Auricular versus Cervical Vagus Nerve Stimulation: A Narrative Perspective on Safety, Mechanisms, and Clinical Applicability
Auricular versus Cervical Vagus Nerve Stimulation: A Narrative Perspective on Safety, Mechanisms, and Clinical Applicability
Weronika Górska, MSc (Eng)1; Dr Mikolaj Raszek, PhD2
*Correspondence to:
1. Weronika Górska, MSc (Eng) in Biotechnology. ORCID: 0009-0002-1938-4369.
2. Miko?aj Raszek, PhD, Merogenomics Inc., Edmonton, Canada; University of Alberta, Edmonton, Canada. ORCID:0009-0003-5755-3415.
Copyright
© 2025 Weronika Górska, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 21 Aug 2025
Published: 01 Sep 2025
DOI: https://doi.org/10.5281/zenodo.17036073
Abstract
The field of vagus nerve stimulation (VNS) is rapidly evolving, with expanding evidence for therapeutic benefits across a wide spectrum of conditions and rising demand for safer, non-invasive alternatives. While cervical VNS has established clinical credibility, its surgical invasiveness and risk of off-target effects limit broader adoption. In contrast, transcutaneous auricular vagus nerve stimulation (taVNS), which targets the auricular branch of the vagus nerve, provides a non-invasive, anatomically precise, and well-tolerated approach.
This review synthesises current knowledge on taVNS mechanisms, safety, clinical feasibility, and future directions, with emphasis on device-specific outcomes. Evidence consistently shows that taVNS is not associated with serious adverse events, even in high-risk populations such as patients with heart failure, atrial fibrillation, and post-viral dysautonomia. Meta-analytic safety data and mechanistic studies support its fibre selectivity and broad applicability, highlighting key distinctions from both implanted and transcutaneous cervical VNS.
As healthcare moves towards decentralised and patient-led care models, taVNS emerges as a promising cornerstone of bioelectronic medicine. Its favourable safety profile, at-home usability, and expanding clinical evidence base position auricular neuromodulation not merely as a safer alternative, but as a scalable and patient-centred innovation shaping the next generation of personalised therapeutics.
Auricular versus Cervical Vagus Nerve Stimulation: A Narrative Perspective on Safety, Mechanisms, and Clinical Applicability
1. Introduction
Vagus nerve stimulation (VNS) is a neuromodulation technique that delivers electrical impulses to the vagus nerve to modulate its activity. VNS has transitioned from a niche intervention to a foundational therapeutic strategy, increasingly applied across several medical disciplines due to its diverse benefits. Originally only deliverable through surgically implanted devices, it’s been US Food and Drug Administration (US FDA) approved for treating refractory epilepsy [1], treatment-resistant depression [2], and, more recently, ischaemic stroke rehabilitation [3]. A VNS system had also been approved for weight management in patients with obesity and related comorbidities [4], though the device is no longer commercialised by the manufacturing company. The VNS field is now undergoing a major shift towards non-invasive alternatives, driven by technological advances and patient demand. Among these new non-invasive techniques, transcutaneous auricular vagus nerve stimulation (taVNS, targeting the auricular branch of the vagus nerve (ABVN), is emerging as a leading modality actively explored not only for neuropsychiatric but also other systemic conditions [5].
While the clinical value of cervical VNS, particularly in its surgically implanted form, has been well established in treatment-refractory conditions, this review does not aim to re-evaluate its effectiveness. Instead, we focus on emerging challenges related to scalability, anatomical precision, and safety, especially in decentralised and outpatient care settings. Auricular VNS is presented as a complementary, and in many cases more suitable, non-invasive strategy for broader clinical use, particularly among patients who are not candidates for surgery but still present with dysautonomia-related symptoms. Throughout this review, we integrate comparisons with both implanted and transcutaneous cervical VNS to clarify mechanistic, safety, and translational distinctions, while also exploring the anatomical limitations that may restrict the wider adoption of cervical neuromodulation in milder cases.
2. Scientific and Anatomical Rationale for Auricular Neuromodulation
2.1 From Cervical to Auricular Neuromodulation: Anatomical and Safety Considerations
Surgical cervical VNS has played a foundational role in establishing clinical validation and securing regulatory approvals for VNS. However, it carries inherent risks, primarily due to the off-target activation of efferent fibres that project to diverse anatomical targets. These include cardiac structures, as well as the recurrent laryngeal nerve, a branch of the vagus that innervates the laryngeal muscles responsible for vocal cord movement [6, 7]. While the transcutaneous version of cervical VNS (tcVNS) is generally recognised as safe, reports have also identified potential serious adverse events (SAEs). Risks associated with this technique include cardiovascular and autonomic disturbances [8–11].
A key mechanistic concern with cervical stimulation is that it activates both afferent (sensory) and efferent (motor) fibres within the vagus nerve. This mixed activation can interfere with autonomic regulation, producing unpredictable physiological effects, such as bradycardia, blood pressure fluctuations, or paradoxical autonomic responses [11]. Additionally, unilateral activation of cardiac efferent fibres may disturb the symmetry of bilateral autonomic output to the heart and thereby increase susceptibility to arrhythmias or other adverse cardiac events in both tcVNS and implanted VNS [12]. In severe cases, this can progress to advanced atrioventricular (AV) block or critical bradyarrhythmia culminating in cardiac arrest, as observed in patients who experienced recurrent loss of consciousness with VNS-induced bradycardia, AV block, and transient cardiac arrest during video EEG monitoring [12–14]. Rarely, implanted cervical VNS has also been associated with fatal outcomes, including sudden cardiac death [15] and trauma-induced death [16]. Finally, the close anatomical proximity of the cervical vagus to other critical nerves, such as the phrenic and recurrent laryngeal nerves, creates a risk of off-target effects. Documented complications include respiratory compromise from phrenic nerve involvement, hoarse voice, cough reflex, and vocal cord paresis or paralysis from recurrent laryngeal dysfunction [11, 17].
Collectively, these anatomical and mechanistic side effects, along with the potential risks, narrow the safety profile and tolerability of tcVNS. As a result, it is usually administered in short bursts. While shorter administration times may be more convenient for patients, they can also limit nerve training and, consequently, reduce the therapeutic potential of this intervention [11]. By contrast, auricular stimulation, through its selective action on afferent fibres, avoids unintentional activation of vagal efferent or of non-vagal nerves, allowing for higher dosing with lower associated risks and side effects. Anatomical considerations provide a compelling rationale to focus on taVNS as a safer and more selective technique [12, 18, 19].
