Computed Tomography for Preoperative Planning in Minimally Invasive Mitral Valve Surgery: A Single Centre Experience
Computed Tomography for Preoperative Planning in Minimally Invasive Mitral Valve Surgery: A Single Centre Experience
Michaela Sasse1, Joon Lee2, Ishtiaq Ahmed3*
*Correspondence to: Ishtiaq Ahmed, England.
Copyright.
© 2025 Ishtiaq AhmedThis is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 14 Aug 2025
Published: 01 Sep 2025
DOI: https://doi.org/10.5281/zenodo.17053226
Abstract:
Objectives: Minimally invasive mitral valve surgery (MIMVS) has been developed as an alternative to conventional median sternotomy for the surgical treatment of mitral valve disease. Computed tomography (CT) and three-dimensional (3D) CT reconstructions play an important role in patient selection and preoperative planning for MIMVS. This study evaluates our centre’s experience of using CT and 3D CT reconstructions for preoperative planning in MIMVS in order to inform future clinical decision making, reduce the potential risk of complications and highlight important considerations when initiating a MIMVS programme.
Methods: CT data from all consecutive patients (n = 79) referred for CT evaluation for MIMVS to our tertiary care centre between January 2016 and February 2021 were retrospectively analysed. All patients underwent a preoperative CT scan of the heart, whole aorta and iliofemoral vessels in order to assess suitability for MIMVS. Two-dimensional (2D) CT data was then used to produce 3D CT reconstructions for further assessment of patient anatomy. Specific cardiac and vascular dimensions were measured and evaluated preoperatively by a cardiac radiologist. Patient suitability for MIMVS and operative strategy were then discussed by a multidisciplinary heart team. Additional measurements were realised retrospectively in order to inform future clinical decision making.
Results: In total, 79 patients were referred for CT evaluation to assess suitability for MIMVS. A total of 55 patients (69.6%) had MIMVS and 16 patients (20.3%) had a conventional sternotomy. A further 8 patients (10.1%) are still awaiting mitral valve surgery. Overall, 11 patients (13.9%) were excluded from MIMVS due to aberrant CT findings alone. CT exclusion criteria included: significant mitral annular calcification; coronary artery calcification; dilatation of the ascending aorta; significant mural thrombus in the descending and abdominal aorta; slender, calcified iliofemoral vessels; dissection flap in the right external iliac artery; incidental persistent left-sided superior vena cava; significant chest wall deformity; incidental liver haemangioma; abdominal aortic aneurysm and aberrant origin of the left circumflex artery. Additional CT findings resulted in modification of minimally invasive operative strategy or required further investigation. For MIMVS, the 30-day mortality rate was 0%, the stroke rate was 1.8%, the repair rate was 100% and there were no conversions to sternotomy.
Conclusions: Preoperative CT and 3D CT reconstructions play a crucial role in appropriate patient selection and individualised procedural planning for MIMVS and may help reduce the potential risk of complications. CT can also highlight extra-cardiac findings that may lead to exclusion from MIMVS or require further investigation. Furthermore, our study demonstrates that MIMVS is a safe and effective procedure. Our study also highlights important considerations when initiating a MIMVS programme.
Computed Tomography for Preoperative Planning in Minimally Invasive Mitral Valve Surgery: A Single Centre Experience
Introduction
Surgical mitral valve repair is the gold standard treatment for patients with severe mitral regurgitation (MR).1 Minimally invasive mitral valve surgery (MIMVS) has been developed as a safe alternative to conventional median sternotomy for the surgical treatment of mitral valve disease.1 The minimally invasive approach is most commonly performed via a right-sided anterolateral minithoracotomy.1 MIMVS is associated with decreased bleeding and pain, less wound infections, reduced ventilation times, shorter hospital stays, faster recovery and improved cosmesis when compared to median sternotomy.2-7 MIMVS also has comparable short-term and long-term mortality and in-hospital morbidity.7 MIMVS has previously been associated with an increased risk of stroke due to longer cardiopulmonary bypass (CPB) and cross-clamp times when compared to sternotomy.7 However, more recent studies suggest that stroke rates are comparable.8,9 Despite the apparent advantages and comparable outcomes of MIMVS, the technique has not yet been widely adopted in the United Kingdom (UK). Conventional sternotomy remains the first-line surgical treatment for severe MR in most general cardiothoracic centres.5 This is possibly owing to steep learning curves for the surgeon, potential complications and lack of conclusive data and randomised controlled trials favouring the technique.3,10
Computed tomography (CT) and three-dimensional (3D) CT reconstructions play an important role in appropriate patient selection and preoperative planning for MIMVS. Precise patient selection and individualised procedural planning may help reduce operative times and the potential risk of complications.3 This study aims to evaluate our centre’s experience of using CT and 3D CT reconstructions for preoperative planning in MIMVS in order to inform future clinical decision making and reduce the potential risk of complications. Additionally, this study aims to highlight important considerations when initiating a MIMVS programme.
Materials and Methods
Population
Patients and/or the public were not involved in the setting of this research due to its retrospective design. CT data from all consecutive patients (n = 79) referred for CT evaluation for MIMVS to a single tertiary care centre in the UK between January 2016 and February 2021 were retrospectively analysed. Initially, only patients with a body mass index (BMI) less than 30 and isolated mitral valve dysfunction with posterior leaflet lesions were considered for MIMVS. However, the inclusion criteria widened with increased experience of the procedure and patients were considered for MIMVS on an individual case-by-case basis.
