A Report of Two Unique Perimenopausal AUB Patients with Severe to Morbid Obesity with Hypothyroidism with Menorrhagia with Differential Response to Norethisterone Acetate with Exposition for Same
A Report of Two Unique Perimenopausal AUB Patients with Severe to Morbid Obesity with Hypothyroidism with Menorrhagia with Differential Response to Norethisterone Acetate with Exposition for Same
Dr Kulvinder Kochar Kaur *
*Correspondence to: Dr Kulvinder Kochar Kaur, M.D Obstt & Gynae, specialist reproductive endocrinology & Infertility specialist, Scientific Director Dr Kulvinder Kaur Centre For Human Reproduction 721,G.T.B. Nagar, Jalandhar-144001, Punjab,India.
Orcid Number- https://orcid.org/0000-0003-1473-3419
Copyright.
© 2025 Dr Kulvinder Kochar Kaur This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 18 Aug 2025
Published: 26 Aug 2025
DOI: https://doi.org/10.5281/zenodo.17038567
Abstract
Abnormal uterine bleeding (AUB) by definition portrays any kind of bleeding from the genital tract which is a deviation from the normal in frequency, cyclicity, duration, or quantity. AUB attributes to about 25-33% of outpatient visits to gynaecologists.
Perimenopausal AUB is frequently observed along with possesses the capability of originating from a complicated crosstalk of hormonal fluctuations as well as structural abnormalities for instance uterine leiomyomas or endometrial polyps. Perimenopausal women are further at escalated risk for oestrogen-guided endometrial hyperplasia as well as have a potentiated risk for endometrial cancer.Hypothyroidism is well acknowledged to influence a wide range of physiological systems, inclusive of menstrual function, in women of reproductive age.
Here we report two perimenopausal AUB patients whose presentation was morbid obesity(first BMI->43. 0Kg/m2 , second BMI>-35.0Kg/m2 with hypothyroidism with menorrhagia. Despite presentation of patients were in the form of perimenopausal AUB, both were distinct from each other.1) first Case Report 1 (was older age wise, with greater BMI, greater thyronorm requirement , greater handicaps,her endometrial thickness(ET-10.6) was substantially lesser in contrast to secondpatient, however she promptly responded to primolut-n, while second was younger, required lesser thyronorm dosages despite getting appropriate medical treatment she illustrated failure of response, the expositions might be i)noncompliance tofollow appropriate prescription ii) Negative thought process andsuggest her management .
Key Words; Perimenopausal AUB; morbid obesity; hypothyroidism.
A Report of Two Unique Perimenopausal AUB Patients with Severe to Morbid Obesity with Hypothyroidism with Menorrhagia with Differential Response to Norethisterone Acetate with Exposition for Same
1. Introduction
Abnormal uterine bleeding (AUB) by definition portrays any kind of bleeding from the genital tract which is a deviation from the normal in frequency, cyclicity, duration, or quantity. AUB attributes to about 25-33% of outpatient visits to gynaecologists [1] The initiation of AUB canonically takes place in the fourth to fifth decade (perimenopausal age group), coinciding with the body's shifting to menopause [1]. The clinical manifestation of AUB possesses the capacity of differing amongst patients with heavy menstrual bleeding (HMB), escalation of as well as continuation of menstrual bleeding, irregular bleeding, intermenstrual bleeding, postcoital bleeding, in addition to postmenopausal bleeding (PMB) [1].
The International Federation of Gynecology as well as Obstetrics (FIGO) has introduced a classification system in 2011 to stratify AUB (PALM-COEIN). The labels for instance polyp, adenomyosis, leiomyoma, malignancy (PALM), in addition to hyperplasia detail botherations with the structure of the uterus, while coagulopathy, ovulatory situations, endometrial, iatrogenic, along with not otherwise classified (COEIN) detail issues with the working of the uterus that result in AUB [1-3]. In the perimenopausal scenario, hyper-estrogenism as well as anovulatory cycles are an etiological factor of AUB [4,5].
Mary Nandini Singhetal[6], performed a three -year retrospective study in reference to histopathological assessment in cases of AUB in a tertiary care centre.Out of the 956 endometrial samples received, 106 were observed to be insufficient for diagnosis, diminishing the sample size to 850. Outcomes evaluation, illustrated that perimenopausal women possess the highest incidence of AUB (379/850; 44.5%). The incidence of working in addition to organic etiologies of AUB were 73.9% (628/850) along with 26.1% (222/850), respectively. The maximum frequent working etiologies of AUB were abnormalities in proliferative endometrium (DPE) (235/628; 27.7%), withii) following proliferative endometrium with 155/850 cases (18.2%).iii) The maximum frequent organic disfigurements were benign endometrial polyp (102/850; 12%).iv) Subsequent to that was non-atypical hyperplasia (55/850; 6.5%) followed byv) atypical endometrial hyperplasia/endometrial intraepithelial neoplasia (AEH/EIN) (41 cases, or 4.8%). Vi)Of the 22 cases of endometrial carcinomas, 81.8% were of the endometrioid type, with following 9.2% cases of carcinosarcoma, as well as the rest were clear cell carcinoma (4.5%) in addition to serous carcinoma (4.5%).