2.2. Clinical Safety Profile and Translational Advantages
Both implanted and transcutaneous cervical VNS modalities have significantly contributed to the evolution of neuromodulation therapies overall. However, their broader clinical applicability is constrained by procedural invasiveness, anatomical complexity, and frequent activation of vagal motor efferents, which can lead to undesirable autonomic effects. These safety limitations are clearly reflected in clinical outcomes. A recent meta-analysis by Kalagara et al. (2025) reviewed complications in over 1,400 patients treated with implanted VNS and identified common adverse effects such as voice alteration or hoarseness (45.5%), paresthesia (15.8%), cough (15.0%), dyspnea (14.3%), and pain (11.5%), underscoring the high rate of side effects linked to cervical nerve and efferent fibre activation [20].
tcVNS additionally suffers from diffuse targeting and poor reproducibility, and far fewer studies exist compared to taVNS [21]. A systematic review of 1,322 participants across tcVNS and taVNS confirmed that the majority of reported adverse events, including cardiovascular and autonomic events, originated from cervical stimulation. Overall, adverse events are more frequently associated with tcVNS than with taVNS. Auricular stimulation appears to be better tolerated [22, 23], highlighting why cervical and auricular approaches should not be conflated within a single category.
taVNS has demonstrated a consistently favourable profile across clinical trials and meta-analyses. Tan et al. (2023) meta-analysis of >900 participants reported no significant difference in adverse events risk between taVNS and controls, with only two severe incidents which were deemed to have no causal link to the device [19, 24]. Kim et al. (2022) meta-analysis of 177 studies involving over 6,300 participants identified 20 SAEs across nine trials. Of these, only five were deemed “possibly or probably” related to taVNS. Reported events included anxiety requiring psychiatric admission, vestibular neuronitis, and a suspected basal cell carcinoma without histopathological confirmation. However, using the Bradford Hill criteria no causal relationship was established by the reviewing authors. Importantly, this analysis also confirmed that taVNS does not significantly increase the rate of adverse events compared to control interventions [25]. The most commonly reported symptoms were mild and localised, including ear pain, headache, tingling, and skin irritation, typically attributed to electrode placement rather than stimulation itself [25, 26]. Similarly, a 2024 meta-analysis of 109 studies involving over 3,000 adults across 21 clinical populations found no causal link between taVNS and any SAEs. Among the 57 studies reporting adverse events, nearly half (27) reported none, and none documented SAEs across diverse clinical settings [27]. Further support comes from a 2024 report summarising data from taVNS trials in high-risk populations, including heart failure, paroxysmal atrial fibrillation, hypertension, and POTS, all treated with the Parasym device (Parasym Ltd., London, UK), which delivers proprietary auricular vagal neuromodulation therapy (AVNT) [28]. Despite frailty and multimorbidity in these cohorts, no serious device-related events were reported, emphasising the importance of device-specific safety evaluations in neuromodulation research. Taken together all these findings, no serious cardiac events (e.g., bradycardia or asystole) have been observed in taVNS trials, in contrast to the adverse cardiac outcomes associated with implanted cervical VNS [24, 25, 28, 29].
This converging evidence further supports the distinct safety advantage of non-invasive auricular neuromodulation. By avoiding direct activation of cardiac efferents, taVNS provides not only an anatomic and mechanistic advantage, but also a validated safety margin in clinical practice.
2.3 Mechanisms of Action and Physiological Rationale
Building on the anatomical and clinical distinctions outlined in Sections 2.1 and 2.2, it is important to consider how these differences translate into mechanisms of action and therapeutic potential. This section reviews the mechanisms of both cervical and auricular approaches, with particular attention to the translational advantages of taVNS, which address the limitations of cervical stimulation previously outlined.
2.3.1 Cervical VNS: Activation and Therapeutic Effects
Cervical VNS, whether implanted or transcutaneous, delivers electrical impulses to the cervical trunk of the vagus nerve, which contains both afferent (sensory) and efferent (motor) fibres. Stimulation of this mixed nerve bundle results in activation of a wide range of brainstem and peripheral targets. Afferent signals are transmitted to the nucleus tractus solitarius (NTS) and also to the spinal trigeminal nucleus (SpV), which relays input via secondary pathways to autonomic regulatory centres including the hypothalamus, amygdala, and insular cortex. In contrast, stimulation of the efferents (originating from the dorsal vagal nucleus DVN, and the nucleus ambiguous NA) produces direct parasympathetic output modulating cardiac, respiratory, and gastrointestinal activity without cortical integration or top-down modulation [30].
Implanted cervical VNS theoretically bypasses patient adherence issues via continuous, programmable stimulation, which could be extremely important in conditions with such as epilepsy and depression. However this is usually not the case due to the severity of side effects and complications considered in 2.1. Chronic exposure, while beneficial for some treatment-refractory populations, comes at the cost of higher procedural complexity, long-term device management, and a broader side effect profile [31].
Figure 1. Schematic mechanism representation of cervical vagus nerve stimulation.
The cervical vagus nerve contains both afferent (ascending towards the brainstem) and efferent (descending from the brainstem) fibres, positioned alongside adjacent nerves in the neck, and separated by the carotid sheath housing the vagus nerve. Because of this anatomical complexity, stimulation at the cervical level often leads to non-selective activation and recruitment of nearby fibres. Clinically, this is associated with cardiac side effects (e.g., bradycardia, dyspnea) and vocal cord alterations (e.g., hoarseness, cough reflex). These risks underscore the limitations of cervical VNS compared with taVNS, which selectively targets afferent input without direct activation of cardiac or laryngeal efferents. Downstream effects post-stimulation are not depicted, only the direct effect of stimulation is depicted.
As noted in Section 2.1, in cervical VNS the absence of central filtering can yield unpredictable physiological responses. VNS effects reflect its anatomical targets: negative chronotropic action on the sinoatrial node and negative dromotropic action on the atrioventricular node can reduce heart rate and conduction therapeutically, yet excessive stimulation may provoke symptomatic bradycardia, dizziness, syncope, or hypotension due to impaired cardiac output [13].
In contrast, tcVNS, a non-invasive method aimed at stimulating the cervical vagus externally, faces significant technical challenges. Anatomical targeting is less precise within the carotid sheath, which lies beneath the skin (≈2 mm), superficial fascia (≈3–6 mm), and sternocleidomastoid muscle (≈5–6 mm). As a result, stimulation often activates adjacent motor structures, contributing to variability in outcomes and a higher risk of off-target effects [12, 32]. Moreover, tcVNS lacks reliable control over selective afferent fibres engagement, which may help explain its relatively inconsistent efficacy across studies [12], as well as its diffuse effects, whereby non-vagal cervical nerves and efferent fibres may also be co-stimulated [32].
2.3.2 Auricular VNS: Selective Afferent Modulation
Figure 2. Pathways associated with the auricular vagus nerve stimulation (taVNS).
