Lidocaine versus Ketamine for Prevention of Postoperative Catheter Related Bladder Discomfort in Patients Undergoing Transurethral Resection of Bladder Tumor: A Randomised Controlled Trial
Lidocaine versus Ketamine for Prevention of Postoperative Catheter Related Bladder Discomfort in Patients Undergoing Transurethral Resection of Bladder Tumor: A Randomised Controlled Trial
Rumit Sood1, Anuja Pandit2*, Gitartha Goswami3, Renoxy Bansal4 , Shirali Agarwal5 , Nivisha Sood6
1,2,3,4. All India Institute of Medical Sciences, New Delhi, India.
5. Artemis Hospital , Gurgaon , Haryana
6. Maharaja Agreson sat Narain Gupta hospital, Haryana
*Correspondence to: Anuja Pandit, All India Institute of Medical Sciences, New Delhi, India.
Copyright.
© 2025 Anuja Pandit This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 02 Sep 2025
Published: 16 Sep 2025
DOI: https://doi.org/10.5281/zenodo.17284975
Abstract
Background: Male patients undergoing transurethral resections of bladder tumor (TURBT) commonly suffer from catheter related bladder discomfort (CRBD). Here, the effect of intravenous lidocaine and intravenous ketamine were evaluated and compared in male patients undergoing TURBT.
Methods: 130 male patients of age group 18-65 years were randomly allocated to receive intravenous lidocaine (1.5mg/kg bolus followed by 2 mg/kg/hr infusion) and intravenous ketamine (0.5 mg/kg bolus followed by 0.25 mg/kg/hr infusion) during the intraoperative period, which were discontinued once the patient was shifted to recovery room. The primary outcome evaluated was incidence of moderate to severe CRBD at 0 hr post surgery, analysed using the Fischer’s exact test. The secondary outcome was opioid requirement in the 24 hour postoperative period and severity of CRBD at 1, 2, 6 and 24 hrs post surgery, postoperative pain, side effects of lidocaine and ketamine, rescue medications (tramadol and fentanyl) used and any other complications were also assessed.
Results: There were no significant differences in incidence and severity of CRBD between the 2 groups. There was a significant reduction in incidence of postoperative pain at 1 hour interval in group B (1.52 ± 1.03 vs 1.17 ± 1.29; p-0.013). Opioid requirement, rescue medication use and drug related side effect did not differ significantly between the 2 groups.
Conclusion: Pre emptive administration of lidocaine reduces incidence and severity of CRBD in early postoperative period, similar to ketamine.
Keywords: catheter related bladder discomfort, ketamine, lidocaine, bladder catheterization.
Lidocaine versus Ketamine for Prevention of Postoperative Catheter Related Bladder Discomfort in Patients Undergoing Transurethral Resection of Bladder Tumor: A Randomised Controlled Trial
Introduction
Urinary Bladder catheterisation can result in feeling of discomfort, urgency and frequency. These symptoms have been referred to as catheter related bladder discomfort (CRBD) which can continue in the postoperative period. The incidence of CRBD has been reported to be 40-80% 1-5. CRBD is strongly associated with bladder catheter size more than 18 Fr, male gender and patients undergoing TURBT. 1, 6 Mild CRBD is tolerable in most of the patients but moderate to severe CRBD can be distressing in the postoperative period impacting the quality of postoperative care and may require additional analgesic therapy. Therefore appropriate management of CRBD is must to improve patient satisfaction and postoperative outcomes.