Preoperative planning
All patients referred for mitral valve surgery at our centre undergo a comprehensive preoperative workup. After initial clinical evaluation, patients undergo transoesophageal echocardiography (TOE) to assess the mechanism and severity of mitral valve dysfunction and any additional valve pathology. If surgery is indicated, the patient is considered for conventional sternotomy or MIMVS. Potential candidates for MIMVS are referred for CT evaluation. Patient suitability for MIMVS and individualised operative strategy are then discussed by a multidisciplinary heart team.
Computed tomography
To assess suitability for MIMVS, all patients included in this study underwent preoperative Electrocardiography (ECG)-gated CT angiography (Somatom Definition Flash scanner, Siemens, Forchheim, Germany) of the heart, whole aorta and iliofemoral vessels. The cardiac scan was acquired in four steps during systolic phase (z-axis coverage: 138 mm). Following ECG-gated cardiac scan acquisition, a separate non-gated CT scan of the whole aorta and iliofemoral vessels was acquired (z-axis coverage: lower half of the neck to the lesser trochanters of the femora). Both scans were acquired following intravenous injection of 100 ml of Iopamidol 300 mg I/ml (Niopam 300, Bracco, High Wycombe, UK) at 4 ml/s followed by 40 ml of saline at 4 ml/s. The ECG-gated images of the heart and aortic root were reconstructed using a narrow field-of-view and a vascular reconstruction filter (Siemens B26F ASA) (slice thickness: 0.75 mm; slice interval: 0.5 mm). The non-gated images of the whole aorta and iliofemoral vessels were reconstructed using a wide field-of-view with a standard reconstruction filter (Siemens B35) (slice thickness: 1.0 mm; slice interval: 1.0 mm).
Image analysis and 3D reconstruction
The image datasets were evaluated by a cardiac radiologist using a dedicated 3D workstation (Syngo.via, Version VB40B_HF01, Siemens, Forchheim, Germany). From the initial datasets, coronal, sagittal and various oblique plane images were reconstructed as well as a 3D rendered model of the thorax. In some cases, additional 3D rendered models of the heart, whole aorta and iliofemoral vessels were reconstructed for further assessment. With the use of CT, the aorta and iliofemoral vessels were assessed for calcification, mural thrombus, aneurysm, dissection flaps, patency and tortuosity. Patients were also screened for coronary artery disease, mitral annular calcification (MAC), aberrant vessel anatomy, chest wall deformity and any other incidental pathologies. Software tools were used to measure the dimensions of the aorta and iliofemoral vessels. CT and 3D CT reconstructions were used to determine the optimal intercostal space (ICS) for right-sided minithoracotomy incision based upon the position of the left atrium, pulmonary vessels and right hemi-diaphragm. 3D CT reconstructions were also used to assess chest wall deformity, aberrant vessel anatomy and aortoiliofemoral vessel tortuosity. Additional measurements were realised retrospectively in order to inform future clinical decision making. Retrospective measurements included: distance from the optimal minithoracotomy incision point to the mitral valve annulus; distance from the optimal minithoracotomy incision point to the ascending aorta and distance from the second ICS incision point to the ascending aorta.
Minimally invasive surgical technique
A right-sided anterolateral minithoracotomy of 5 cm is made in the predetermined ICS (Figure 1). The right lung is deflated and visualisation is achieved using a 5 mm 30° endoscope (KARL STORZ, Slough, UK) placed through a port that is positioned 5 cm posteriorly to the minithoracotomy incision in the same ICS. CPB and retrograde perfusion is established by cannulation of the femoral artery and vein under TOE guidance. Antegrade cardioplegia is delivered via the aortic root. Cross-clamping is achieved by insertion of a transthoracic aortic (Chitwood) clamp (Geister, Tuttlingen, Germany) through an incision in the second ICS on the right side. The mitral valve is approached directly via Sondergaard’s groove and is then repaired or replaced. Repair techniques include: resection; neochordal placement; cleft closure and annuloplasty. TOE is then used to determine repair success, valvular gradient, the presence of systolic anterior motion and any regional wall abnormalities.
Statistics
Descriptive statistics were performed using SPSS version 26 (SPSS, IBM, Chicago, USA). Frequencies are expressed as absolute numbers and percentages. Continuous variables are expressed as mean ± standard deviation (Range).
Figure 1. Right-sided anterolateral minithoracotomy for minimally invasive mitral valve surgery. A 5 cm incision is made in the chosen intercostal space in order to access the mitral valve.
Results
A total of 79 patients were referred for CT evaluation in order to assess suitability for MIMVS between January 2016 and February 2021. Of the 79 patients who were referred for CT evaluation, 71 patients (89.9%) underwent mitral valve surgery. A total of 55 patients (69.6%) had MIMVS and 16 patients (20.3%) had a conventional sternotomy. A further 8 patients (10.1%) are still awaiting mitral valve surgery or under surveillance; 3 of these patients are considered suitable candidates for MIMVS. Baseline characteristics for patients who underwent mitral valve surgery (n = 71) are displayed in Table 1. CT findings for all patients (n = 79) referred for CT evaluation for MIMVS are displayed in Table 2.