Schematic illustration of afferent-selective input engaged during auricular stimulation. Electrical activation from the ear is transmitted via vagal afferents (black pathway) to the nucleus tractus solitarius (NTS) and associated brainstem nuclei. This input is dominated by Aδ afferents, with minimal Aβ involvement and no motor fibre recruitment. Importantly, efferent fibres mediating cardiac or laryngeal activity are not directly engaged at the auricular site, although they may be influenced indirectly through central integration. The absence of direct efferent recruitment underlies the favourable safety profile of taVNS. Downstream effects post-stimulation are not depicted, only the direct effect of stimulation is depicted.
taVNS exerts its effects by targeting the ABVN, which innervates specific regions of the outer ear. Unlike the cervical vagus, the ABVN is a purely sensory branch and contains no motor fibres, meaning stimulation avoids direct activation of any efferents. Low-level electrical currents therefore preferentially recruit myelinated sensory axons, with no involvement of unmyelinated C fibres. Safi et al. (2016) reported that approximately half of ABVN myelinated axons fall within the 2.5–4.4 μm diameter range, consistent with Aδ fibres, which are considered the most relevant for mediating therapeutic vagal afferent input to central autonomic circuits [33]. Around 20% of axons exceeded 7 μm in diameter, corresponding to Aβ fibres. However, Aβ activation is generally less desirable, as these fibres primarily transmit mechanoreceptive input and are less likely to engage parasympathetic regulatory circuits compared with Aδ afferents. Their recruitment may therefore contribute more to cutaneous sensation than to therapeutic efficacy, potentially explaining weaker effects on heart rate variability (HRV), inflammation, or autonomic regulation in some individuals. Notably, the ABVN contains almost six times fewer Aβ fibres than the cervical vagus, and the proportion varies considerably between individuals [18, 33, 34].
Stimulation of the ABVN activates vagal afferent fibres that project to the brainstem, where they synapse in the NTS and, to a lesser extent, the SpV From these primary sites, secondary projections engage autonomic motor nuclei, including the DVN and NA, thereby modulating parasympathetic output [18]. Additional studies have demonstrated that vagal afferent activation, via secondary projections from the NTS and SpV, recruits broader networks including the parabrachial nucleus, locus coeruleus, and dorsal raphe nuclei, which are implicated in broader autonomic, arousal, and neuroimmune modulation. Within the central nervous system, interruption and decrease in afferent pathways can reduce vagal efferent outflow and increase cardiac sympathetic activity, which clearly point out the functional impact of afferent signalling [35]. This afferent-selective targeting avoids direct efferent activation, thereby reducing the risk of motor-related adverse effects such as bradycardia, hoarseness, or dysphagia [32, 35].
Inputs to these brainstem nuclei are integrated within central autonomic networks and relayed to subcortical and cortical structures, including the hypothalamus, insular cortex, amygdala, and anterior cingulate cortex. Through these pathways, taVNS modulates parasympathetic tone, stress reactivity, and inflammatory signalling [36–38]. Importantly, by enhancing afferent signalling, taVNS may restore autonomic balance, counteracting sympathetic dominance, and exerting anti-inflammatory effects via the cholinergic anti-inflammatory reflex and broader neuroimmune mechanisms. In parallel, taVNS-driven activation of the locus coeruleus supports norepinephrine release, cognitive flexibility, and stress resilience, while influencing the hypothalamic–pituitary–adrenal axis and adaptive immune responses [30, 35, 36].
Together, these anatomical and physiological mechanisms provide a coherent rationale for taVNS as a selective, safe, and clinically scalable approach. By combining afferent-specific activation (dominated by Aδ fibres), avoidance of efferent recruitment, and engagement of central autonomic networks, taVNS establishes a strong mechanistic foundation for its therapeutic effects across autonomic, inflammatory, and neuropsychiatric conditions.
3. Implementation, Optimisation, and User Experience
3.1. Practical and Comparative Use
While surgically implanted cervical VNS remains a viable intervention for selected conditions, its broader applicability is restricted by surgical implantation requirements, high costs, perioperative risks, long-term device management, and frequent adverse effects (see Section 2.1) [6–16]. These limitations confine its use largely to severe cases within specialist clinical settings. Moreover, many patients are unsuitable candidates due to comorbidities, anaesthetic risk, or anatomical contraindications such as prior neck surgery, while those with milder or earlier-stage conditions often do not meet thresholds for invasive intervention [39].
Both tcVNS and taVNS offer practical advantages over implanted VNS, including lower cost and reduced procedural complexity, while eliminating the need for anatomical imaging or surgical navigation. However, effective tcVNS stimulation, as exemplified by the gammaCore device (electroCore Inc., Basking Ridge, US), requires precise cervical placement and must be manually held during use, with its dose-limited design reducing long-term cost-effectiveness [12, 21].
TaVNS, in particular, benefits from the superficial location of the ABVN (see Section 2.3.2) [11, 12, 17, 18]. Combined with its non-invasive nature and excellent tolerability, as detailed in Section 2.2, taVNS emerges as a scalable and cost-effective neuromodulatory modality [24–27].
3.1. Clinical Feasibility and Adherence
Building on the favourable safety profile discussed in Section 2.2, taVNS demonstrates strong clinical feasibility across outpatient and home-based contexts [25, 28]. Its straightforward application and minimal technical requirements make it accessible to a broad range of patients without specialised training or the logistical burden of clinical oversight. Importantly, this ease of integration supports sustained use in everyday care settings, making taVNS highly compatible with decentralised care models. [28, 5]. These features make taVNS, especially relevant in chronic disease management and long-term adherence contexts [37, 38], even among high-risk populations [28].
Importantly, adherence rates with taVNS appear to be substantially higher than those observed with standard pharmacological therapies. In the clinical study by Stavrakis et al. (2022) on patients with heart failure with preserved ejection fraction, which employed the Parasym AVNT device, adherence to daily stimulation over 3 months reached 92%, even among older heart failure patients, a group typically associated with poor long-term adherence [40]. By comparison, only 43% of patients maintain adherence to cardiovascular preventsion drugs, 54% at 1–3 months for heart failure medications, and 74–78% for oral anticoagulants in atrial fibrillation [41–43].
These findings suggest that taVNS may provide a more sustainable, user-friendly alternative to conventional treatments, particularly in populations burdened by polypharmacy, cognitive decline, or limited access to follow-up care. The combination of safety, ease of use, and high compliance also supports the integration of taVNS into remote monitoring frameworks and self-guided chronic care programs. Nonetheless, ongoing evaluation of long-term safety and durability of therapeutic effects remains essential, especially as taVNS expands into chronic care pathways and younger or otherwise healthy populations.
3.3 Stimulation Thresholds and Technical Considerations
Recent developments in taVNS technology have highlighted the critical role of targeting accuracy and dose optimisation in achieving therapeutic efficacy while minimising off-target effects. The Parasym device employs threshold calibration and electrode orientation features to support consistent stimulation of the ABVN [28]. When applied correctly to the tragus, or to other ABVN-innervated auricular regions such as the cymba conchae, this approach allows for selective activation of afferent sensory fibres, avoiding deeper motor fibre activation and related discomfort as discussed in the section 2.3.2 [12, 18, 34].