Perimenopausal AUB is frequently observed along with possesses the capability of originating from a complicated crosstalk of hormonal fluctuations as well as structural abnormalities for instance uterine leiomyomas or endometrial polyps. Perimenopausal women are further at escalated risk for oestrogen-guided endometrial hyperplasia as well as have a potentiated risk for endometrial cancer [7]. Their study observed that women with low parity possessed greater plausibility to have AUB (66.2%) in contrast to multiparous (30.8%) women who were grand multiparous (1.5%), or those women who had never been pregnant (1.5%). This is converse to other studies in which AUB possessed greater prevalence in multiparous groups [8,9]. Global cultural trends, in addition to, the greater literacy rate, economic status of Kerala, with a greater proportion of small nuclear families,might possessthe capability of partly exposition the contrasting data in association with parity.
Hypothyroidism is well acknowledged to influence a wide range of physiological systems, inclusive of menstrual function, in women of reproductive age [10-12].
Here we report two perimenopausal AUB patients whose presentation was morbid obesity with hypothyroidism with menorrhagia.
2.1 Case Report 1
A 50 yr old patient’s presentation was at the age of 50 yrs with escalated bleeding with passage of clots particularly in evening ,now escalated since 9-7-2025 .
She gave a history of hypothyroidism, was on thyronorm 100μg as well as when omitted it furthergot deranged. Apart from that she was handicapped in lieu of morbid obesity with her knees badly affected for which she possessed the incapacity of walking in lieu of which the orthopaedician initiated methotrexate as well as folinic acid, joincerin(containing glucosamine 800mg, diacerin 50mg and sulphonyl methane25mg),besides 2 more medications(methyl prednisolone4mg as well as etoricoxib 50mg.
She had been married for -27yrs with3 children 2 males-25&24yrs and 1female 22yrs subsequent to which ligation of fallopian tube was performed with her attributing obesity to tubal ligation.
On 10/7/25 General Physical Examination(GPE)revealed -pulse-98/min,BP-177/109mm Hg,wt-99kg,ht-151cm,BMI-43.70Kg/m2
Trans vaginal Ultrasonography (TVS) was tough to perform in lieu of morbid obesity.
TVS revealed - Uterus 46.5x39.6mm with ET-10.6 mm,paradoxically she got awrong report of endometrial thickness(ET-16.2) reported in some other center which actually was the cortical thickness of kidney -16.2 mm due to presenceof pelvic kidney which measured 5.9cmx2.2cm.Both ovaries were normal in size.
Figure1-Pelvic Kidney (on right sidewith uteruson left side)
Investigations at that time revealed -HB-9.3gdec,PCV-36.7,ESR-82,TLC-9830/Cumm,DLC-P63L24E0B3, Platelet count 3.4lakh (1.25-3.5)BT-3’45’’(2-7),CT-6.25(3-10),PT-12.9/12.0,INR-1.01,MCV-76.5(82-100)fLdec. MCH20.8(27.0-34.0)pgdec MCHC27.2(31.6-35.4)g/dl dec RDW-CV17.4(11.5-15.0%, RDW-SD-48.6(35-56)fL, LFT-SB(T)-0.71mg/dl(0.2-3.2), SB(D)- 0.23mg/dl(0.0-0.39), SB(ID)- 0.48mg/dl(0.0-0.8), SGOT-20.44U/L(0-46), SGPT-30.41 U/L(0-46), AP-107.4 mg/dl(30-130), TP-6.98g/dl(6.0-8.3), Alb3.94g/dl(3.4-5), Glob3.94g/dl(1.9-3.9),A:G1.3(1-2), HCV-Negative, HIV-Negative, HBsAg-Negative,VDRL Negative, oral glucose tolerance test ( OGTT)- F-89mg,1’-92mg,2’-92 mg subsequent to 75 g glucose.
Thyroid functiontest illustrated TSH was 17.7uIU/L,repeat 10.4 uIU/L,TT4-12(5.0-13,0),T3 2.0 ng/ml(0.80-12.9) was also with in normallimits. RFT-S Creat-0.52 mg/dl(0.51-0. 95),eGFR -113ml/min/173m2,BUrea-22.10ng/dl(6.0-20),UricAcid-5.09,Scalcium-8.03 mg/dl(8.60-10.30)dec
P43.17 mg/dl(2.4-4.40),Na 139mEq/L(139-146),K3.98 mEq/L(139-146),Cl-107 mEq/L (101-104)
The patient had been prescribed a combination of telmisartan 40mg with 5mgamlodipine however she discontinued it ,so she has been emphasized the significance of its utilization in lieu of pelvic Kidney which was small sized. Additionally, she was prescribed norethisterone acetate(tradename primolut-n tidx3days which was followed by norethisterone bdx21 days. Her bleeding got controlled within aweek, she was recommended persistence of thyronorm 100μg in view of borderline control as well as in lieu of physical handicap,exercise was not feasible,so she was told to start high protein Diet intake 1250-1500 Kcal mg divided in morning(breakfast) in addition afternoon & least calories at evening/dinner along with avoidance of proteins at evening/night as recommended inobesity,eat in morning breakfast, (eat like a king)afternoon(eat like a middleman) at evening/dinner,(eat like a pauper) in addition to morning/ breakfast / between moring and breakfast was prescribed garcinia cambogia capsules(possessing hydroxycitric acid in the form of anti obesity agents) The patient was further advised to get lipids profileinclusive of cholesterol, triglyceride, high-density lipoprotein cholesterol (HDL-C), LDL-C, however she didnot get them donetill now on which she has lost 1kg in 1 month. methyl prednisolone 4mg, methotrexate as well as etoricoxib 50mg have been omitted along with in contrast to joincerin(containing glucosamine 800mg,diacerin50mg and sulphonyl methane 25mg) now replaced with projoint, asdiacerin 50mg might be toxic to Kidney (containing glucosamine 750mg, rose hip extract137.5mg,undenatured type II collagen peptide and methyl sulphonyl methane200mg,vitamin B12-2.2μg).
