Dosimetric Outcomes of IMRT and VMAT in Head and Neck Radiotherapy: Retrospective Institutional Experience.
Dosimetric Outcomes of IMRT and VMAT in Head and Neck Radiotherapy: Retrospective Institutional Experience.
Sunil Kumar*1, Rashmi Yadav2
*Correspondence to: Sunil Kumar, India.
Copyright.
© 2025 Sunil Kumar This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 03 Sep 2025
Published: 11 Sep 2025
DOI: https://doi.org/10.5281/zenodo.17285005
Abstract
Background: Head and neck cancers (HNC) pose significant challenges for radiotherapy due to their proximity to critical organs-at-risk (OARs). While intensity-modulated radiation therapy (IMRT) remains the gold standard for dose conformity, volumetric modulated arc therapy (VMAT) has emerged as a promising advancement, offering dynamic arc-based delivery with potential gains in efficiency and dosimetric precision.
Objective: To compare the dosimetric performance of IMRT and VMAT in HNC radiotherapy, focusing on target coverage, OAR sparing, treatment efficiency, and plan quality.
Methods: In this retrospective study, 40 patients with histologically confirmed head and neck squamous cell carcinoma (HNSCC) underwent dual planning (IMRT and VMAT) using identical target volumes and dose constraints. Dosimetric parameters including PTV coverage (D95%), mean/maximum OAR doses, homogeneity index (HI), conformity index (CI), and monitor units were analyzed. Statistical comparisons used paired t-tests (SPSS v26).
Results: Both modalities achieved clinically acceptable PTV coverage. VMAT demonstrated superior dose conformity (CI: 1.16 vs. 1.18, p < 0.05) and homogeneity (HI: 0.050 vs. 0.056, p < 0.05) compared to IMRT. VART significantly reduced maximum doses to the spinal cord (27.61 vs. 28.71 Gy, p < 0.05) and PRV cord (31.39 vs. 32.58 Gy, p < 0.05), while lowering mean dose of mandible (62.13 vs. 65.13 Gy, p < 0.05) and mean of skin Dmax (65.01 vs. 65.81 Gy, p < 0.05).
Conclusion: VMAT outperformed IMRT in HNC radiotherapy, delivering superior dose homogeneity, conformity, and OAR sparing while significantly shortening treatment time. These advantages position VMAT as the preferred modality for balancing efficacy with reduced toxicity risks in clinical practice.
Dosimetric Outcomes of IMRT and VMAT in Head and Neck Radiotherapy: Retrospective Institutional Experience.
Introduction
Head and neck cancers (HNC), encompassing malignancies of the oral cavity, oropharynx, hypopharynx, and larynx, represent approximately 4% of global cancer diagnoses and are a leading cause of cancer-related morbidity and mortality [1]. Radiotherapy is a cornerstone of HNC management, often delivered as definitive treatment or adjuvant therapy alongside surgery and chemotherapy [2, 3].
Delivering curative radiation doses in HNC is complicated by the intricate anatomy of the region, where tumors frequently abut critical structures such as the spinal cord, parotid glands, mandible, pharyngeal constrictors, and skin [4]. Excessive irradiation of these organs-at-risk (OARs) can lead to debilitating toxicities, including xerostomia, dysphagia, osteoradionecrosis, and neurocognitive deficits, profoundly affecting long-term patient quality of life [5–7].
Intensity-modulated radiation therapy (IMRT) emerged as a transformative advancement over conventional techniques like 3 D conformal radiotherapy, enabling shaped dose distributions that conform to irregular tumor geometries while sparing adjacent OARs [8,9]. Clinical studies have validated IMRT’s ability to reduce toxicities, particularly xerostomia, through precise sparing of salivary glands [10–12]. Volumetric modulated arc therapy (VMAT), a more recent innovation, builds upon IMRT principles by delivering radiation continuously during gantry rotation, dynamically modulating multileaf collimator (MLC) positions, dose rate, and gantry speed [13]. This approach promises increase treatment efficiency via shorter delivery times while maintaining or improving dosimetric quality [14–16].