While the cymba concha is a common stimulation target, several studies mentioned practical limitations due to anatomical variability, including inconsistent electrode fit and the need for custom supports to ensure stable contact [44, 45, 46]. The tragus and external acoustic meatus (EAM) provide larger, more accessible contact surfaces that enable higher focality and selectivity of current flow. This may offer more consistent, user-friendly stimulation, supporting broader clinical and home-based use [45, 46]. Unlike cervical invasive approaches, taVNS does not require identification of deep neurovascular structures or operative access, thereby avoiding surgical implantation, perioperative care, and device explantation in non-responders [47].
The importance of fibre-specific engagement has been further underscored by recent findings in cervical VNS. A study by Blanz et al. (2023) demonstrated that even minor changes in electrode placement along the cervical vagus nerve can shift the balance between desired therapeutic effects (e.g., heart rate modulation) and unintended side effects (e.g., neck muscle contractions), due to the underlying fascicular anatomy [7]. While their work focused on implanted epineural electrodes in large animal models, the principles of spatial selectivity and neuroanatomy-guided stimulation are also relevant for non-invasive taVNS approaches, where electrode positioning and contact precision remain central to clinical outcomes [7].
Techniques to improve fibre specificity, such as neural sheath-targeted stimulation and research-guided threshold titration, are being explored in the context of taVNS. The Parasym AVNT system applies research-guided titration through anatomical targeting and waveform design, using stimulation parameters derived from experimental studies to standardise application for broader use. As taVNS continues to expand across clinical indications, the development of standardised stimulation protocols, including intensity, frequency, and duration, will be essential to ensure reproducibility, safety, and cross-study comparability [7, 48]. However, at present, no universally accepted protocols have been adopted for taVNS, even within specific clinical indications. Published studies vary considerably in terms of stimulation site, intensity, frequency, pulse width, and session duration. This heterogeneity complicates direct comparison across trials and limits the ability to draw firm conclusions about optimal dosing parameters or treatment efficacy [5, 25, 48, 49].
Reported stimulation parameters vary widely: intensity was generally titrated to remain below the individual pain threshold but above the sensory threshold, within the milliampere ranges permitted by International Electrotechnical Commission standards in medical equipment (IEC 60601-1) for vagal stimulation, pulse widths ranged from 100–500 µs, and frequencies typically centred around 5–50?Hz. Treatment schedules varied widely, with daily durations ranging from 12 to 360 minutes and total intervention periods spanning from a few sessions to up to one year. Importantly, stimulation is determined not only by intensity, pulse width, and frequency but also by waveform features, such as shape, polarity, and temporal profile, as well as electrode-related factors such as conductivity and contact quality. These elements vary between studies and devices, and are often overlooked, making results obtained through different devices virtually uncompilable [5, 12]. Such variability reflects both the evolving nature of the field and differences in targeted conditions, underscoring the urgent need for protocol standardisation to optimise therapeutic outcomes.
4. Therapeutic Scope and Emerging Evidence
4.1 Broad Applicability Across Conditions
Cervical VNS has shown promise in selected indications, its broader therapeutic application is limited by procedural complexity, and by practical challenges in conducting research within emerging clinical contexts, particularly in preventsive, or lower-acuity indications, where surgical implantation is neither feasible nor appropriate compared to auricular approaches [20]. TaVNS is currently being investigated across a growing range of clinical conditions, reflecting its engagement with core physiological functions such as autonomic regulation, inflammation, and neurocognitive function. Active research spans several therapeutic domains, including mental health conditions, neurocognitive conditions, inflammatory and autoimmune conditions, cardiovascular and metabolic conditions [49].
This wide therapeutic reach likely stems from the integrative role of the vagus nerve in maintaining physiological homeostasis. Through its afferent and efferent pathways, vagal activity influences cardiovascular control, inflammation, gastrointestinal function, and neurocognitive processes [50]. As a result, taVNS is increasingly viewed as a versatile neuromodulation tool, with expanding clinical trials exploring its use across acute, chronic, and post-infectious conditions.
4.2 Focus on Dysautonomia and Comorbidities
Dysautonomia has emerged as a key therapeutic focus for taVNS, particularly in conditions such as postural orthostatic tachycardia syndrome (POTS) and long COVID, where autonomic dysfunction, immune dysregulation, and chronic inflammation are prominent and well documented. These syndromes are often post-viral in origin (e.g., following SARS-CoV-2 infection) and associated with mechanisms such as microclot formation, autoantibody generation, and impaired interferon signaling, all of which may be influenced by vagal modulation [51].
A recent randomised controlled trial published in Journal of the American College of Cardiology in patients with POTS (n = 26) employed daily taVNS using the Parasym device (1 hour/day for 2 months). The study reported significant reductions in postural tachycardia, decreased autoantibodies against α1- and β1-adrenergic receptors, and improved HRV. Nearly half the participants (46%) had a confirmed COVID-19 infection within the prior 12 months. Notably, no device-related adverse events were observed [52].
Additional uncontrolled studies using the same taVNS platform in patients with long COVID, ME/CFS, and fibromyalgia have also demonstrated improvements across multiple clinical domains. Reported benefits included reductions in fatigue, anxiety, depression, and widespread pain, as well as improvements in sleep, cognitive symptoms, and overall quality of life. Notably, positive effects on oxygen saturation and grip strength were observed following stimulation protocols lasting from 10 days up to 6 weeks. Importantly, no SAEs were reported in any of these studies [53–56].
Mechanistic investigations have further shown that taVNS with the Parasym system enhances parasympathetic tone [57], lowers pro-inflammatory cytokines [37, 58], and improves baroreflex sensitivity [38], physiological changes that align with key aspects of dysautonomia pathophysiology. To date, across a combined cohort of more than 390 patients treated with this system for dysautonomia-related conditions, no SAEs have been reported [59].
Taken together, these findings suggest that taVNS, particularly when delivered through anatomically targeted systems such as the Parasym device, offers a well-tolerated and potentially effective non-pharmacological treatment for patients with complex post-infectious syndromes. While early data are encouraging, it is important to acknowledge that dysautonomia encompasses a heterogeneous group of syndromes with diverse etiologies, diagnostic challenges, and clinical trajectories. Larger, placebo-controlled studies with longer-term follow-up will be essential to confirm efficacy, establish durability of benefit, and define the patient populations most likely to respond.
5. Regulatory Trends and Technological Development
5.1 Movement Towards Non-Invasive Solutions
The evolution of neuromodulatory therapies reflects a broader trend in medicine towards non-invasive, patient-friendly interventions. Given the favourable safety profile, ease of use, and growing evidence supporting clinical efficacy, regulatory interest is expected to increasingly focus on transcutaneous rather than surgically implanted solutions. At present, US FDA approvals remain weighted towards invasive modalities: implanted cervical VNS, which is approved for epilepsy, resistant depression, and stroke rehabilitation [1–3] as well as some percutaneous devices [60]. Non-invasive approvals are more limited; to the best of current knowledge, only one tcVNS device gammaCore cleared by the FDA for migraine and cluster headache [21, 31]. taVNS originated in Europe, where devices are regulated under CE-mark for medical devices frameworks, the former Medical Device Directive and the current Medical Device Regulation. Based on current evidence in the literature, only two systems: Parasym and NEMOS (Cerbomed, Erlangen, Germany), have been cleared for medical use [28, 31].