CRBD is induced by involuntary contractions of the bladder smooth muscle due to activation of the bladder muscarinic receptors or inflammatory stimulation.7 Therefore a range of agents, such as anticholinergics, ketamine, tramadol have been evaluated in various studies for the prevention or treatment of CRBD.2 Despite availability of all these agents, there is a very little evidence of their usage for the treatment without side effects.8,9
Ketamine is known to interact with multiple binding sites, including NMDA and non-NMDA glutamine receptors, mono-aminogenic and opioid receptors, nicotinic and muscarinic cholinergic receptors. Theoretically, ketamine can reduce the contractile response of the smooth muscles by an effect on the central nervous system, the preganglionic fibres, the nicotinic receptors in the postsynaptic membrane of the intramural ganglion, the postsynaptic fibres, the muscarinic receptors, the chemical signal transmission between the receptors and the contractile elements, the contractile element or any combination above. Durieux demonstrated that clinically relevant concentration of ketamine profoundly inhibit muscarinic receptor signalling.10
Whereas intravenous lidocaine has got multifactorial pharmacological effects including analgesic, anticholinergic, anti-inflammatory and antihyperalgesic properties. Based on these considerations we hypothesized that intravenous lidocaine can reduce the incidence of moderate to severe CRBD. So, in this study we compared the role of intravenous ketamine and intravenous lidocaine for prevention of CRBD in patients undergoing TURBT. The study also notes the postoperative pain, rescue medications used, side effects of both the drugs and any complications during the surgery.
Methods
This was a prospective randomised trial registered prior to patients enrolment with Clinical trial registry India ICMR-NIMS (CTRI/2022/09/045172, date of registration – 2/09/2022) and was approved by All India Institute of Medical Sciences New Delhi institutional ethical committee. 130 consenting male patients of age group 18-65 yrs, ASA grade I & II undergoing TURBT were assessed for eligibility and included in the study after taking written informed consents. Patients were excluded from the study if there was delayed extubation, admission to intensive care unit, history of opium abuse or chronic pain, patient already catheterized, history of urinary tract obstruction requiring surgical intervention, history of neurological disorder, liver, heart disease, overactive bladder, and morbid obesity. The consort’s flow chart is depicted in the study (fig 1).
Fig 1 . CONSORT flow diagram of patients included in the study. Group A comprised patients who received intravenous ketamine. Group B comprised patients who received intravenous lidocaine. CONSORT indicates Consolidated standards of reporting trials.
All the patients were educated about symptoms of CRBD a day prior to surgery. After shifting patient for surgery, NIBP, pulse oximetry, ECG, capnography, skin temperature probe, and BIS were applied to all patients. General anesthesia induction was performed with an injection of fentanyl 2mcg/kg, propofol 1.5 mg/kg, atracurium 0.5mg/kg. Once the patient was unconscious, laryngeal mask airway (LMA) was inserted and anesthesia was maintained using sevoflurane in an air/oxygen mixture and end tidal concentration of sevoflurane was adjusted to maintain a target BIS value of 40-60 and appropriate vital signs. The lidocaine and ketamine dose regime was determined based on the previous study with some modifications. In group A intravenous bolus of ketamine 0.5mg/kg just prior to induction followed by infusion of ketamine at 0.25 mg/kg/h. In group B intravenous bolus of 2% lidocaine 1.5 mg/kg just prior to induction followed by 2mg/kg/hr infusion was used. To reverse neuromuscular blockade, neostigmine 0.04 mg/kg and glycopyrrolate 8mcg/kg were administered. At the end of surgery Urinary catheter more than 20 Fr was inserted and balloon was inflated with 10 ml distilled water. A 2% lidocaine gel was used to lubricate the catheter. Normal saline was infused continuously through the urinary catheter to irrigate the bladder. After confirming that the patient is fully conscious and recovered from neuromuscular blockade, Laryngeal mask airway was removed and patient was shifted to Postoperative care unit.
In Postoperative care unit, Inj. paracetamol 1 gm was administered as rescue analgesic and inj. fentanyl was given to patients complaining of pain NRS>4 and inj. tramadol 50 mg was given to the patient complaining of moderate to severe CRBD at any point of time .If patient complained of both postoperative pain with NRS >4 and moderate-severe CRBD simultaneously then either tramadol or fentanyl was administered according to the more significant complaint. The patient was reassessed after 15 minutes. The same analgesic protocol was maintained in ward.