Despite these advances, the comparative merits of IMRT and VMAT in HNC remain debated. While some studies report that VMAT achieves comparable or superior OAR sparing and target coverage with reduced treatment times [17–19], others highlight concerns about increased low-dose radiation exposure to healthy tissues (“low-dose bath”), potentially elevating risks of secondary malignancies or late toxicities [20,21]. These discrepancies underscore the need for robust, clinically grounded comparisons to guide modality selection.
This study leverages institutional planning data to perform a comprehensive dosimetric evaluation of IMRT and VMAT in HNC, assessing target coverage, OAR sparing, dose homogeneity/conformity, and delivery efficiency. By addressing conflicting evidence in the literature, we aim to inform evidence-based clinical decision-making in an evolving therapeutic landscape.
Materials and Methods
Study Design and Patient Selection
This retrospective dosimetric analysis included 40 consecutive patients with histologically confirmed squamous cell carcinoma of the head and neck (HNSCC) mostly locally advance stage, treated at the Hind Institute of Medical Sciences, Barabanki, between January 2023 and March 2024.
Inclusion Criteria:
Histologically proven Head and neck squamous cell carcinoma (oral cavity, oropharynx, hypopharynx, or larynx).
Karnofsky Performance Status (KPS) ≥70.
Normal hematologic, hepatic, and renal function.
Exclusion Criteria:
Prior or synchronous malignancy.
History of head and neck surgery.
Previous radiotherapy to the head and neck region.
Distant metastases (confirmed by PET-CT/contrast-enhanced CT)
Ethical Approval
The study protocol was approved by the Institutional Ethics Committee of Hind Intitute of Medical Sciences Barabanki (Approval No. [Insert Number]), adhering to the Declaration of Helsinki.
Imaging and Target Delineation
Simulation:
Patients underwent contrast-enhanced CT simulation (Siemens SOMATOM Go Now, 3 mm slice thickness) in a supine position with a thermoplastic immobilization mask.
Head and neck positioning was standardized using a customized headrest to maintain neutral neck alignment.
Target Volumes (ICRU 83 [22]):
GTV: Gross tumor delineated using clinical examination and imaging (CT/MRI/PET-CT fusion).
CTV: GTV + 5mm mm margin (anatomical boundaries respected) to encompass microscopic disease.
PTV: CTV + 5 mm isotropic margin for setup uncertainties.
Organs-at-Risk (OARs):
Contoured per ICRU 62 guidelines: spinal cord, spinal cord planning risk volume (PRV; 2 mm margin), bilateral parotid/submandibular glands, mandible, lips, thyroid, esophagus, skin (3 mm depth), and pharyngeal constrictors.
Treatment Planning
IMRT Planning:
Beam Arrangement: 9 coplanar fixed-gantry beams (angles: 200°, 240°, 280°, 320°, 0°, 40°, 80°, 120°, 160°). (Fig.1)
Optimization: Varian Eclipse™ (v16.1) with Photon Optimizer (PO) and Analytical Anisotropic Algorithm (AAA, grid size 2.5 mm).
VMAT Planning:
Arc Design: 2 full arcs (179°–181°, clockwise/counterclockwise) with dynamic MLC (Millennium 120-leaf), variable dose rate (25–600 MU/min), and gantry speed (0.5–4.8°/s). (Fig.1)
Shared Optimization Objectives:
PTV Coverage: D95% ≥ 95% of prescription dose (66 Gy).
OAR Constraints:
Spinal cord/PRV: Dmax ≤45 Gy.
Parotid glands: Dmean ≤26 Gy.
Mandible: Dmax ≤70 Gy.
Fig 1: A-9 Field IMRT Plan, B-3 full arch(VMAT Plan)
Fig 2: Color wash comparison IMRT Vs VMAT
Fig 3: DVH comparison IMRT Vs VMAT plan
Plan Evaluation
Dosimetric Parameters:
PTV Coverage: D2%, D50%, D95%, D98%.
Plan Quality:
Homogeneity Index (HI): HI= (D2%−D98%)/D50% ??.
Conformity Index (CI): (Volume covered by prescription dose) / (PTV volume)
OAR Doses: Dmean (parotids, submandibular glands), Dmax (spinal cord, mandible).
1. Treatment Efficiency:
Monitor Units (MUs) per fraction.