2.2 Case Report 2
A 48 yr old patient’s presentation was at the age of 48 yrs on 4/8/25 withescalated bleeding with passage of clotsfor the last 4-5 mths which was temporarily controlled by norethisterone but recurred repeatedly -sheconsulted 4-5 gynecologists . She gave a history of hypothyroidism, was on thyronorm 50μg on which hypothyroidism was well controlled.
On 4/8/25 General Physical Examination(GPE)revealed –pulse-82/min, BP-130/87mm Hg, wt-79kg,ht-149cm,BMI-35.755Kg/m2
Trans vaginal Ultrasonography(TVS)
Uterus was enlarged to 76.2x39.6mm with ET-15.7mm with small fibroids measuring 2x2submucusfibroid but the endometrial thickness(ET-15.7mm) was substantially thick which was extending to the cervical canal upto 1.5cm as well as uterus was acutely retroverted with right ovarian size1.8x1.7cm,left 1.7x1.65cm.
Figure 2- Acutely retroverted uterus with thick endometrium ET-15.7mm extending to the cervical canal upto 1.5cm with ovarian size1.8x1.7cm
Investigations at that time revealed – HCV-Negative, HIV-Negative, HbsAg- Negative TSH was 2.2uIU/L,TT3-2.39ng/ml(0.80-1.9), TT4-11.37(5.0-13,0).The patient refused to get too many investigations in lieu of escalated bleeding with passage of clots particularly in evening for the past 4-5mths for which she was given norethisterone athough in case of dosages were adequate or not was not clear from the history of patient by 4-5 variable gynecologists dosages was plausibly not adequate in reference to body weight as well as BMI. Additionally, since we had not earlier seen such thick ET correlated with AUB along withit was extending to the cervical canal upto 1.5cm we tried to rule out any association with pregnancy or retained placental products byperforming a urinary pregnancy test to rule out both (RPOC) or any germ cells tumors which liberate human chorionic Gonadotropins(HCG), but it was Negative.So since norethisterone hadfailed as well as patient was extensively apprehensive she was not convinced with diagnosis thereby she refused to comply with the prescribed agents mifepristone in combination with elagolix in addition to since she was not willing for any surgery or even any repeat D&Cs for ruling out any malignancy( which apparently she had got done by 1 of earlier gynecologists& patient gave a verbal report of normal no knowledge, if proliferative endometrium or atypical endometrial hyperplasia which apparently should have reverted subsequentto norethisterone, in case if received adequate dosages of norethisterone as well as isrunning from pillar to post for knowledge of diagnosis as shewas incapable of understanding despite my extensive explaination in her language please try this medications as she hadfibroid uterus with substantially thick endometrium but all she was bothered about her avoidance of surgery & giveall reports for a newer consultation so outcomes of thiscomplicated patient not known as she refused to follow prescribed agents and justwants let me know if this will totally cure her,which one needs to follow up to gain knowledge.
3. Discussion
Earlier we had reviewed how Thyroid hormones(TH) control body weight, lipid as well as the carbohydrate metabolism in addition to thermogenesis. They regulate lipid metabolism by demonstrating specific actions on the liver along with adipose tissue(AT), summarized in Figure 1[reviewed in ref no 13], in a coordinated way however rarely with debatable effects [14].
Figure 3
Courtesy ref no-13-Possible mechanisms in the association between hypothyroidism and NAFLD. LPL: Lipoprotein Lipase; ROS: Reactive Oxygen Species; TRG: Triglyceride; Chol: Cholesterol.
T3 regulates the expression of genes involved in hepatic lipogenesis as well as the genes involved in the fatty acid oxidation(FFA) through the thyroid hormone receptor-β, that is the main isoform expressed in the liver [15,16]. Thyroid hormone receptor-α is the main modulator of thyroid hormone(TH) actions in the heart in addition to brown adipose tissue(BAT) 13. Thus, TH regulates lipid metabolism in a tissue- dependentmanner, along with this was validated by studies in knockout(KO) mice. THR-α-knockout mice demonstrate reduced liver fat levels as well as white adipose tissue (WAT)mass through diminished genes involved in lipogenesis. They possess lesser insulin resistance(IR) in addition to hepatic steatosis [16]. THR-β-knockout animals reveal an escalated liver mass along with the hepatic lipid accrual through escalation of lipogenic genes as well as reduction in fatty acid β-oxidation but no significant changes in WAT [17].