In total, 11 patients (13.9%) were excluded from MIMVS due to CT findings alone. CT exclusion criteria included: significant MAC (Figure 2A); significant coronary artery calcification (Figure 2B); dilatation of the ascending aorta to 4.4 cm (Figure 2C); significant mural thrombus in descending and abdominal aorta (Figure 2D); slender, calcified iliofemoral vessels (Figure 2E); dissection flap in the right external iliac artery (Figure 2F); incidental persistent left-sided superior vena cava (SVC) (Figure 2G); significant chest wall deformity (Figure 2H and 3A); incidental liver haemangioma (Figure 2I); 4.6 cm abdominal aortic aneurysm (AAA) (Figure 3B) and aberrant origin of the left circumflex artery (Figure 3C). A further 10 patients (12.7%) were excluded from MIMVS following multidisciplinary team (MDT) discussion due to: complexity of repair; non-isolated procedure and evidence of previous inferolateral myocardial infarction (MI). Additional CT findings resulting in modification of minimally invasive operative strategy included a partially bifid 4th rib (Figure 4A) and rib crowding (Figure 4B). Significant incidental findings that resulted in further investigation and referral to other pathways included a 10 mm lung nodule (Figure 5A) and 4 cm renal mass (Figure 5B).
|
Table 1: Baseline characteristics for patients who underwent mitral valve surgery |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
MIMVS (n = 55) |
Sternotomy (n = 16) |
Total (n = 71) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Age (years) |
66 ± 9.7 (41-81) |
67.5 ± 12.3 (34-84) |
66.3 ± 10.3 (34-84) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Male |
43 (78.2%) |
12 (0.75%) |
55 (77.5%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
BMI (kg/m2) |
25.1 ± 3.7 (20.1-34) |
25.5 ± 4.0 (18.6-32.9) |
25.2 ± 3.7 (18.6-34) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Logistic EuroSCORE |
5.7 ± 6.9 (1.5-37) |
5.7 ± 4.8 (1.5-30) |
5.68 ± 6.5 (1.5-37) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Elective surgery |
52 (94.5%) |
16 (100%) |
68 (95.8%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
LVEF >50% |
41 (74.5%) |
10 (63%) |
51 (71.8%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Diabetes mellitus |
2 (3.6%) |
1 (6.3%) |
3 (4.2%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Preoperative atrial fibrillation |
5 (9.1%) |
5 (31.3%) |
10 (14%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Severe renal impairment |
3 (5.5%) |
1 (6.3%) |
4 (5.6%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Extra-cardiac arteriopathy |
1 (1.8%) |
1 (6.3%) |
2 (2.8%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Previous stroke or TIA |
1 (1.8%) |
1 (6.3%) |
2 (2.8%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Previous MI |
1 (1.8%) |
1 (6.3%) |
2 (2.8%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Previous cardiac surgery |
3 (5.5%) |
1 (6.3%) |
4 (5.6%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Hypertension |
21 (38.2%) |
8 (50%) |
29 (40.8%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Chronic pulmonary disease |
4 (7.3%) |
2 (12.5%) |
6 (8.4%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Pulmonary hypertension |
10 (18.2%) |
2 (12.5%) |
12 (16.9%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Isolated MR |
51 (92.7%) |
13 (81.3%) |
64 (90.1%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Combined MR/TR |
3 (5.5%) |
1 (6.3%) |
4 (5.5%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Combined MR/AR |
1 (1.8%) |
2 (12.5%) |
3 (4.2%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Referred for MV repair |
52 (94.5%) |
16 (100%) |
68 (95.8%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Referred for redo MV replacement |
3 (5.5%) |
0 (0%) |
3 (4.2%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Non-isolated procedure |
8 (14.5%) |
8 (50%) |
16 (22.5%) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 2. Contraindications for minimally invasive mitral valve surgery (MIMVS). Preoperative CT revealing: A) Significant mitral annular calcification; B) Significant coronary artery calcification; C) Dilatation of the ascending aorta to 4.4 cm; D) Significant mural thrombus in the abdominal aorta; E) Slender, calcified iliofemoral vessels; F) Dissection flap in right external iliac artery; G) Incidental persistent left-sided superior vena cava; H) Significant pectus excavatum and I) Incidental liver haemangioma.
Figure 3. Contraindications for minimally invasive mitral valve surgery (MIMVS). 3D CT reconstructions revealing: A) Significant pectus excavatum; B) 4.6 cm abdominal aortic aneurysm and C) Aberrant origin of the left circumflex artery: [1] RCA: Right coronary artery and [2] LCx: Left circumflex artery.
Figure 4. Significant findings resulting in modification of minimally invasive operative strategy. 3D CT reconstructions revealing: A) Rib crowding of ribs 4 to 7 and B) Partially bifid 4th rib.
Figure 5. Significant incidental findings resulting in further investigation and referral. Preoperative CT revealing: A) 10 mm nodule in left lung and B) 4 cm mass in left kidney.
Postoperative complications and 30-day mortality following MIMVS are displayed in Table 3. The 30-day mortality rate was 0%. One postoperative stroke was reported (stroke rate: 1.8%). This was due to an occlusive fat embolus and associated with prolonged CPB time. There were no cases of MI or aortic dissection. Three patients (5.5%) required reintervention for postoperative bleeding or tamponade. The repair rate was 100% and there were no conversions to sternotomy.
Discussion
This descriptive retrospective study presents our centre’s experience of using CT and 3D CT reconstructions for preoperative planning in MIMVS. The aim of this study was to evaluate the use of CT and 3D CT reconstructions for preoperative planning in MIMVS in order to inform future clinical decision making and reduce the potential risk of complications. The study also aimed to highlight important considerations when initiating a MIMVS programme. Overall, 11 patients (13.9%) were excluded from MIMVS due to aberrant CT findings alone. Additional CT findings resulted in modification of minimally invasive operative strategy or further investigation and referral. Our findings demonstrate that preoperative CT and 3D CT reconstructions play a crucial role in informing suitable patient selection and individualised procedural planning for MIMVS and may help reduce the risk of potential complications.
