid hormone (PTH) is to increase the concentration of plasma calcium by enhancing the release of calcium and phosphate from the bone matrix, increasing calcium reabsorption by the kidneys, and boosting renal production of 1.25 dihydroxy vitamin D-3 (Calcitriol), which elevates plasma calcium levels [1, 2]. Usually, there are four parathyroid glands located posterior to the thyroid gland in the neck. Primary hyperparathyroidism (pHPT) is a disease characterized by hypercalcemia attributable to the autonomous overproduction of PTH. Although some patients with pHPT may have normal serum calcium concentrations, most of them have hypercalcemia. Therefore, pHPT can often be detected by routine serum calcium measurement. pHPT is present in about 1% of the adult population. The incidence of the disease increases to 2% or higher after age 55.
Imaging and nuclear medicine studies will be beneficial, especially in patients who are candidates for surgical removal of the abnormal parathyroid gland. PTH also causes phosphaturia, thereby decreasing serum phosphate levels [1, 3, 4].
In approximately 80–85% of cases with primary hyperparathyroidism, a single adenoma is found [2, 5]. Multiple gland hyperplasia or neoplasia is present in approximately 15% of the remaining cases [4, 7, 8]. pHPT is the commonest cause, affecting approximately 4 per 100,000 populations per annum, and has a peak age of incidence of 50–60. It affects females more than males with a ratio of 3: 1 [2, 4]. Advanced skeletal disease is the most common pattern of presentation at a young age [2]
Clinical Presentation
A 36-year-old male was referred to the general surgery clinic with features suggestive of hyperparathyroidism. His past medical history revealed a known specific complaint without a definitive diagnosis. Furthermore, 1 year ago, he presented with nonspecific generalized joint pain. He gave a history of pulmonary tuberculosis.
His recent condition started two months ago when he presented with a right humeral pathological fracture, which did not union after two months of management. Then, thorough investigations were done to determine the cause of his pathological fractures. His laboratory test is shown in Table 1. His chest x-ray showed osteoporotic features. His extremities and vertebrae showed osteoporotic features. At the T99m MIBI nuclear scan, a focal and persistent active spot at the lower pole of the right thyroid gland, consistent with parathyroid adenoma, was detected (Figure 2).
A classic neck exploration with a horizontal collar incision was performed. A 4.3 *3*2.8 cm yellowish-brown rubbery mass at the inferior parathyroid region on the right side was detected and excised. The serum parathyroid hormone detected intraoperatively dropped from 2650 to 235. A histological study confirmed the diagnosis of parathyroid adenoma. The serum calcium level returned to normal 24 hours postoperatively. The PTH level on the third postoperative day was in the normal range. The patient's complaint of muscle weakness and bone pain disappeared during the first week postoperatively.
|
Table 1: Laboratory data of the patient with their related normal range. |
||
|
Laboratory variables |
Patient’s values |
Normal values |
|
Calcium, mg/dl |
3.15 |
|
|
Phosphor, mg/dl |
|
2.5–5 |
|
Alkaline phosphate, u/l |
2721 |
100–290 |
|
Parathyroid hormone, pg/mL |
2130 |
9 –55 |
Figure 1water silk parathyroid adenoma. Top left: well circumscribed and encapsulated tumor x40. Top right: closely packed nests separated by rich thin fibrovascular capillaries x100. Bottom left: Clear cytoplasm rich in glycogen x200. Bottom right: Occasional enlarged nuclei typical of neuroendocrine atypia x400.
Figure 2
Figure 3
Figure 4
Figure 5
Discussion
There are striking similarities between clinical and laboratory findings of pHPT from Iran and other eastern regions [8, 9]. Testing of intact PTH level is the core of the diagnosis of hyperparathyroidism [3]. Elevated PTH and serum-ionized calcium levels are a diagnostic method for pHPT [4]. A 24-hour urinary calcium measurement is necessary to rule out. Figure 2: The T99m nuclear scan shows a focal and persistent active spot at the lower pole of the right thyroid gland, consistent with parathyroid adenoma. out familial hypocalciuric hypercalcemia [4]. Patients with pHPT usually excrete more than 200 mg calcium per 24- hour, a calcium/creatinine clearance ratio.