TaVNS minimal risk of adverse effects and suitability for outpatient or at-home use, aligns well with emerging health system priorities, including decentralised care, long-term management of chronic disease, and the integration of neuromodulation into digital therapeutics platforms. These attributes also support the potential for broader population-level implementation, particularly in settings where invasive approaches may be less feasible. However, despite promising early findings, most clinical studies to date have involved small sample sizes, short follow-up durations, or limited methodological consistency, underscoring the need for larger, multicentre trials to confirm efficacy across indications [25, 5, 48, 49].
5.2 Industry Innovation
Technological innovation in taVNS is playing a central role in enabling broader clinical use. Recent developments have focused on improving stimulation precision, optimising dosing parameters, and enhancing user adherence. Multiple manufacturers are contributing to this space, with devices increasingly incorporating individualised threshold calibration and electrode positioning. Among these, the Parasym system has been utilised in numerous peer-reviewed studies, contributing significantly to the current clinical evidence base [52–58]. However, it remains essential that future research continues to evaluate device-specific outcomes, as differences in design and stimulation parameters may influence safety and efficacy. This approach will help to ensure generalisability and inform evidence-based regulatory approval processes.
Substantial variability in stimulation parameters, electrode configurations, and calibration protocols persists across devices, highlighting the need for standardised technical guidelines and outcome measures [5, 25, 48, 49]. Establishing such standards will be critical to achieving consistent therapeutic effects, supporting regulatory review, and enabling meaningful comparison across clinical studies. Current evidence is limited in identifying predictors of treatment response, and factors such as age, sex, baseline autonomic function, and comorbidities may influence outcomes. Establishing clear inclusion frameworks will be essential for tailoring treatment, improving efficacy, and guiding future clinical trial design.
6. Conclusion
While this review emphasises the translational potential and usability of taVNS, it does not aim to diminish the established clinical value of cervical VNS, particularly in specialist settings or cases requiring long-term implanted solutions. Implanted cervical VNS may still offer viable therapies in severe or treatment-refractory conditions, especially where continuous, programmable stimulation is therapeutically advantageous.
By contrast, non-invasive tcVNS has yet to demonstrate consistent efficacy or fibre-specific targeting. Its anatomical imprecision and increased risk of off-target effects limit its reliability as a neuromodulatory option. In this context, taVNS emerges as the most promising non-invasive alternative currently available.
This review provides a narrative perspective contrasting taVNS with both implanted and transcutaneous cervical VNS across mechanistic, safety, and usability domains, highlighting considerations relevant to side-effect profiles, potential clinical utility and future neuromodulation research. TaVNS represents a paradigm shift in bioelectronic medicine. It lays a strong foundation for broader clinical adoption by offering a non-invasive, anatomically targeted, and well-tolerated alternative to traditional cervical approaches, it addresses key limitations in safety, scalability, and user accessibility.
As clinical evidence accumulates, particularly in cardiovascular regulation, immune modulation, and autonomic dysfunction, taVNS is steadily transitioning from an investigational tool into a clinically actionable therapy. Future directions will likely focus on refining stimulation parameters, standardising protocols, expanding regulatory indications, and integrating taVNS into multimodal care pathways. Identifying patient populations most likely to benefit, as well as optimal treatment timing, will be crucial for maximising therapeutic impact and enabling personalised interventions.
Importantly, while many systematic reviews have addressed VNS, few have focused specifically on contrasting cervical and auricular approaches. This review adds to existing literature by summarising current knowledge on the safety and usability of taVNS in comparison with cervical VNS, with the goal of supporting further investigation into its clinical potential.
In this evolving therapeutic landscape, auricular neuromodulation is well positioned not only to complement existing treatments, but to lead the next wave of safe, scalable, and patient-centred innovation. Continued interdisciplinary collaboration among clinicians, engineers, and regulatory bodies will be essential to fully realise its potential in routine clinical practice.
References
1. U.S. Food and Drug Administration. (1997). PMA approval P970003 for VNS Therapy System. Retrieved June 6, 2025, from [URL]
2. U.S. Food and Drug Administration. (2005). PMA approval P970003 for VNS Therapy System. Retrieved June 6, 2025, from [URL]
3. U.S. Food and Drug Administration. (2021). PMA approval P2100007 for Vivistim System. Retrieved June 24, 2025, from [URL]
4. Hampton, T., et al. (2015). Electric stimulation device approved to treat obesity. JAMA, 313(8), 785. https://doi.org/10.1001/jama.2015.234
5. Gerges, A. N. H., & Williams, E. E. R., et al. (2024). Clinical application of transcutaneous auricular vagus nerve stimulation: A scoping review. Disability and Rehabilitation, 46(24), 5730–5760. https://doi.org/10.1080/09638288.2024.2313123
6. Hammer, N., & Löffler, S., et al. (2018). Cervical vagus nerve morphometry and vascularity in the context of nerve stimulation: A cadaveric study. Scientific Reports, 8, 7997. https://doi.org/10.1038/s41598-018-26135-8
7. Stephan, L., & Blanz, S., et al. (2023). Spatially selective stimulation of the pig vagus nerve to modulate target effect versus side effect. Journal of Neural Engineering, 20(1), 016051. https://doi.org/10.1088/1741-2552/acb3fd