Patients with significant MAC are not considered optimal candidates for MIMVS.11 In our centre, the cardiac radiologist subjectively assesses MAC using CT. Moderate or severe MAC is considered to be significant. One patient was excluded from MIMVS due to significant MAC (Figure 2A). Identification, localisation and quantification of MAC is a relative strength of CT.11 The location and extent of MAC correlates with complexity of repair.11 Therefore, the choice of operative strategy will be dependent upon the surgeon’s experience. For surgeons at the start of the learning curve, MAC should be considered a contraindication for MIMVS. In addition to CT, preoperative TOE is a valuable imaging modality for the assessment of other valvular pathology and complexity of mitral valve dysfunction. A sternotomy is indicated for patients with complex valve disease and patients who require concomitant aortic valve surgery.
Significant coronary artery disease is a contraindication for MIMVS as patients will require concomitant coronary artery bypass grafting (CABG) via conventional sternotomy. Two patients were excluded from MIMVS due to significant coronary artery calcification (Figure 2B) that was initially picked up on CT and required referral for invasive coronary angiography. Both of these patients required concomitant CABG via sternotomy. The left circumflex artery (CxA) lies within close proximity to the posterior mitral valve annulus, this makes it susceptible to injury during MV surgery.12 Possible mechanisms of injury to the CxA include: direct laceration, occlusion with a suture, distortion due to tissue retraction, spasm and external compression by the annuloplasty ring.11 Injury to the CxA during MV surgery is a rare complication, recent studies have reported an incidence of 0.3% to 0.5%.12,13 However, injury to the CxA can lead to MI and is associated with a considerable risk of mortality.12 Therefore, it is important to measure the distance between the mitral annulus and the left CxA using preoperative CT in order to reduce the potential risk of injury occurring. In our study, the closest distance between the left CxA and MV annulus was 2 mm (Table 2) and there were no reported injuries to the CxA during MIMVS. Technical aspects such as careful placement of annular sutures and avoiding large quadrangular leaflet resections may help to prevent injury and should be considered when planning MIMVS.13 Additionally, one patient was excluded from MIMVS due to aberrant origin of the left CxA (Figure 3C). Aberrant origin of the left CxA is associated with an increased risk of mechanical coronary occlusion and patients may require a concomitant myocardial revascularisation procedure.14
CPB with retrograde perfusion is established by cannulation of the femoral artery and vein and occlusion of the ascending aorta with an endo-aortic balloon or cross-clamp.11 In our centre, we use transthoracic external aortic cross-clamping (TTC) to occlude the ascending aorta. CPB with retrograde perfusion and cross-clamping are associated with an increased risk of aortic dissection, retrograde embolization and stroke.15 CT allows for visualisation and assessment of the great vessels of the heart. The ascending aorta is screened for significant atherosclerotic burden in order to assess suitability for CPB and TTC. The identification and quantification of atherosclerotic changes in vessel walls is a relative strength of CT.11 It is also important to measure the diameter of the ascending aorta as dilatation increases the risk of aortic dissection and rupture.16 Dilatation of more than 4 cm is a contraindication for MIMVS.2 One patient was excluded from MIMVS due to dilatation of the ascending aorta to 4.4 cm (Figure 2C). There have been no reported cases of aortic dissection during MIMVS in our centre. Aortic elongation has previously been reported as a potential risk factor for Type A aortic dissection and is considered to be a relative contraindication for MIMVS.17 Currently, we do not screen for aortic elongation as part of our preoperative CT workup for MIMVS. However, it is recommended that aortic elongation is evaluated during preoperative planning in order to prevent potentially avoidable complications. CT also revealed an incidental persistent left-sided SVC (Figure 2G). The patient was excluded from MIMVS due to the potential risk of impaired venous drainage which may have led to excess blood in the operative field.
CT is also used to assess suitability of the aortoiliofemoral vasculature for cannulation and CPB in order to reduce the potential risk of dissection, embolization and stroke.15 In the absence of significant vascular disease, femoral arterial cannulation is the method of choice in our centre. Significant aortoiliofemoral thrombus, calcification, aneurysmal disease, tortuosity and evidence of dissection are all contraindications for femoral arterial cannulation.18 Slender iliofemoral vessels with a luminal diameter of less than 6 mm are also unsuitable for cannulation.2 Slender and/or calcified iliofemoral vessels (Figure 2E) resulted in the exclusion of two patients from MIMVS. One patient was excluded from MIMVS as they were found to have a dissection flap in the right external iliac artery (Figure 2F) and a tortuous left femoral artery. Another patient was excluded due to the presence of a 4.6 cm AAA (Figure 3B) and significant mural thrombus in the descending and abdominal aorta (Figure 2D). One other patient was found to have a 3.1 cm AAA but was deemed suitable for MIMVS as the aorta was only mildly dilated and there was a low risk of rupture. A sternotomy with antegrade perfusion is considered a safer approach for patients with significant aortic or iliofemoral disease.7 A recent meta-analysis of data from 57 studies (13,731 patients) demonstrated that preoperative CT for MIMVS is associated with a reduced risk of perioperative stroke (1.5%) and a trend towards lower operative mortality (0.8%).19 In our present study, the 30-day mortality and stroke rates for MIMVS were 0.0% and 1.8% respectively. Comprehensive preoperative CT evaluation for MIMVS may help reduce the potential risk of mortality and stroke as it allows for individualisation of the surgical approach and exclusion of patients with significant vascular disease.