Imaging studies are not used to diagnose, confirm, or decide on surgical therapy of hyperparathyroidism [9, 10]. If a limited parathyroid exploration is to be attempted, a localizing study is necessary [9–11]. In patients who have recurrent or persistent hyperparathyroidism after a previous surgery, an imaging study will be necessary [12], in which the T99m nuclear scan is the best initial test [4, 10]. T99m nuclear scan is highly specific for abnormal parathyroid tissue, and its sensitivity is more than 90% in solitary adenoma, but in multiglandular disease, its sensitivity is very low (55%) [10].
Combination of ultrasound and Computed Tomography (CT) has incremental value in accurately localizing solitary parathyroid adenomas over either technique alone [10].
Ultrasonography, CT scans, and Magnetic Resonance Imaging (MRI) have all been used for localization, but they have been largely replaced with 99mTc-sestamibi. In the case of recurrent or persistent disease and in ectopic locations such as the mediastinum, particularly, MRI may be useful [4, 9, 10]. Bilateral internal jugular venous sampling for parathyroid hormone determination may be used in patients with nonlocalized pHPT. Subperiosteal bone resorption and osteitis fibrosa cystica are now less commonly seen in pHPT. Osteitis fibrosa cystica (brown tumour) was seen only in 10–15% in older reports, but new is now seen rarely, because of the increased incidence of milder forms of the disease [7]. The pathognomonic feature of the disease is increased giant multinucleated osteoclasts in scalloped areas of the surface of the bone (How ship’s lacunae) and replacement of the normal cellular and marrow element by fibrous tissue [2]. Suspecting malignancy, the clinician should be highly alert to other possible causes of bony lesions. Brown tumour should be kept in mind during our practice [13]. Multiple maxillofacial brown tumours can be the primary hyperparathyroidism manifestation [14]. In this case decreased bone density and two pathologic fractures in the neck of the right femur and in the radius and ulna of the right hand were obvious on the first visit. We should remember that several types of malignancies present in the lung, head and neck, esophagus, breast, and renal cells, can cause paraneoplastic hypercalcemia and mimic signs and symptoms of parathyroid adenoma [15]. In the last 30 years the most contemporary series show an incidence of 20% or less. In most published series of patients presenting with urolithiasis the incidence of concurrent PHPT is between 2% and 8% [16].
Greater than 50% of patients with hyperparathyroidism have renal symptoms manifested by nephrolithiasis and nephrocalcinosis [17]. Recurrent acute pancreatitis can be the first and sole presentation of undiagnosed pHPT. Muscle weakness, particularly in the proximal extremity muscles, together with progressive fatigue and malaise, may occur in symptomatic pHPT [4]. Various degrees of depression, nervousness, and cognitive dysfunction may commonly occur in pHPT [4]. Hypertension is more prevalent among patients with hyperparathyroidism [18]. Nonspecific renal, neuralgic, gastrointestinal, or bone and muscle system signs and symptoms, can mislead the physician and cause significant delay in diagnosis. As you see in the present case, he had complained from those symptoms for more than 18 months and two previous surgeries without a definite diagnosis during the last six months.
Some clinicians advocate surgical therapy in all patients with primary hyperparathyroidism, but currently acceptable indications for surgery if a serum calcium level is less than 11.5 mg/dl, symptomatic disease, and a 24-hour urinary calcium excretion of more than 400 mg [4–6]. Our patient satisfied all of these criteria. The standard operation is complete neck exploration with identification of all parathyroid glands and removal of all abnormal glands. In the case of 4-gland disease, a 3.5- gland parathyroidectomy must be performed. Approximately 50–70 mg of the most normal appearing glands must be left intact [4, 8, 11]. In this case, three other parathyroid glands examined during surgery were normal; thus, only the adenomatous gland was removed. Parathyroidectomy reduced the risk of fracture in all patients with pHPT, when compared with observation [19, 20]. The benefits of parathyroidectomy were reported in all patients with pHPT, regardless of age, calcium level, or bone mineral density [20]. Although offering parathyroidectomy to all patients with pHPT, regardless of age and other variables, may have good advantages, it should be considered very carefully in older patients [21]. A recent cost analysis study emphasizes the importance of early parathyroidectomy, demonstrating that parathyroidectomy is cost-saving compared to observation and serial monitoring of patients with PHPT.