8. Kreuzer, P. M., & Landgrebe, M., et al. (2012). Transcutaneous vagus nerve stimulation: Retrospective assessment of cardiac safety in a pilot study. Frontiers in Psychiatry, 3, 70. https://doi.org/10.3389/fpsyt.2012.00070
9. Muthulingam, J. A., & Olesen, S. S., et al. (2021). Cervical transcutaneous vagal neuromodulation in chronic pancreatitis patients with chronic pain: A randomized sham-controlled clinical trial. PLoS ONE, 16(2), e0247653. https://doi.org/10.1371/journal.pone.0247653
10. Fitchett, A., & Mastitskaya, S., et al. (2021). Selective neuromodulation of the vagus nerve. Frontiers in Neuroscience, 15, 685872. https://doi.org/10.3389/fnins.2021.685872
11. Chen, M., & Yu, L., et al. (2015). The right side or left side of noninvasive transcutaneous vagus nerve stimulation: Based on conventional wisdom or scientific evidence? International Journal of Cardiology, 187:44-45. https://doi.org/10.1016/j.ijcard.2015.03.351
12. Yap, J. Y. Y., & Keatch, C., et al. (2020). Critical review of transcutaneous vagus nerve stimulation: Challenges for translation to clinical practice. Frontiers in Neuroscience, 14, 284. https://doi.org/10.3389/fnins.2020.00284
13. Capilupi, M. J., & Kerath, S. M., et al. (2020). Vagus nerve stimulation and the cardiovascular system. Cold Spring Harbor Perspectives in Medicine, 10(2), a034173. https://doi.org/10.1101/cshperspect.a034173
14. Kato, H., & Fujimoto, A., et al. (2018). New onset syncopal events following vagus nerve stimulator implantation might be key to preventsing vagus nerve stimulation-induced symptomatic bradycardia: A case report and review. Epilepsy & Behavior Case Reports, 10, 57–60. https://doi.org/10.1016/j.ebcr.2018.04.004
15. Kumar, A., & Goyal, A., et al. (2023). Vagal stimulation causing intra-operative cardiac arrest: A major dilemma whether to proceed or to defer surgery: A case report. Exploratory Research and Hypothesis in Medicine, 8(1), 77–80. https://doi.org/10.14218/ERHM.2021.00063
16. Potenza, S., & Tavone, A. M., et al. (2021). Death by stimulation of the left vagus nerve in a case of neck grasping in a healthy young man. Medico-Legal Update, 21(2), 1122–1125. [URL]
17. Brauer, P. R., & Lamarre, E. D., et al. (2023). Laryngology outcomes following implantable vagus nerve stimulation. JAMA Otolaryngology–Head & Neck Surgery, 149(1), 49–53. https://doi.org/10.1001/jamaoto.2022.3699
18. Butt, M. F., & Albusoda, A., et al. (2019). The anatomical basis for transcutaneous auricular vagus nerve stimulation. Journal of Anatomy, 235(3), 489–510. https://doi.org/10.1111/joa.13122
19. Wang, L., & Gao, F., et al. (2023). Transcutaneous auricular vagus nerve stimulation in the treatment of disorders of consciousness: Mechanisms and applications. Frontiers in Neuroscience, 17, 1286267. https://doi.org/10.3389/fnins.2023.1286267
20. Kalagara, R., & Chennareddy, S., et al. (2025). Complications of implanted vagus nerve stimulation: A systematic review and meta-analysis. Cerebrovascular Diseases, 54(1), 112–120. https://doi.org/10.1159/000539878
21. Mwamburi, M., & Liebler, E. J., et al. (2017). Review of non-invasive vagus nerve stimulation (gammaCore): Efficacy, safety, potential impact on comorbidities, and economic burden for episodic and chronic cluster headache. American Journal of Managed Care, 23(17 Suppl), S317–S325. [URL]
22. Costa, V., & Gianlorenço, A. C., et al. (2024). Transcutaneous vagus nerve stimulation effects on chronic pain: Systematic review and meta-analysis. Pain Reports, 9(5), e1171. https://doi.org/10.1097/PR9.0000000000001171
23. Redgrave, J., & Day, D., et al. (2018). Safety and tolerability of transcutaneous vagus nerve stimulation in humans: A systematic review. Brain Stimulation, 11(6), 1225–1238. https://doi.org/10.1016/j.brs.2018.08.010
24. Tan, H., & Qiao, M., et al. (2023). The efficacy and safety of transcutaneous auricular vagus nerve stimulation in depressive disorder: A systematic review and meta-analysis of randomized controlled trials. Journal of Affective Disorders, 337, 37–49. https://doi.org/10.1016/j.jad.2023.05.048
25. Kim, A. Y., & Marduy, A., et al. (2022). Safety of transcutaneous auricular vagus nerve stimulation (taVNS): A systematic review and meta-analysis. Scientific Reports, 12, 22055. https://doi.org/10.1038/s41598-022-25864-1
26. van der Groen, O., & Rafique, S. A., et al. (2025). Transcutaneous and transcranial electrical stimulation for enhancing military performance: An update and systematic review. Frontiers in Human Neuroscience, 19, 1501209. https://doi.org/10.3389/fnhum.2025.1501209
27. Gerges, A. N. H., & Williams, E. E. R., et al. (2024). Clinical application of transcutaneous auricular vagus nerve stimulation: A scoping review. Disability and Rehabilitation, 46(24), 5730–5760. https://doi.org/10.1080/09638288.2024.2313123
28. Luche, G., & Dundovic, S., et al. (2024). First report of safety and tolerability of low-level tragus vagal neuromodulation in cardiovascular patients. Journal of the American College of Cardiology, 83(13 Suppl), 178. https://doi.org/10.1016/S0735-1097(24)02168-5
29. Wang, W., & Li, R., et al. (2024). Advances in VNS efficiency and mechanisms of action on cognitive functions. Frontiers in Physiology, 15, 1452490. https://doi.org/10.3389/fphys.2024.1452490
30. Fang, Y. T., & Lin, Y. T., et al. (2023). Neuroimmunomodulation of vagus nerve stimulation and the therapeutic implications. Frontiers in Aging Neuroscience, 15, 1173987. https://doi.org/10.3389/fnagi.2023.1173987
31. Ben-Menachem, E., & Revesz, D., et al. (2015). Surgically implanted and non-invasive vagus nerve stimulation: A review of efficacy, safety and tolerability. European Journal of Neurology, 22(9), 1260–1268. https://doi.org/10.1111/ene.12629
32. Farmer, A. D., & Strzelczyk, A., et al. (2021). International consensus-based recommendations for minimum reporting standards in transcutaneous vagus nerve stimulation research. Frontiers in Human Neuroscience, 14, 568051.https://doi.org/10.3389/fnhum.2020.568051
33. Safi, S., & Ellrich, J., et al. (2016). Myelinated axons in the auricular branch of the human vagus nerve. Anatomical Record, 299(9), 1184–1191.https://doi.org/10.1002/ar.23391