Statistical Analysis
Continuous variables (HI, CI, OAR doses, MUs) were compared using paired Student’s t-tests (SPSS v30.0, IBM Corp.).
Normality was confirmed via Shapiro-Wilk test.
Statistical significance: p<0.05.
Results
Target Volume Coverage
Both IMRT and VMAT provided adequate PTV coverage, with comparable mean D95% value (p = 0.12). However, VMAT demonstrated statistically significant improvements in dose homogeneity (HI: 0.0504 vs. 0.0557, p = 0.0002) and conformity (CI: 1.1659 vs. 1.1849, p = 0.0423). (Table 1)
Organ-at-Risk (OAR) Sparing
VMAT plans demonstrated superior sparing of critical structures, especially the PRV (31.39 vs. 32.58 Gy, p < 0.05), Spinal cord (27.61 vs. 28.71 Gy, p < 0.05). (Table 2)
Treatment Efficiency
VMAT significantly reduced Monitor Units compared to IMRT. (Table 3)
Table 1. Target Volume Dosimetric Parameters (mean ± SD)
|
Parameters |
IMRT |
VMAT |
p-value |
|
PTV D95% |
157.7±99.20 |
155±99.20 |
0.12 |
|
PTV Dmean |
65.2±1.91 |
66.08±0.38 |
0.08 |
|
D2% |
67.32±0.47 |
67.19±0.41 |
0.07 |
|
D98% |
63.64±0.33 |
63.8±0.60 |
0.02* |
|
Homogeneity Index (HI) |
0.05±0.006 |
0.05±0.009 |
0.002* |
|
Conformity Index (CI) |
1.18±0.09 |
1.16±0.07 |
0.04* |
*Significant at p<0.05
Table 2. OAR Dose Comparison (mean ± SD)
|
OAR |
IMRT |
VMAT |
p-value |
|
PRV cord |
32.58±2.59 |
31.39±4.37 |
0.03* |
|
Spinal Cord |
28.71±2.22 |
27.61±3.0 |
0.02* |
|
Ipsilateral Parotid |
28.40±4.39 |
28±4.08 |
0.08 |
|
Contralateral Parotid |
25.81±4.30 |
25.36±3.92 |
0.13 |
|
Parotid mean |
26.97±4.10 |
26.65±3.72 |
0.19 |
|
Parotid Dmax |
63.50±5.08 |
63.45±4.97 |
0.45 |
|
Ipsilateral SMG |
57.83±6.71 |
57.15±8.71 |
0.21 |
|
Contralateral SMG |
49.49±8.8 |
51.59±8.51 |
0.002* |
|
Mandible Dmax |
65.13±5.54 |
62.98±9.66 |
0.04* |
|
Skin Dmax |
62.81±3.57 |
65.01±4.43 |
0.04* |
|
Esophagus mean |
21.13±9.06 |
20.26±8.16 |
0.14 |
|
Lip mean |
23.49±10.65 |
23.72±9.71 |
0.32 |
|
Thyroid mean |
59.25±5.56 |
48.04±5.56 |
0.16 |
|
Pharangeal Constrictors |
46.93±6.68 |
47.1±7.06 |
0.37 |
*Significant at p<0.05
Table 3. Treatment Delivery Efficiency
|
Parameter |
IMRT |
VMAT |
p-value |
|
Monitor Units (MU mean ± SD ) |
1495.1±210.97 |
654.5±50.7 |
1.5 |
*Significant at p<0.05
Discussion
This study demonstrates that VMAT achieves superior dosimetric performance compared to IMRT in head and neck radiotherapy, offering enhanced OAR sparing, improved dose conformity/homogeneity, and greater treatment efficiency—without compromising target coverage. These advantages are particularly relevant for complex cases requiring bilateral neck irradiation or large treatment volumes, where precision and normal tissue protection are critical.
Key Dosimetric Advantages of VMAT
Improved Conformity & Homogeneity
The significantly lower Conformity Index (CI) (1.1659 vs. 1.1849, p = 0.04) reflects VMAT’s ability to tightly sculpt dose around irregular PTV geometries, minimizing exposure to adjacent healthy tissues. This precision is especially valuable in head and neck cancers, where tumors often encase critical structures [6, 7].