Conclusion
It is difficult to diagnose (pHPT) in primary and secondary health-providing settings. This can lead to delayed presentation and complications. However, diagnosis and treatment if the disease is suspected are not difficult.
References:
1. Allen, C. E., Merad, M., & McClain, K. L. (2018). Langerhans cell histiocytosis. Nature Reviews Disease Primers, 4, 13. https://doi.org/10.1038/s41572-018-0004-4
2. Badalian-Very, G., et al. (2010). BRAF mutations are present in Langerhans cell histiocytosis in children and adults. Blood, 116(11), 1919–1923. https://doi.org/10.1182/blood-2010-03-273242
3. Berres, M. L., et al. (2014). BRAF V600E expression in histiocytic disorders. Blood, 124(20), 3174–3184. https://doi.org/10.1182/blood-2014-05-577834
4. Rao, D. G., Trivedi, M. V., Havale, R., & Shrutha, S. P. (2016). A rare and unusual case report of Langerhans cell histiocytosis. PMC, 28479703.
5. Abla O, Egeler RM, Weitzman S. Langerhans cell histiocytosis: Current concepts and treatments. Cancer Treatment Reviews. 2010;36(4):354-359.
6. Ladisch S, Gadner H. Treatment of Langerhans cell histiocytosis—evolution and current approaches. British Journal of Cancer. 1994;70(23):S41-S46.
7. Egeler RM, D'Angio GJ. Langerhans cell histiocytosis. Journal of Pediatrics. 1995;127(1):1-11.
8. Howarth DM, Gilchrist GS, Mullan BP, Wiseman GA, Edmonson JH, Schomberg PJ. Langerhans cell histiocytosis: Diagnosis, natural history, management, and outcome. Cancer. 1999;85(10):2278-2290...
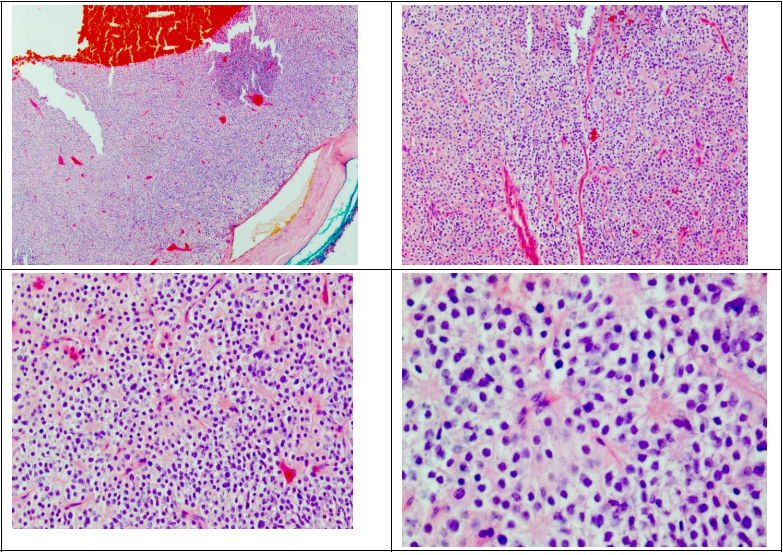
Figure 1
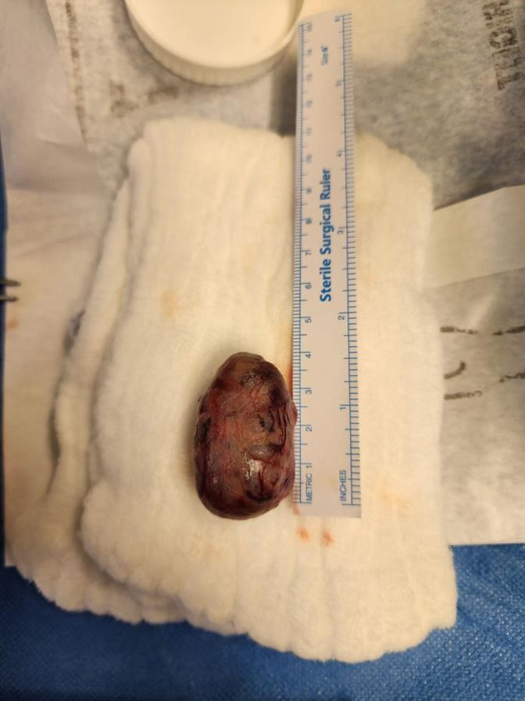
Figure 2
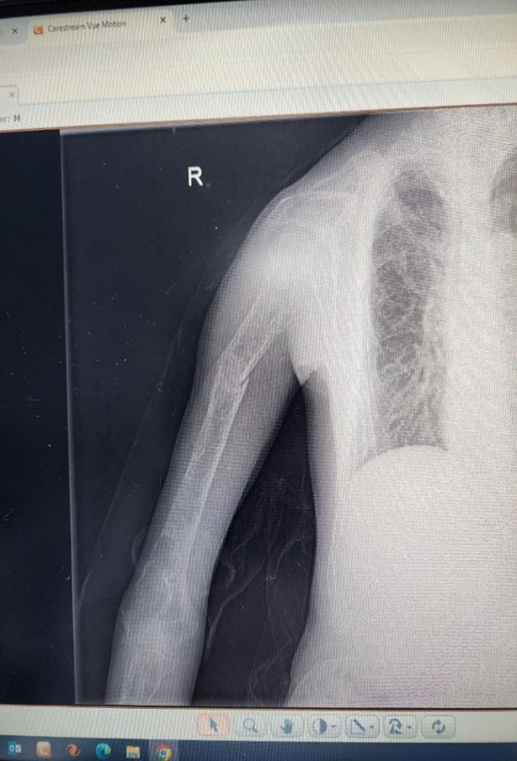
Figure 3
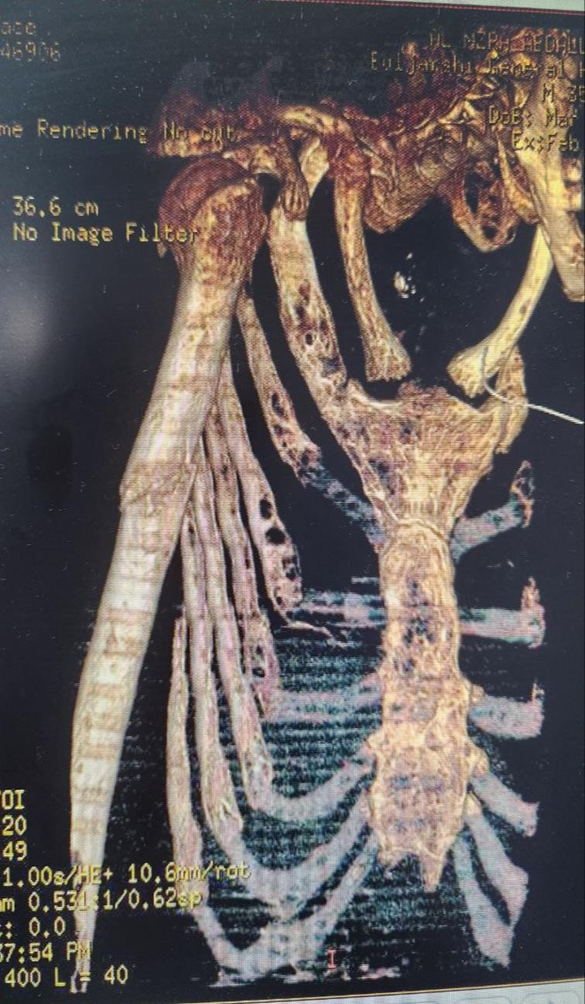
Figure 4
Figure 5