34. Kollarik, M., & Ru, F., et al. (2010). Vagal afferent nerves with the properties of nociceptors. Autonomic Neuroscience, 153(1–2), 12–20. https://doi.org/10.1016/j.autneu.2009.08.001
35. van Weperen, V. Y. H., & Vaseghi, M. (2023). Cardiac vagal afferent neurotransmission in health and disease: Review and knowledge gaps. Frontiers in Neuroscience, 17, 1192188. https://doi.org/10.3389/fnins.2023.1192188
36. Farhat, K., & Niewiadomska, M., et al. (2022). Transcutaneous vagus nerve stimulation restores the cardiac phenotype in heart failure with preserved ejection fraction by modulating the immune cell profile. Circulation, 146. https://doi.org/10.1161/circ.146.suppl_1.13645
37. Dasari, T. W., & Akhtar, K. H., et al. (2023). Effects of low level tragus stimulation on inflammation in acute decompensated heart failure. Journal of Cardiovascular Electrophysiology, 29(4), 660–661.https://doi.org/10.1111/jce.15827
38. Gentile, F., & Giannoni, A., et al. (2025). Acute right-sided transcutaneous vagus nerve stimulation improves cardio-vagal baroreflex gain in patients with chronic heart failure. Clinical Autonomic Research, 35, 75–85. https://doi.org/10.1007/s10286-024-01074-9
39. Spuck, S., & Tronnier, V., et al. (2010). Operative and technical complications of vagus nerve stimulator implantation. Neurosurgery, 67, 489–494. https://doi.org/10.1227/NEU.0b013e3181f88867
40. Stavrakis, S., & Elkholey, K., et al. (2022). Neuromodulation of inflammation to treat heart failure with preserved ejection fraction: A pilot randomized clinical trial. Journal of the American Heart Association, 11(3), e023582. https://doi.org/10.1161/JAHA.121.023582
41. Kronish, I. M., & Ye, S. (2013). Adherence to cardiovascular medications: Lessons learned and future directions. Progress in Cardiovascular Diseases, 55(6), 590–600. https://doi.org/10.1016/j.pcad.2013.02.001
42. Wu, J. R., & Moser, D. K., et al. (2018). Medication adherence in patients who have heart failure: A review of the literature. Journal of Cardiovascular Nursing, 33(1), 40–47. https://doi.org/10.1016/j.cnur.2007.10.006
43. An, Y., & Xu, H., et al. (2021). Real?world persistence and adherence to oral anticoagulants in patients with atrial fibrillation: A population?based study. Journal of the American Heart Association, 10(21), e021601. https://doi.org/10.1161/JAHA.121.021601
44. Bretherton, B., & Murray, A., et al. (2023). The autonomic effects of transcutaneous auricular nerve stimulation at different sites on the external auricle of the ear. bioRxiv. https://doi.org/10.1101/2023.09.14.557755
45. Phillips, I., & Johns, M. A., et al. (2025). Three hundred hertz transcutaneous auricular vagus nerve stimulation (taVNS) impacts pupil size non-linearly as a function of intensity. Psychophysiology, 62(2), e70011. https://doi.org/10.1111/psyp.70011
46. Kreisberg, E., & Esmaeilpour, Z., et al. (2022). High-resolution computational modeling of the current flow in the outer ear during transcutaneous auricular vagus nerve stimulation (taVNS). Brain Stimulation, 15(1), 108–120. https://doi.org/10.1016/j.brs.2021.09.001
47. Ben-Menachem, E., & Hellström, K., et al. (2002). Analysis of direct hospital costs before and 18 months after treatment with vagus nerve stimulation therapy in 43 patients. Neurology, 59(6 Suppl 4), S44–S47.
48. Badran, B. W., & DeVries, W. H., et al. (2018). Neurophysiologic effects of transcutaneous auricular vagus nerve stimulation via electrical stimulation of the tragus: A concurrent taVNS/fMRI study and review. Brain Stimulation, 11(3), 492–500. https://doi.org/10.1016/j.brs.2017.12.009
49. Machetanz K, Berelidze L, & Guggenberger R and Gharabaghi A. (2021) Brain–Heart Interaction During Transcutaneous Auricular Vagus Nerve Stimulation. Front. Neurosci. 15:632697.https://doi.org/10.3389/fnins.2021.632697
50. Ottaviani, M. M., & Macefield, V. G. (2022). Structure and functions of the vagus nerve in mammals. Comprehensive Physiology, 12(4), 3989–4037. https://doi.org/10.1002/cphy.c210042
51. Chakraborty, P., & Farhat, K., et al. (2023). Non-invasive vagus nerve stimulation in postural orthostatic tachycardia syndrome. Arrhythmia & Electrophysiology Review, 12, e31 .https://doi.org/10.15420/aer.2023.20
52. Stavrakis, S., & Chakraborty, P., et al. (2024). Noninvasive vagus nerve stimulation in postural tachycardia syndrome: A randomized clinical trial. JACC: Clinical Electrophysiology, 10(2), 346–355. https://doi.org/10.1016/j.jacep.2023.10.015
53. Dolcini, G., & Favretti, M., et al. (2025). Vagal nerve stimulation and fibromyalgia: An additional therapeutic option. Clinical and Experimental Rheumatology, 43(6), 1095–1104. https://doi.org/10.55563/clinexprheumatol/johqvo
54. Verbanck, P., & Clarinval, A. M., et al. (2021). Transcutaneous auricular vagus nerve stimulation (tVNS) can reverse the manifestations of the long-COVID syndrome: A pilot study. Frontiers in Neurology and Neuroscience Research, 2, 100011. https://doi.org/10.51956/FNNR.100011
55. Zheng, Z. S., & Simonian, N., et al. (2024). Transcutaneous vagus nerve stimulation improves Long COVID symptoms in a female cohort: A pilot study. Frontiers in Neurology, 15, 1393371. https://doi.org/10.3389/fneur.2024.1393371
56. Natelson, B. H., & Blate, M., et al. (2022). Transcutaneous vagus nerve stimulation in the treatment of long COVID–chronic fatigue syndrome. medRxiv. https://doi.org/10.1101/2022.11.08.22281807
57. Stavrakis, S., & Elkholey, K., et al. (2022). Neuromodulation of inflammation to treat heart failure with preserved ejection fraction: A pilot randomized clinical trial. Journal of the American Heart Association, 11(3), e023582. https://doi.org/10.1161/JAHA.121.023582
58. Nagai, M., & Dote, K., et al. (2023). Afterload reduction after non-invasive vagus nerve stimulation in acute heart failure. Frontiers in Human Neuroscience, 17, 1149449. https://doi.org/10.1002/ar.23391
59. Dalle Luche, G., & Burchi, E., et al. (2025). Auricular vagal neuromodulation therapy explained: How one technique can affect multiple autonomic-related mechanisms [Poster presentation]. Dysautonomia International Conference, Parasym Scientific Ltd, London, UK
60. Zagorski N. FDA Clears Way for Stimulation Device to Treat Opioid Withdrawal Symptoms.(2017). Psychiatrist News 53 (1). https://doi.org/10.1176/appi.pn.2018.1a16.