Three-dimensional CT reconstructions of the thorax allow for assessment of chest wall anatomy and visualisation of the operating field in order to aid preoperative planning, reduce operative times and prevent potentially avoidable complications. Preoperative understanding of patient anatomy is particularly important in minimally invasive surgery as exposure is limited.2 In our experience, we have found that CT and 3D CT reconstructions are useful tools for accurately determining the optimal ICS for right-sided minithoracotomy incision based upon the position of the left atrium, pulmonary vessels and right hemi-diaphragm. The fourth ICS is the optimal incision point in 65.8% of patients (Table 2). We also use CT and 3D CT reconstructions to assess chest wall deformity. Significant chest wall deformity may be a contraindication for MIMVS depending upon the surgeon’s experience. One patient was considered unsuitable for MIMVS due to significant pectus excavatum (Figures 2H and 3A) that may have caused difficulty with surgical approach. Other patients required modification of minimally invasive operative strategy due to rib crowding (Figure 4A) and a partially bifid 4th rib (Figure 4B). CT and 3D CT reconstructions are also used to visualise aberrant vessel anatomy (Figure 3C) and subjectively assess iliofemoral tortuosity in order to aid preoperative procedural planning and prevent potentially avoidable complications.2 Additionally, preoperative CT may highlight incidental extra-cardiac findings that may exclude the patient from MIMVS (Figure 2I) or require further investigation and referral to additional pathways (Figures 5A and 5B).
We measured specific distances using CT in order to inform choice of surgical instruments. Long-shafted needle holders and cross-clamps (Figure 6) are available in a range of sizes. We retrospectively measured the distance from the optimal incision point for minithoracotomy to the mitral annulus in order to determine the optimal needle holder length. The average distance was 18 cm but ranged between 15.2 cm to 22.1 cm (Table 2). We determined that from the catalogue of sizes available, a long-shafted needle holder with a working length of 17 cm would be the appropriate choice in most cases. In addition to this, a long-shafted needle holder with a working length of 25 cm is required for patients who require deeper access. It is important to choose the correct size for the patient as if the needle holder is too long, the surgeon may encounter difficulties with manoeuvrability within the restricted surgical field.20 However, if it is too short then it will be difficult to access the mitral annulus.
Additionally, we retrospectively measured the distance from the second intercostal incision point to the ascending aorta in order to determine the optimal cross-clamp size. The average distance was 10.8 cm and ranged between 7.5 cm to 14.2 cm (Table 2). Furthermore, we estimated the width of the aorta once clamped (0.5 x circumference) in order to inform the choice of jaw size for the clamp. The average estimated width of the aorta once clamped is 5.1 cm but ranges between 3.6 cm to 6.7 cm (Table 2). From the range of cross-clamp sizes available, we have determined that a cross-clamp with a working length of 22 cm and jaw size of 5 cm is suitable for most patients. However, for larger aortas, a cross-clamp with a working length of 24.5 cm and jaw size of 7.5 cm is the most appropriate choice. Again, if the clamp is too long then the surgeon may encounter difficulties with manoeuvrability. If the jaw of the cross-clamp is too long, there is a potential risk of injury to the left atrial appendage.21 If the jaw is too short, then it may lead to incomplete occlusion of the ascending aorta. Preoperatively determining the appropriate choice of surgical instruments may theoretically help to reduce operative times and the potential risk of complications. However, these measurements were realised retrospectively. Further prospective research is needed to determine if these specific measurements will translate to a significant clinical benefit. Furthermore, our study findings may be informative for those who are considering initiating a MIMVS programme. One of the reported barriers to the adoption of minimally invasive techniques is the higher costs associated with the equipment and instruments required.22 The instrument sizes we have highlighted in this study should be suitable for most cases and may prevent the purchase of unnecessary or incorrect surgical instruments when initiating a MIMVS programme.
Figure 6. Surgical instruments. Top: Long-shafted needle holder. Bottom: Transthoracic aortic cross-clamp.
Other institutions have demonstrated that 3D software can be used to produce virtual preoperative simulations of the working port, insertion of surgical instruments, femoral arterial cannulation and mitral valve repair techniques.3,20,23 Additionally, one institution has developed a patient-specific simulation model for mitral valve repair with the use of 3D TOE, 3D CT and 3D printing (rapid prototyping) in order to facilitate surgical training and preoperative planning of MIMVS.24 Finally, 3D virtual reality (VR) is a novel imaging technique that allows immersive 3D assessment of CT scans in a VR environment with promising results in other fields of surgery.25 A recent preliminary study on immersive 3D VR imaging in planning minimally invasive and complex cardiac surgery demonstrated that 3D VR is a potentially beneficial supplementary tool for preoperative planning in cardiothoracic surgery.25 Further research is needed to determine the benefits of simulations and 3D VR for preoperative planning in MIMVS.
Limitations
Our study only presents a single tertiary care centre’s experience of using CT for preoperative planning in MIMVS for a relatively small study population (n = 79). Although most of the preoperative planning was realised prospectively, the study was conducted retrospectively and some additional measurements were realised retrospectively. Furthermore, we do not compare outcomes of using CT versus not using CT in order to establish superiority of CT for preoperative planning in MIMVS. Further prospective research is needed in this area.