Hyperthyroidism has been demonstrated, to escalate AT lipolysis [18] as well as hepatic lipogenesis in addition to is correlated with lesser body weight, prominently in view of increased catabolism [19]. Such effects get modulated by a T3- stimulated escalation of the expression of variable lipogenic genes (for instance acyl-CoA-synthetase, fatty acid synthase(FAS), acetyl-CoA carboxylase(ACC) along with glucose-6-P dehydrogenase(G6PD)) as well as genes responsible in FFA (for instance lipoprotein lipase(LPL), fatty acid-binding protein (FABP) as well as fatty acid transporter(FAT) [15]. Hypothyroidism diminished liver uptake of FFA procured from triglycerides [20], in additionto is associated with a decreased lipolysis in the AT along with decreased cholesterol clearance [21], resulting in ,β oxidation of FFA as well as triglyceride clearance is diminished, with a concomitant hepatic accrual of triglycerides along with escalation of low density lipoprotein (LDL) uptake. Hypothyroidism decreases hepatic lipase activity, which modulates FFA in addition to oxidation of long-chain fatty acids(lCFAs) for energy generation.Lipid storage in the liver is further increased by obesity along with lesser resting energy expenditure(EE), both escalated by hypothyroidism [20]. TH treatment in human along with murine models reverts hepatic lipase decreased actions.
In the mitochondria, TH induces carnitine palmitoyltransferase-1a(Cpt1a), the rate-limiting enzyme in FFA, leading to accumulationof FFA.
Obesity, in both human as well as animal studies, is found to lead to lipid accumulation in the liver, resulting in fibrosis in additionto cirrhosis. Escalated hepatic lipid accumulation stimulates downregulation of variable metabolism- correlated genes, which are based on T3 actions [15].Thyroid hormones are activators of lipogenesis via direct in additionto indirect modes .T3 stimulates enzymes that catalyze variable significant steps of hepatic fatty acid generation, for instance ACC (which catalyzes the carboxylation of acetyl-CoA to malonyl-CoA, the first step of hepatic fatty acid synthesis) along with FAS [22].T3 also stimulates various transcription factors whichare implicated in de novo lipogenesis(DNL), for instance carbohydrate responsive element-binding protein (ChREBP), a robust lipogenic controller [21]. Thyroid-stimulating hormone(TSH) participating is believed to stimulate hepatic lipogenesis via binding with the TSH-receptor expressed at the surface of the hepatocytes, that further leads to inductionof the peroxisome proliferator-activated receptor-γ (PPARγ)pathway along with activation of sterol regulatory element-binding transcription factor 1 (SREBP-1c) [24,25]. TSH directly escalates hepatic gluconeogenesis as well as diminishes phosphorylationof 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, the major target of statins, thereby inducing stimulation of hypercholesterolemia [26].Animal studies have suggested a role of T3 in hepatic mitochondrial turnover, which is altered in nonalcoholic fatty liver disease(NAFLD).TH apparently enhances mitochondrial biogeneration along with mitophagy of nuclear receptors [27]. Conversely, hepatic steatosis leads to the repression of T3- based genes involved in metabolism in both humans in addition to animal models [28].
Thyroid hormone signaling further reacts to cross-talk amongst thyroid receptors along with other nuclear receptors sensitive to circulating metabolite quantities, for instance PPARs as well as the liver X receptor (LXR) [29]. Alterations of lipophagy, the modes of autophagy of lipid droplets, which is a significant step of lipid mobilization in the liver, is further believed to take part in NAFLD, as well as T3 has been demonstrated to stimulate lipophagyin vitro along with in vivo [30].Oxidative stress from β-oxidation is thought to be aiding in the propagation of nonalcoholic steatohepatitis (NASH) to hepatocyte inflammation as well as liver fibrosis. Hyperthyroidism has been demonstrated toescalate oxidative stress(OS), leading to liver cell damage [31], whereas hypothyroidism reduces OS levels through decreasing energy expenditure[32].
Thereby, TH might aid in the propagation of NAFLD to NASH, however the precise pathophysiological modes need clarification[rev by us in ref 33]..
4. Conclusions and Directions
Thereby, here we report two perimenopausal AUB patients whose presentation was morbid obesity(first BMI->43. 0Kg/m2, second BMI>-35.0Kg/m2 with hypothyroidism with menorrhagia. Despite presentation of patients were in the form of perimenopausal AUB, both were distinct from each other.1) first Case Report 1 (was older age wise, with greater BMI, greater thyronorm requirement, greater handicaps, her endometrial thickness(ET-10.6) was substantially lesser in contrast to secondpatient, however she promptly responded to primolut-n, while second was younger, required lesser thyronorm dosages despite getting appropriate medical treatment she illustrated failure of response, the expositions might be i)noncompliance tofollow appropriate prescription ii) Negative thought process.