The severity of CRBD was recorded at 0, 1, 2, 6 and 24 hrs with none as no complaint, mild as reported by patient only when asked, moderate as reported by patients independently and severe as reported by patients with behavioral response such as flailing limbs, strong vocal response, and attempts to pull out catheter.2 Postoperative pain was also evaluated at 0, 1, 2, 6 and 24 hr with a Numerical rating scale (NRS) in which 0 is no pain and 10 is worst pain. Additionally Postoperative sedation was evaluated with Ramsay sedation scale11 along with postoperative nausea vomiting, hallucinations, diplopia, respiratory depression, cardiovascular depression and seizures. The duration of surgery and rescue medications (fentanyl or tramadol) used were also recorded. All the evaluations were recorded by blinded anaesthesiologist in post operative care unit.
Statistical analysis
The study of Reza, et al observed that incidence of CRBD at 0 hour was 38.60% and at 1 hour was 22.81% in Ketamine group. Taking these values as reference and assuming difference of 25% in incidence of CRBD between Ketamine and Lidocaine, the minimum required sample size with 80% power of study and 5% level of significance is 59 patients in each study group. To reduce margin of error, total sample size taken was 130 (65 patients per group).
Formula used was:-
n>=((pc*(1-pc)+pe*(1-pe))*(Zα + Zβ)2)/(pc-pe)2
with
pc= incidence of CRBD in Ketamine group
pe= incidence of CRBD in Lidocaine group
Where Zα is value of Z at two sided alpha error of 5% and Zβ is value of Z at power of 80% .Categorical variables were presented in number and percentage (%) and continuous variables was presented as mean ± SD and median. Normality of data was tested by Kolmogorov-Smirnov test. The data normality was checked by using Kolmogorov-Smirnov test. The cases in which the data was not normal, we used non parametric tests. The comparison of the variables which were quantitative and not normally distributed in nature were analysed using Mann-Whitney Test and variables which were quantitative and normally distributed in nature were analysed using Independent t test. The comparison of the variables which were qualitative in nature were analysed using Chi-Square test. If any cell had an expected value of less than 5 then Fisher’s exact test was used. The data entry was done in the Microsoft EXCEL spreadsheet and the final analysis was done with the use of Statistical Package for Social Sciences (SPSS) software, IBM manufacturer, Chicago, USA, version 25.0.Randomization was done with block randomization with sealed envelope system:- In this, we prepared ten randomly generated treatment allocations within sealed opaque envelopes assigning A and B in 5 envelopes each, where A represents ketamine group and B represents lidocaine group. Once a patient consented to enter a trial an envelope was opened and the patient was then offered the allocated group. Hence, patients were randomized in a series of blocks of ten.
Results
Initially 65 patients were allocated in each group but in group B there was one drop out as the patient was shifted to Intensive care unit. The 2 groups did not have significant differences in anthropometric baseline characteristics (Table 1). Duration of surgery was similar in both the groups (P>0.05; Table 1) but the groups were not similar on the basis of age (P<0.05).
In post operative care unit, incidence and severity of CRBD were similar in both the groups at 0, 1, 2, 6 and 24 hours (P>0.05; Table 2,fig 2)
There was significant reduction in incidence of pain at 1 hour interval in group B when compared to group A. (P-0.013, Table 3)
Incidence of sedation was 1.56% in group B and 0% in group A (P>0.05). Incidence of PONV was similar in both the groups. 5 patients in group A and 6 patients in group B received fentanyl for relief of pain; this difference was not statistically significant. None of the patient in either group had respiratory depression, seizures, delayed extubation and local anesthetic systemic toxicity. Hallucinations were seen in one of the patient in group A, but it resolved without any need for intervention at the 1-hour visit. (Table 4)
Table 1. Age, Anthropometric characteristics of the patients, and surgery duration between group A and B
|
|
Group A |
Group B |
P value |
|
Age (yr) (mean±SD) |
52.15 ± 9.82 |
56.03 ± 8.94 |
0.021 |
|
Weight (kg) (mean±SD) |
62.8 ± 11.04 |
62.62 ± 8.51 |
0.92 |
|
BMI (kg/m2) (mean±SD) |
23.01 ± 3.15 |
23.19 ± 2.52 |
0.73 |
|
Height (m) (mean±SD) |
1.65 ± 0.07 |
1.64 ± 0.05 |
0.52 |
|
Surgery duration (min) (mean±SD) |
57.25 ± 42.17 |
56.97 ± 36.28 |
0.81 |
Table 2. Comparison of CRBD between group A and B.