Conclusion
Our study demonstrates that preoperative CT and 3D CT reconstructions play a crucial role in informing appropriate patient selection and individualised procedural planning for MIMVS. CT can also highlight extra-cardiac findings that may lead to exclusion from MIMVS or require further investigation. Furthermore, our study demonstrates that MIMVS is a safe and effective procedure with a low rate of postoperative complications, low 30-day mortality rate and 100% repair rate. Precise preoperative procedural planning and exclusion of patients who are unsuitable for MIMVS may potentially reduce operative times and the risk of complications. The study also highlights important considerations when initiating a MIMVS programme. Further prospective research is needed in order to establish superiority of preoperative planning with the use of CT.
References
1. Van Praet K, Stamm C, Sündermann S, Meyer A, Unbehaun A, Montagner M et al. Minimally Invasive Surgical Mitral Valve Repair: State of the Art Review. Interventional Cardiology Review. 2018;13(1):14-19.
2. Heuts S, Maessen J, Sardari Nia P. Preoperative planning of left-sided valve surgery with 3D computed tomography reconstruction models: sternotomy or a minimally invasive approach?†. Interactive CardioVascular and Thoracic Surgery. 2016;22(5):587-593.
3. Heuts S, Olsthoorn J, Maessen J, Sardari Nia P. Planning minimally invasive mitral valve surgery. Journal of Visualized Surgery. 2018;4:212-212.
4. Modi P, Hassan A, Chitwood W. Minimally invasive mitral valve surgery: a systematic review and meta-analysis. European Journal of Cardio-Thoracic Surgery. 2008;34(5):943-952.
5. Vohra H, Salmasi M, Chien L, Baghai M, Deshpande R, Akowuah E et al. BISMICS consensus statement: implementing a safe minimally invasive mitral programme in the UK healthcare setting. Open Heart. 2020;7(2):e001259.
6. Grossi E, Galloway A, Ribakove G, Zakow P, Derivaux C, Baumann F et al. Impact of minimally invasive valvular heart surgery: a case-control study. The Annals of Thoracic Surgery. 2001;71(3):807-810.
7. Falk V, Cheng D, Martin J, Diegeler A, Folliguet T, Nifong L et al. Minimally Invasive versus Open Mitral Valve Surgery a Consensus Statement of the International Society of Minimally Invasive Coronary Surgery (ISMICS) 2010. Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery. 2011;6(2):66-76.
8. Al Otaibi A, Gupta S, Belley-cote E, Alsagheir A, Spence J, Parry D et al. Mini-thoracotomy vs. conventional sternotomy mitral valve surgery: a systematic review and meta-analysis. J Cardiovasc Surg (Torino). 2016;58(3):489-496.
9. Sündermann S, Sromicki J, Rodriguez Cetina Biefer H, Seifert B, Holubec T, Falk V et al. Mitral valve surgery: Right lateral minithoracotomy or sternotomy? A systematic review and meta-analysis. The Journal of Thoracic and Cardiovascular Surgery. 2014;148(5):1989-1995.e4.
10. Holzhey D, Seeburger J, Misfeld M, Borger M, Mohr F. Learning minimally invasive mitral valve surgery: a cumulative sum sequential probability analysis of 3895 operations from a single high-volume center. Circulation. 2013;128(5):483-491.
11. Moodley S, Schoenhagen P, Gillinov A, Mihaljevic T, Flamm S, Griffin B et al. Preoperative multidetector computed tomography angiography for planning of minimally invasive robotic mitral valve surgery: Impact on decision making. The Journal of Thoracic and Cardiovascular Surgery. 2013;146(2):262-268.e1.
12. Husain A, Alsanei A, Tahir M, Dahdouh Z, AlHalees Z, AlMasood A. Left circumflex artery injury postmitral valve surgery, single center experience. Journal of the Saudi Heart Association. 2019;31(2):94-99.
13. Coutinho G, Leite F, Antunes M. Circumflex artery injury during mitral valve repair: Not well known, perhaps not so infrequent—lessons learned from a 6-case experience. The Journal of Thoracic and Cardiovascular Surgery. 2017;154(5):1613-1620.
14. Fabozzo A, Rossi E, Facci G, Bottio T, Gerosa G. Mitral Valve Repair and Anomalous Origin of Circumflex Artery: To Ring or Not to Ring. JACC: Case Reports. 2019;1(4):503-507.
15. Lamelas J, Aberle C, Macias A, Alnajar A. Cannulation Strategies for Minimally Invasive Cardiac Surgery. Innovations: Technology and Techniques in Cardiothoracic and Vascular Surgery. 2020;15(3):261-269.
16. Cozijnsen L, Braam R, Waalewijn R, Schepens M, Loeys B, van Oosterhout M et al. What Is New in Dilatation of the Ascending Aorta?. Circulation. 2011;123(8):924-928.
17. Hermans S, Heuts S, Olsthoorn J, Nia P. Antegrade type A aortic dissection under endoscopic vision during minimally invasive mitral valve repair: a case report. Journal of Visualized Surgery. 2018;4:211-211.
18. Van Praet K, Kofler M, Montagner M, Heck R, Eggert-Doktor D, Stamm C et al. Minimally invasive mitral valve repair using external clamping— pearls and pitfalls. Journal of Visualized Surgery. 2020;6:45-45.
19. Leonard J, Henry M, Rahouma M, Khan F, Wingo M, Hameed I et al. Systematic preoperative CT scan is associated with reduced risk of stroke in minimally invasive mitral valve surgery: A meta-analysis. International Journal of Cardiology. 2019;278:300-306.