Since we acknowledge cholesterol is the precursor of gonadal sexsteroids both in men as well as women.With perimenopausal age cyclespossess a tendency of menstrual cycle irregularities to start. Appearance of menstrual cycle irregularities takes place particularly in the presence of hypothyroidism. Here in obesity mechanistic modesare analogous to that observed in case of Polycystic other ovary syndrome (PCOS). The manner illustrated in Figre3, in obese PCOS, where escalated androgens generation takes place followed by pregnenolone=->17OH pregnenolone=->, Dehydroepiandrosteronesulphate (DHEAS)=-> =>androstenedione=> testosterone( T), production in the theca cells by Luteinizing hormone(LH), whereasin the granulosa cells androstenedione as well as testosterone( T) transformation to estrogen takes place[rev in ref 34].
Figure 4
Courtesy ref no-34-Classical pathway of androgen synthesis. Luteinising hormone stimulates the classical pathway of androgen synthesis in ovarian theca cells. Cholesterol is transported to the inner mitochrondrial membrane by steroidogenic acute regulatory protein (StAR). A cleavage system of the cytochrome P450 enzyme, CYP11A1, ferrodoxin, and ferrodoxin reductase converts cholesterol to pregnenolone. Expression of CYP11A1 is stimulated by activation of the luteinising hormone receptor. Pregnenolone is transported to smooth endoplasmic reticulum where it is converted to 17-hydroxypregnenolone and subsequently to dehydroepiandrosterone by the 17-hydroxylase and 17,20-lyase subunit of the CYP17A1 enzyme, respectively. Dehydroepiandrosterone is then converted to androstenedione or androstenediol and subsequently to testosterone by a combination of 3β-hydroxysteroid dehydrogenase type II (HSD3B2) and aldo-keto reductase type 1C3 (AKR1C3). 17β-hydroxysteroid dehydrogenase 1 (HSD17B1) also catalyses the conversion of dehydroepiandrosterone to androstenediol. HSD3B2 converts pregnenolone and 17-hydroxypregnenolone to progesterone and 17-hydroxyprogesterone, respectively, which are substrates for a back door alternative pathway of androgen synthesis. Androstenedione and testosterone diffuse into granulosa cells where they are converted to oestrogens by the action of aromatase (CYP19A1), under the control of follicle stimulating hormone receptor activation. Testosterone can be converted to dihydrotestosterone by steroid 5α-reductase (SRD5A) in peripheral tissues
This might be enough toexplain escalated production of estrogens. Neverthelessit, has been well acknowledged for long that Adipose tissue(AT) possesses the capability of converting androgens to estrogens[35].Thereby, obesity alone can explain in perimenopausal women whose follicular cohort is at verge of finishing ,however escalated production of estrogens in AT are capable of resulting in menstrual cycle irregularities, with TH issues aggravating bleeding due to unopposed actions of estrogens . Thereby, administration of 19 nor testosterone derivatives progesterone (Pg) was efficacious, in the first patient. However second patient in view of increased stress along with negative thought process, adrenal gland participates in escalated production of androgens, particularly 11-oxygenated androgen followed by that of estrogens therefore no response to Pg ( see Figure5).
Figure 5
Courtesy ref no-34-Pathway for 11-oxygenated androgen synthesis, which begins in the adrenal cortex. Androstenedione and testosterone are produced by the classical pathway (4). Dehydroepiandrosterone is diverted to downstream androgens or sulphonated to dehydroepiandrosterone sulphate by the sulphotransferase, SULT2A1. Androstenedione and testosterone are hydroxylated by 11β-hydroxylase (CYP11B) to produce abundant 11β-hydroxyandrostenedione (11OHA4) and smaller amounts of 11β-hydroxytestosterone (11OHT). Renal 11β-hydroxysteroid dehydrogenase type 2 (HSD11B2) converts 11OHT to 11-ketotestosterone (11KT) and 11OHA4 to 11-ketoandrostenedione (11KA4). In adipose tissue, 11KA4 is metabolised to 11KT and 11-ketodihydrotestosterone (11DHKT) by aldo-keto reductase type 1C3 (AKR1C3) and steroid-5α-reductase (SRD5A), respectively. 11OHA4 is metabolised to 11OHT and 11β-hydroxydihydrotestosterone (11OHDHT) by 17β-hydroxysteroid dehydrogenase 2 (HSD17B2) and SRD5A, respectively. 11KT and 11KDHT are potent agonists of the androgen receptor whereas 11OHT and 11OHDHT have milder potency. StAR=steroidogenic acute regulatory protein; HSD3B2=3β-hydroxysteroid dehydrogenase type II; CYP11A1, CYP17A1, CYP11B1=cytochrome P450 enzymes.