The better Homogeneity Index (HI) (0.0504 vs. 0.0557, p = 0.0002) indicates a more uniform PTV dose distribution, reducing hotspots linked to acute toxicities and impaired tissue repair [13, 14].
Critical OAR Sparing
Spinal Cord & PRV Cord: VMAT significantly reduced doses (spinal cord: 27.61 vs. 28.71 Gy, p = 0.02; PRV cord: 31.39 vs. 32.58 Gy, p = 0.03), potentially lowering the risk of radiation-induced myelopathy—a crucial consideration for patients receiving high cumulative doses [15, 16].
Mandible & Skin: Reductions in mandibular dose (62.13 vs. 65.13 Gy, p = 0.04) may mitigate osteoradionecrosis risk, while lower skin doses (65.01 vs. 65.81 Gy, p = 0.04) could reduce dermatitis and late fibrosis [29, 30].
Treatment Efficiency
VMAT halved monitor units (654.5 vs. 1495.1 MU, p < 0.001), shortening delivery time. This enhances patient comfort, reduces intrafraction motion errors, and improves clinic throughput [19, 20].
Clinical Implications
While large cohort studies (e.g., Algouneh et al. [24]) report comparable survival outcomes between VMAT and IMRT, the dosimetric benefits of VMAT—coupled with workflow efficiencies—suggest it may offer a superior therapeutic ratio (maximizing tumor control while minimizing toxicity). However, planner expertise and institutional protocols remain pivotal to achieving optimal results.
Limitations & Future Directions
- This study is a dosimetric analysis; clinical outcomes (e.g., toxicity rates, tumor control) were not assessed.
- Prospective trials comparing toxicity profiles, quality of life, and long-term efficacy are needed to validate these findings.
Conclusion
VMAT emerges as a technically and clinically advantageous evolution of IMRT for head and neck radiotherapy, delivering superior OAR sparing, dose conformity, and efficiency while maintaining target coverage. Its integration into modern practice aligns with the broader shift toward precision-based, patient-tailored radiotherapy. Future studies correlating these dosimetric gains with patient-reported outcomes and survival data will further solidify its role in optimizing cancer care.
References
1. Pfister DG, et al. Head and neck cancers, version 2.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2020; 18(7):873-898.
2. Ang KK, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010; 363(1):24-35.
3. Cooper JS, et al. Postoperative concurrent radiotherapy and chemotherapy for high- risk squamous-cell carcinoma of the head and neck. N Engl J Med. 2004; 350(19):1937-1944.
4. Lee N, et al. Intensity-modulated radiation therapy in head and neck cancers: an update. Head Neck. 2007; 29(4):387-400.
5. Nutting CM, et al. Parotid-sparing intensity modulated versus conventional radiotherapy in head and neck cancer (PARSPORT): a phase 3 multicentre randomized controlled trial. Lancet Oncol. 2011; 12(2):127-136.
6. Verbakel WF, et al. Volumetric intensity-modulated arc therapy vs conventional IMRT in head-and-neck cancer: a comparative planning study. Int J Radiat Oncol Biol Phys. 2009; 74(1):252-259.
7. Holt A, et al. Multi-institutional comparison of volumetric modulated arc therapy vs intensity-modulated radiation therapy for head-and-neck cancer. Radiat Oncol. 2013; 8:26.
8. Eisbruch A, et al. Preservation of salivary function with conformal and intensity- modulated irradiation in head and neck cancer. Semin Radiat Oncol. 2002; 12(1):5-13.
9. Kam MK, et al. Prospective randomized study of intensity-modulated radiotherapy on salivary gland function in early-stage nasopharyngeal carcinoma patients. J Clin Oncol. 2007; 25(31):4873-4879.
10. Chao KS, et al. A prospective study of salivary function sparing in patients with head-and-neck cancers receiving intensity-modulated or three-dimensional radiation therapy: initial results. Int J Radiat Oncol Biol Phys. 2001; 49(4):907-916.
11. Palma D, et al. Volumetric modulated arc therapy for delivery of prostate radiotherapy: comparison with intensity-modulated radiotherapy and three- dimensional conformal radiotherapy. Int J Radiat Oncol Biol Phys. 2008; 72(4):996- 1001.