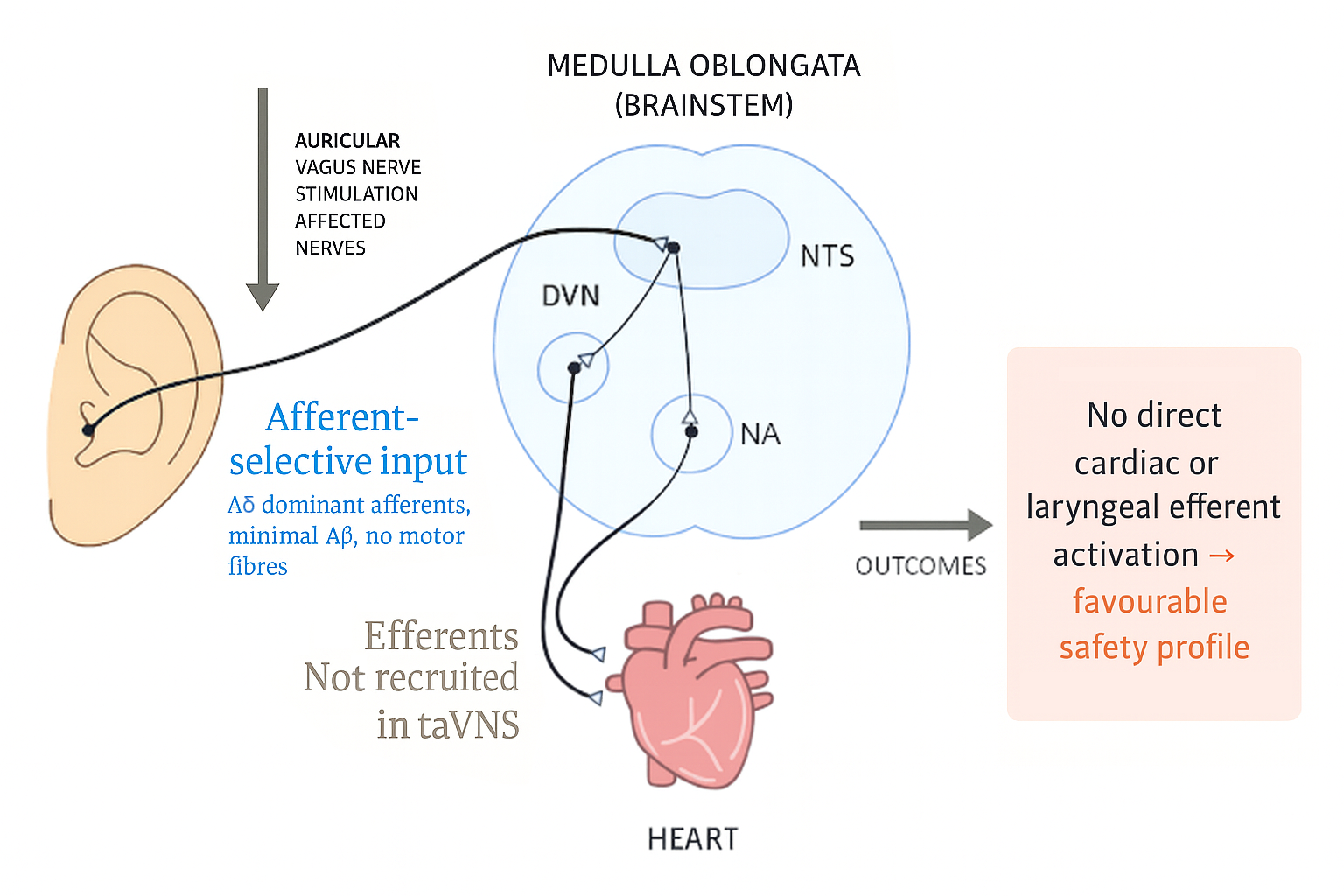
Figure 1
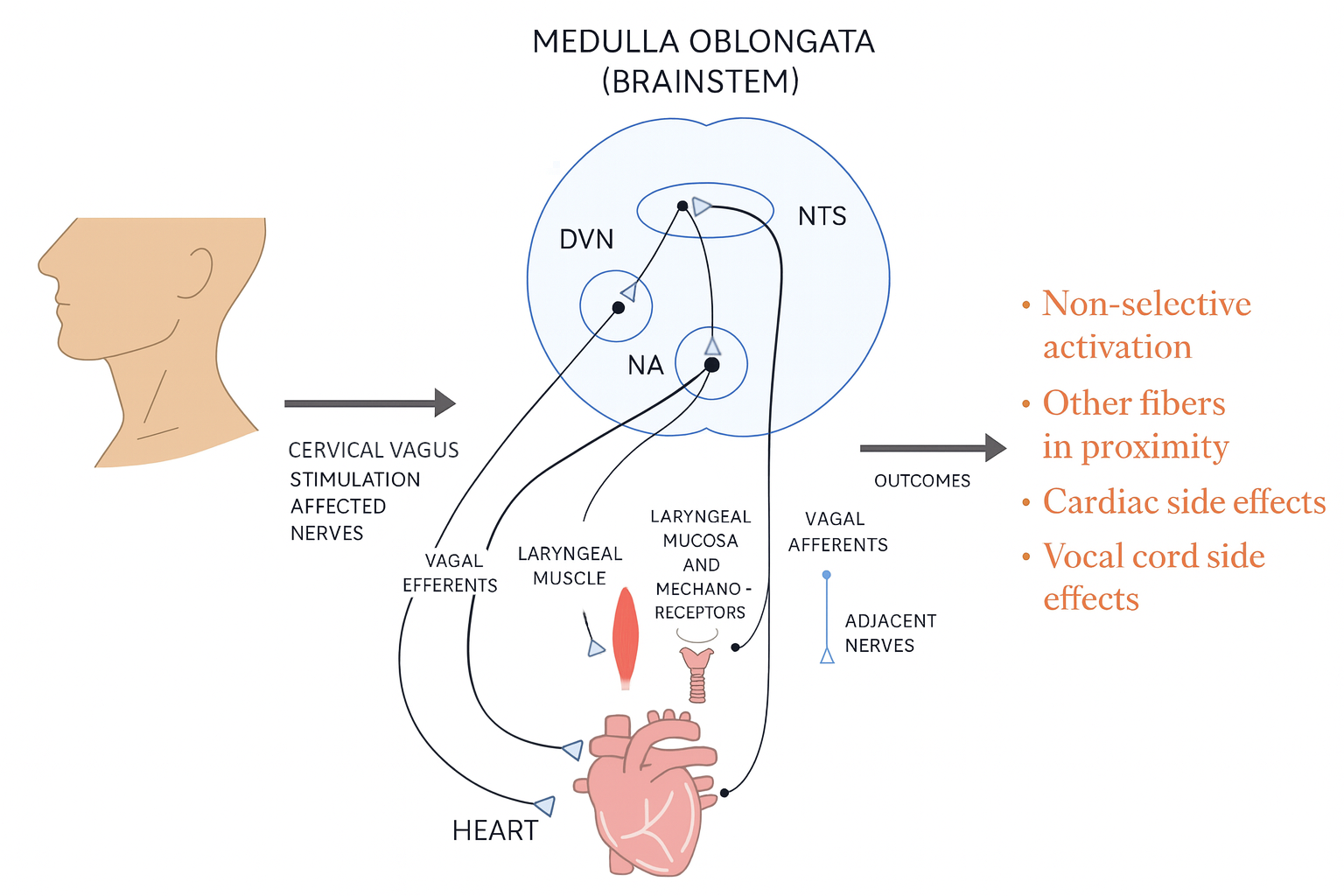
Figure 2