Recently we reviewed the part of N-Acetylcysteine (Nac), & Selenium in tackling Ferroptosis, Oxidative stress (OS) & PCOS [rev in 36]. Selenium represents a component of antioxidant enzymes for instance glutathione peroxidase(GPx), which helps in scavenging ROS along with diminishing OS. Recognized that selenium’s antioxidant properties might be advantageous in diminishing oxidative injury, Selenium might possess role in improving insulin sensitivity, however not by direct hormonal actions [rev in 36]. Additionally, selenium has been acknowledged to modulate immune reactions, inflammation, in addition to affect hormonal balance. Chronic low-grade inflammation is correlated with PCOS, along with selenium’s anti-inflammatory actions might aid in better management of PCOS symptoms [rev in 36]. Selenium is a critical component of enzymes implicated in the generation as well as transformation of thyroid hormones [rev in 36]. Thyroid peroxidase (TPO) enzyme is imperative for the generation of thyroid hormones. TPO catalyzes the iodination of tyrosine residues in thyroglobulin, a protein generated by the thyroid gland, which is a crucial step in the production of thyroid hormones, the triiodothyronine (T3) along with thyroxine (T4). Without sufficient selenium, TPO action might be jeopardized, resulting in thyroid hormone dysequilibrium [rev in36]. In turn, deiodinase enzymes are implicated in transforming one kind of thyroid hormone (T4) into the greater active kind (T3) in addition to as controlling their accessiblity in variable tissues [rev in36]. Selenium is required for the appropriate working of these deiodinase enzymes [revin 36]. Certain persons with PCOS might further experience thyroid impairment [rev in] 36. Hormonal deorchesteration in addition to IR correlated with PCOS are capable of plausibly affecting thyroid working. Thyroid impairment, particularly situations for instance hypothyroidism, possesses the capability of helping in development of PCOS symptoms, inclusive of irregular menstrual cycles along with hurdles in weight management [rev in 36]. Therefore, there might be part of antioxidants for instance N-acetylcysteine as well as selenium in such patients with hypothyroidism menorrhagia or other menstrual cycle irregularities . Additionally, in second patient I advocate rajyoga meditation for practicing positive thought process[revin 37]. Furthermore, once acknowledge obesity reproductive perimenopausal women try controlling obesity for avoidance of postponed menopause.
References
1. Howkins J, Bourne G: Shaw’s textbook of gynaecology, 17th edition. Kumar S, Padubidri VG, Daftary SN (ed): Elsevier , New Delhi; 2018.
2. Munro MG, Critchley HO, Broder MS, Fraser IS: FIGO classification system (PALM-COEIN) for causes of abnormal uterine bleeding in nongravid women of reproductive age. Int J Gynaecol Obstet. 2011, 113:3-13. 10.1016/j.ijgo.2010.11.011
3. Betha K, Malavatu L, Talasani S: Distribution of causes of abnormal uterine bleeding using new FIGO classification system-PALM COEIN: a rural tertiary hospital-based study. Int J Reprod Contracept Obstet Gynecol. 2017, 6:3523-7. 10.18203/2320-1770.ijrcog20173476 .
4. American College of Obstetricians and Gynecologists: ACOG practice bulletin: management of anovulatory bleeding . Int J Gynaecol Obstet. 2001, 72:263-71. 10.1016/S0020-7292(01)00357-5.
5. Kafle N, Shaukin S, Kafle SU, Singh M, Parajuli SB: Histopathological pattern of endometrial biopsies in patients with abnormal uterine bleeding attending Birat Medical College Teaching Hospital. Birat J Health Sci. 2020, 5:1035-9. 10.3126/bjhs.v5i2.31378.
6. Singh MN, SasidharanA, PaulB ParambathSP., Histopathological Spectrum of Endometrium in Abnormal Uterine Bleeding in a Tertiary Care Centre: A Three-Year Retrospective Study. Cureus2024 Jun 25;16(6):e63158. doi: 10.7759/cureus.63158. eCollection 2024
7. Khurram N, Ghumman NA, Yusuf NW: Morphological pattern of endometrial biopsy in women with clinical diagnosis of abnormal uterine bleeding. Esculapio. 2023, 16:3-7. 10.51273/esc20.251641
8. 15. Golecha N, Porwal V: Abnormal uterine bleeding: a study of its clinical spectrum and incidence of histopathological patterns of endometrium as a function of age with parity. Indian J Obstet Gynecol Res. 2018, 5:339-43. 10.18231/2394-2754.2018.0078
9. 16. Jahan I, Rumana R, Munni N, Akter T, Humayra ZU: Spectrum of clinico-pathological correlation of abnormal uterine bleeding cases in a tertiary care hospital. Surg Res. 2020, 2:1-5.
10. Hannah Himabindu P, Swathi G, Padmavathi K, PenumallaS, Kandimalla R.Hypothyroidism and Its Impact on Menstrual Irregularities in Reproductive-Age Women: A Comprehensive Analysis at a Tertiary Care Center .Cureus2024 16(6):e63158. doi: 10.7759/cureus.63158. eCollection 2024
11. Brown ED, Obeng-Gyasi B, Hall JE, Shekhar S: The thyroid hormone axis and female reproduction. Int J Mol Sci. 2023, 24:9815. 10.3390/ijms24129815
12. Usha SM, Bindu CM, Chandrika N: Thyroid dysfunction: an alternate plausibility in perimenopausal women!. J Midlife Health. 2022, 13:300-3. 10.4103/jmh.jmh_67_22
13. MavromatiM,Jornavyaz FR. Hypothyroidism- associated dyslipidemia: potential molecularmechanisms leading to NAFLD. Int J Mol Sci 2021;22:12797.