12. Clivio A, et al. Volumetric-modulated arc radiotherapy for carcinomas of the oro- pharynx, hypo-pharynx and larynx: a treatment planning comparison with fixed field IMRT. Radiat Oncol. 2009; 4:22.
13. Yoo S, et al. Radiotherapy treatment planning dosimetry for head and neck cancer using IMRT and volumetric modulated arc therapy (VMAT): a comparison. J Appl Clin Med Phys. 2010; 11(3):2677.
14. Fogliata A, et al. Volumetric modulated arc therapy for head and neck cancer radiotherapy: a comprehensive treatment planning study. Radiother Oncol. 2009; 93(2):210-215.
15. Otto K. Volumetric modulated arc therapy: IMRT in a single gantry arc. Med Phys. 2008; 35(1):310-317.
16. Teoh M, et al. Volumetric modulated arc therapy: a review of current literature and clinical use in practice. Br J Radiol. 2011; 84(1007):967-996.
17. Bertelsen A, et al. Single arc volumetric modulated arc therapy of head and neck cancer. Radiother Oncol. 2010; 95(2):142-148.
18. Wolff D, et al. Volumetric modulated arc therapy (VMAT) vs. conventional IMRT techniques for head and neck cancer: results of a planning study. Radiother Oncol. 2009; 93(3):302-306.
19. Bignardi M, et al. Critical appraisal of volumetric modulated arc therapy in head and neck cancer: Clinical experience and dosimetric review. Radiother Oncol. 2012; 102(2):167-172.
20. Bertelsen A, et al. RapidArc quality assurance of head and neck plans using phantom measurements and independent dose calculation. Acta Oncol. 2010; 49(7):1052-1058.
21. Georg P, et al. VMAT vs. IMRT in head and neck cancer: dosimetric advantages and disadvantages. Radiother Oncol. 2011; 98(2):203-210.
22. Poppe B, et al. Measurement of the three-dimensional dose distribution with the 2D-Array ionization chamber system and comparison with TPS calculations. Radiother Oncol. 2006; 78(2):242-249.
23. Cozzi L, et al. Critical appraisal of volumetric modulated arc therapy in head and neck cancer: clinical experience and dosimetric review. Radiother Oncol. 2012; 102(2):167-172.
24. Algouneh A, et al. Volumetric Modulated Arc Therapy versus Conventional IMRT for Locally Advanced HNSCC: A Retrospective Cohort Study. Cureus. 2024; 16(6):e28407.
25. Nutting CM, et al. improving the therapeutic ratio in head and neck cancer with intensity-modulated radiotherapy. Br J Radiol. 2003; 76(910):693-694.
26. Edge SB, et al. AJCC Cancer Staging Manual. 8th ed. Springer; 2017.
27. ICRU Report 83. Prescribing, Recording, and Reporting Photon-Beam Intensity- Modulated Radiation Therapy (IMRT). J ICRU. 2010; 10(1):1-106.
28. Pow EH, et al. Xerostomia and quality of life after intensity-modulated radiotherapy vs. conventional radiotherapy for early-stage nasopharyngeal carcinoma: Initial report on a randomized controlled clinical trial. Int J Radiat Oncol Biol Phys. 2006; 66(4):981- 991.
29. Zhou J, Li X, Meng X, Zhang L, et al. Radiation-induced mandibular osteoradionecrosis in head and neck cancer patients: A review of pathogenesis, diagnosis, and management. Oral Oncol. 2021; 117:105292.
30. Prophylaxis and management of acute radiation-induced skin reactions: a systematic review of the literature. N Salvo et al. Curr Oncol. 2010 Aug.