20. Di Perna D, Castro M, Gasc Y, Haigron P, Verhoye J, Anselmi A. Patient-specific access planning in minimally invasive mitral valve surgery. Medical Hypotheses. 2020;136:109475.
21. Nissen A, Abreu J, Ornekian V, Nguyen S, Nguyen T. Percutaneous and open cannulation for peripheral cardiopulmonary bypass: pearls and pitfalls. Journal of Visualized Surgery. 2018;4:256-256.
22. Ali J, Abu-Omar Y. Minimally invasive cardiac surgery—a Fad or the Future?. Journal of Thoracic Disease. 2021;13(3):1882-1885.
23. Rim Y, Choi A, McPherson D, Kim H. Personalized Computational Modeling of Mitral Valve Prolapse: Virtual Leaflet Resection. PLOS ONE. 2015;10(6):e0130906.
24. Sardari Nia P, Heuts S, Daemen J, Luyten P, Vainer J, Hoorntje J et al. Preoperative planning with three-dimensional reconstruction of patient's anatomy, rapid prototyping and simulation for endoscopic mitral valve repair. Interactive CardioVascular and Thoracic Surgery. 2016;:ivw308.
25. Sadeghi A, Bakhuis W, Van Schaagen F, Oei F, Bekkers J, Maat A et al. Immersive 3D virtual reality imaging in planning minimally invasive and complex adult cardiac surgery. European Heart Journal - Digital Health. 2020;1(1):62-70..
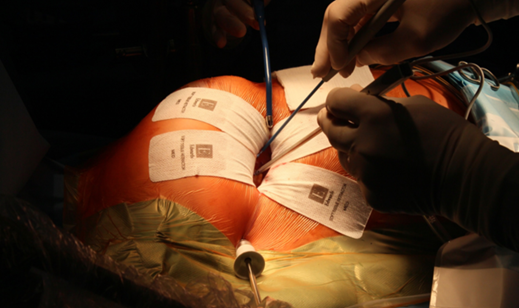
Figure 1
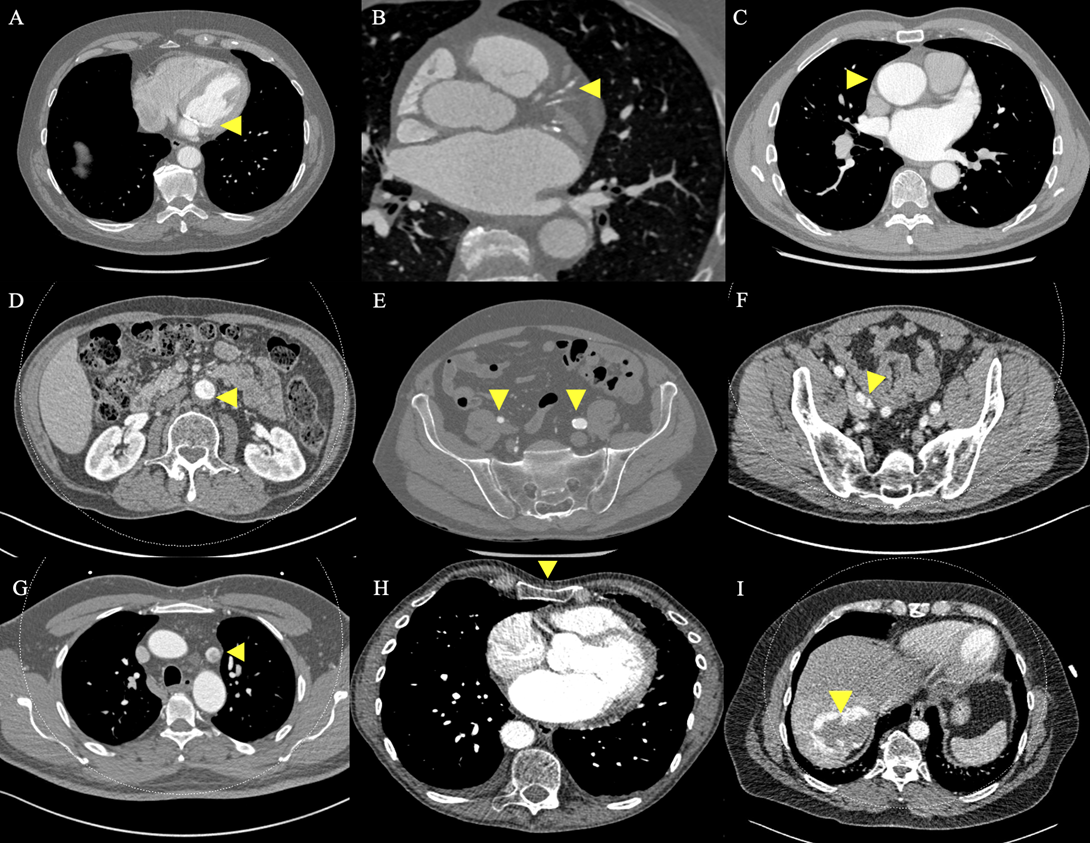
Figure 2
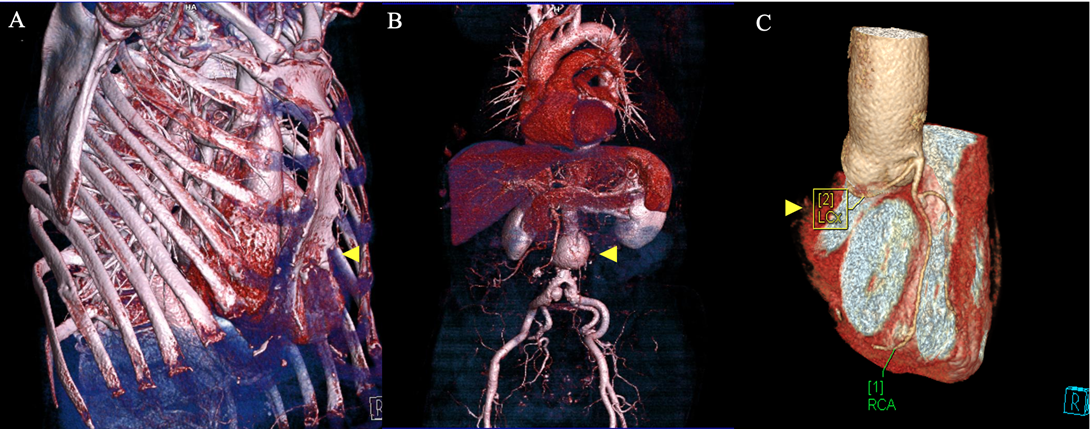
Figure 3
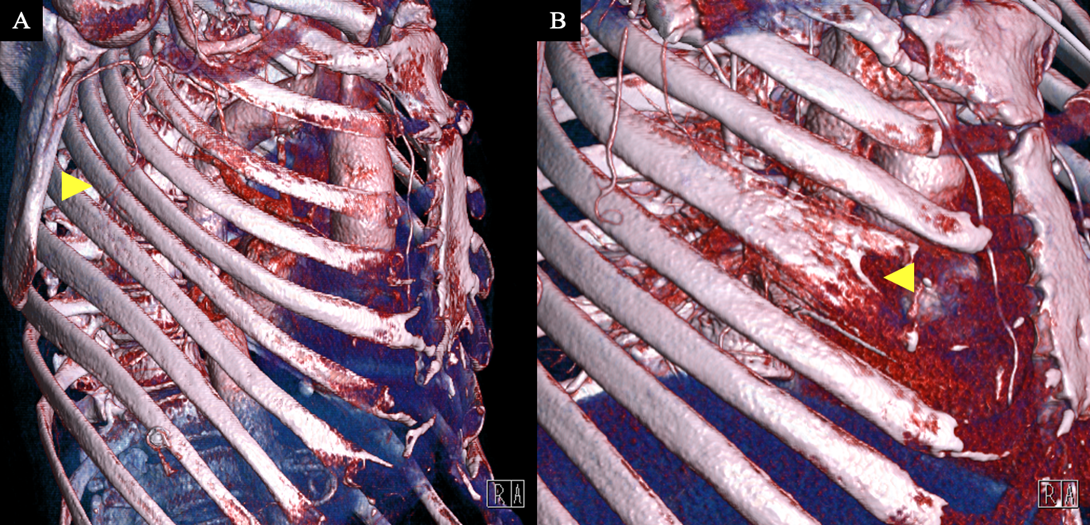
Figure 4
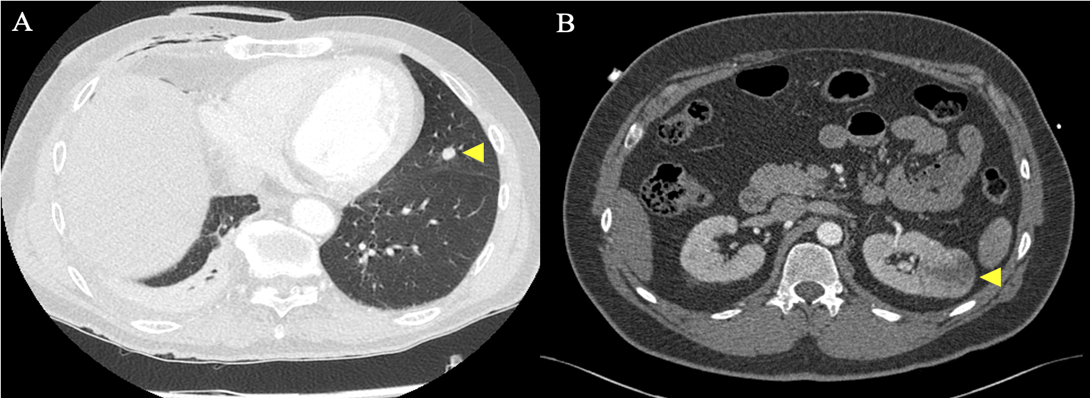
Figure 5

Figure 6
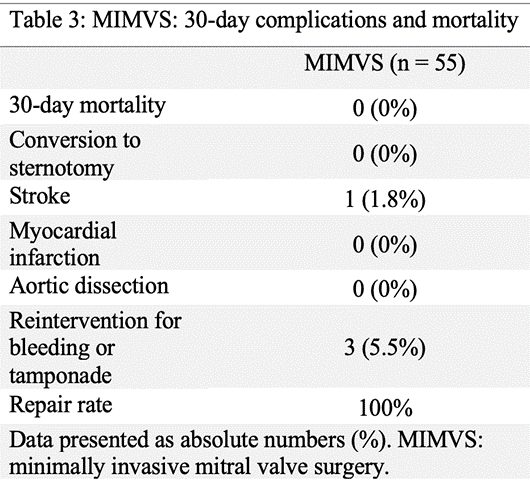
Figure 7