14. Sinha RA,You SHZhou J, SiddiqueMM,Bay BH, Zhu Xet al. Thyroid hormone stimulates hepatic lipid catabolism via activation of autophagy. J. Clin. Investig. 2012, 122, 2428–2438.
15. HuangYY,Gusdon AM, Qu S. Cross-talk between the thyroid and liver: A new target for nonalcoholic fatty liver disease
treatment. World J Gastroenterol 2013; 1:, 8238–8246.
16. Flores-Morales A,Gullberg H, Fernandez L, Stahlberg N, LeeNH, VennstromB, Norstedt G. Patterns of liver gene expression governed by TRbeta. Mol Endocrinol 2002; 16: 1257–1268.
17. Jornayvaz FR,LeeHY, Jurczak MJ,Alves TC,Guebre-EgziabheF, Guigni BA,etal.Thyroid hormone receptor-alpha gene knockout mice are protected from diet-induced hepatic insulin resistance.Endocrinology 2012, 153, 583–591.
18. Riis AL,Gravholt CH, Djurhuus CB,NorrelundJorgensenJO ,Weeke J,MollerN. Elevated regional lipolysis inhyperthyroidism. J Clin Endocrinol Metab 2002; 87:4747–4753.
19. CachefoA,BoucherP,Vidon C, DusserreE,Diraison F, BeylotM. Hepatic lipogenesis and cholesterol synthesis in hyperthyroid patients. J Clin Endocrinol Metab 2001; 86; 5353–5357.
20. KlieverikLP,Coomans CP,EndertE, Sauerwein HP,HavekesLM,Voshol PJ,etal.Thyroid hormone effects on whole-body energy homeostasis and tissue-specific fatty acid uptake in vivo. Endocrinology2009; 150 : 5639–5648.
21. NedvidkovaJ,HaluzikM,BartakV,Dostalova I, VlcekP,RacekP,etal.Changes of noradrenergic activity and lipolysis in the subcutaneous abdominal adipose tissue of hypo- and hyperthyroid patients:An in vivo microdialysis study. Ann N Y Acad Sci 2004; 101,:541–549.
22. D’Ambrosio R,Campi I, MaggioniM,PerbelliniR, GiammonaE,Stucchi R,etal.The relationship between liver histology and thyroid function tests in patients with non-alcoholic fatty liverdisease (NAFLD). PLoS ONE 2021; 16:e0249614.
23. Yin L, Zhang Y,Hillgartner FB .Sterol regulatory element-binding protein-1 interacts with the nuclear thyroid hormonereceptor to enhance acetyl-CoA carboxylase-alpha transcription in hepatocytes. J Biol Chem 2002; 277: 19554–19565
24. Dentin, R.; Girard, J.; Postic, C. Carbohydrate responsive element binding protein (ChREBP) and sterol regulatory elementbinding protein-1c (SREBP-1c): Two key regulators of glucose metabolism and lipid synthesis in liver. Biochimie 2005; 87:81–86.
25. Gariani K,JornayvazFR. Pathophysiology of NASH in endocrine diseases. Endocrin Connect 2021; 10: R52–R65.
26. Ritter MJ, AmanoI,HollenbergAN. Thyroid Hormone Signaling and the Liver. Hepatology 2020; 72 : 742–752.
27. LiY, WangL,ZhouL, SongY,MaS,Yu C, Zhao J, Xu, C.; Gao, L. Thyroid stimulating hormone increases hepaticgluconeogenesis via CRTC2. Mol Cell Endocrinol 2017, 446, 70–80.
28. Pihlajamaki , BoesT,Kim EY, DearieF,KimBW,SchroederJ,etal. Thyroidhormone-related regulation of gene expression in human fatty liver. J Clin Endocrinol Metab 2009; 94: 3521–3529.
29. ArakiO, YingH,FuruyaF, Zhu X, ChengSY. Thyroid hormone receptor beta mutants: Dominant negative regulators ofperoxisome proliferator-activated receptor gamma action. Proc NatlAcad Sci USA 2005; 102: 16251–16256.
30. Rautou PE, MansouriA,LebrecD,Durand F,VallaD ,etal. Autophagy in liver diseases. J Hepatol 2010;53:1123–1134.
31. MessarahM,BoumendjelA,ChouabiaA,Klibet F,AbdennourC,BoulakoudMS,etal. Influence of thyroiddysfunction on liver lipid peroxidation and antioxidant status in experimental rats. Exp Toxicol Pathol 2010; 62:301–310.
32. Loria P, CarulliL, BertolottiM, LonardoA. Endocrine and liver interaction: The role of endocrine pathways in NASH. Nat Rev Gastroenterol Hepatol 2009; 6: 236–247.