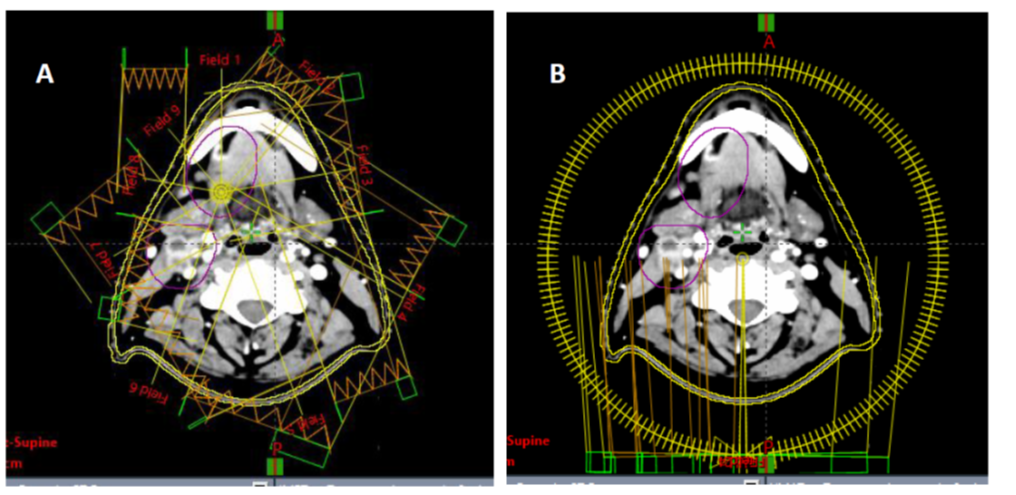
Figure 1
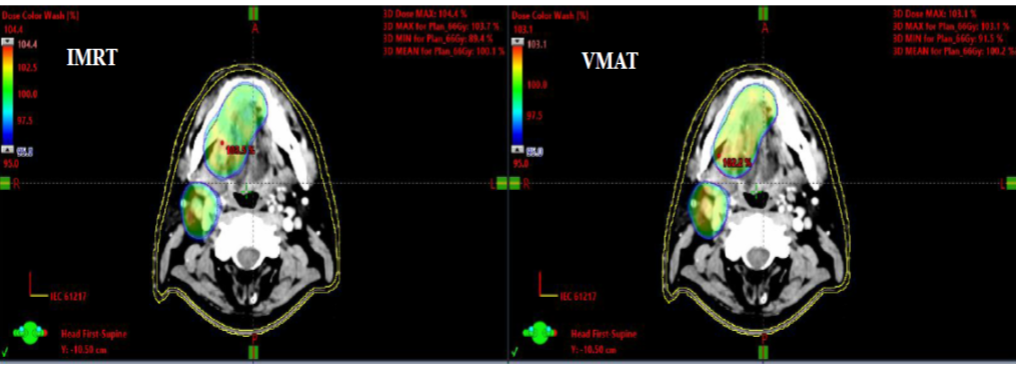
Figure 2
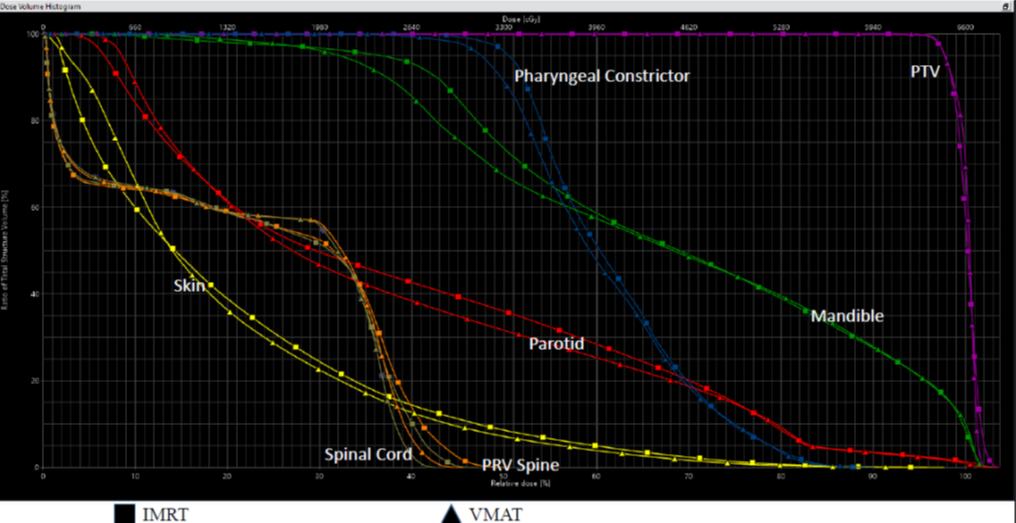
Figure 3