33. Ishrat Kaur, Kulvinder Kochar Kaur. Study of Correlation of Dyslipidaemia and Diabetes Mellitus with Hypothyroidism in Middle Aged Punjabi Females & Its Impact on Non-Alcoholic Fatty Liver Disease, International Clinical and Medical Case Reports BioRes Scientia Publishers. 3(1):1-10. DOI: 10.59657/2837-5998.brs.24.031
34. DongJ,ReesAD.Polycystic ovary syndrome: pathophysiology and therapeutic opportunities BMJ MedOct 12;2(1):e000548. doi: 10.1136/bmjmed-2023-000548
35. NelsonSL, BulunSE. Estrogen production and action .J Am Acad Dermatol2001 Sep;45(3 Suppl):S116-24. doi: 10.1067/mjd.2001.117432
36. Kulvinder Kochar Kaur,Allahbadia GN,Singh M.“ An Update on PCOS Pathophysiology in Reference to Oxidative Stress (Os), Ferroptosis: A Comprehensive Narrative Review on Part of N-Acetylcysteine (Nac), & Selenium in Tackling Ferroptosis, Os & PCOS. MAR Clinical Case Reports2025; 5:12(1-25). DOI: https://doi.org/10.5281/zenodo.15387844
37. Kulvinder Kochar Kaur,Allahbadia GN,Singh M.“The Science behind Rajyoga Meditation-A Narrative Review .Adv Yoga Physical Ther, 1(2), 05-22.
Figure 1
Figure 2
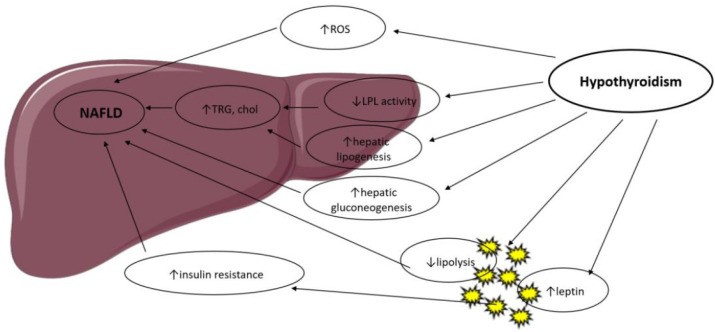
Figure 3
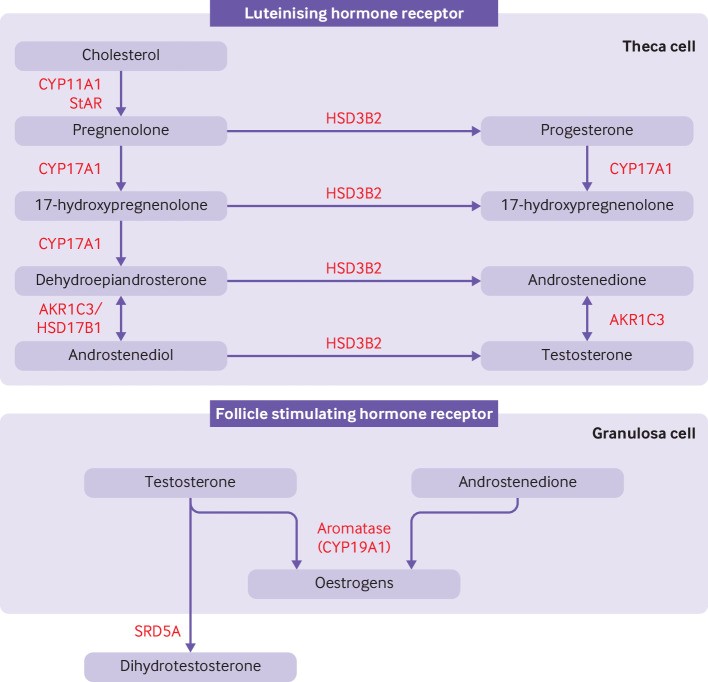
Figure 4
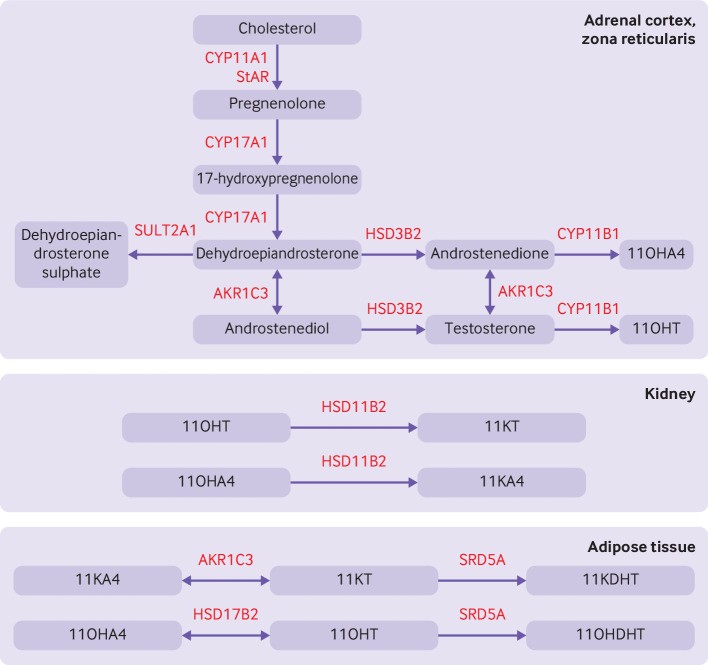
Figure 5