|
CRBD |
Group A(n=65) |
Group B(n=64) |
Total |
P value |
|
At 0 hour |
||||
|
None |
46 (70.77%) |
43 (67.19%) |
89 (68.99%) |
0.927 |
|
Mild |
11 (16.92%) |
11 (17.19%) |
22 (17.05%) |
|
|
Moderate |
7 (10.77%) |
9 (14.06%) |
16 (12.40%) |
|
|
Severe |
1 (1.54%) |
1 (1.56%) |
2 (1.55%) |
|
|
At 1 hour |
||||
|
None |
41 (63.08%) |
45 (70.31%) |
86 (66.67%) |
0.337 |
|
Mild |
16 (24.62%) |
15 (23.44%) |
31 (24.03%) |
|
|
Moderate |
8 (12.31%) |
3 (4.69%) |
11 (8.53%) |
|
|
Severe |
0 (0%) |
1 (1.56%) |
1 (0.78%) |
|
|
At 2 hours |
||||
|
None |
49 (75.38%) |
51 (79.69%) |
100 (77.52%) |
0.869 |
|
Mild |
14 (21.54%) |
11 (17.19%) |
25 (19.38%) |
|
|
Moderate |
2 (3.08%) |
2 (3.13%) |
4 (3.10%) |
|
|
At 6 hours |
||||
|
None |
62 (95.38%) |
63 (98.44%) |
125 (96.90%) |
0.619 |
|
Mild |
3 (4.62%) |
1 (1.56%) |
4 (3.10%) |
|
|
At 24 hours |
||||
|
None |
65 (100%) |
64 (100%) |
129 (100%) |
NA |
Table 3. Comparison of pain between group A and B.
|
Pain |
Group A(n=65) |
Group B(n=64) |
Total |
P value |
|
At 0 hour |
||||
|
Mean ± SD |
1.77 ± 1.13 |
2.05 ± 1.1 |
1.91 ± 1.12 |
0.193 |
|
Median(25th-75th percentile) |
2(1-2) |
2(1-2) |
2(1-2) |
|
|
Range |
0-6 |
1-7 |
0-7 |
|
|
At 1 hour |
||||
|
Mean ± SD |
1.52 ± 1.03 |
1.17 ± 1.29 |
1.35 ± 1.18 |
0.013 |
|
Median(25th-75th percentile) |
2(1-2) |
1(0-2) |
1(0-2) |
|
|
Range |
0-5 |
0-5 |
0-5 |
|
|
At 2 hours |
||||
|
Mean ± SD |
1.06 ± 1.04 |
0.83 ± 1.05 |
0.95 ± 1.05 |
0.064 |
|
Median(25th-75th percentile) |
1(0-1) |
1(0-1) |
1(0-1) |
|
|
Range |
0-5 |
0-5 |
0-5 |
|
|
At 6 hours |
||||
|
Mean ± SD |
0.34 ± 0.57 |
0.3 ± 0.46 |
0.32 ± 0.52 |
0.91 |
|
Median(25th-75th percentile) |
0(0-1) |
0(0-1) |
0(0-1) |
|
|
Range |
0-2 |
0-1 |
0-2 |
|
|
At 24 hours |
||||
|
Mean ± SD |
0.06 ± 0.24 |
0.12 ± 0.33 |
0.09 ± 0.29 |
0.217 |
|
Median(25th-75th percentile) |
0(0-0) |
0(0-0) |
0(0-0) |
|
|
Range |
0-1 |
0-1 |
0-1 |
|
Table 4. Comparison of adverse effects between group A and B.
|
Adverse effects |
Group A(n=65) |
Group B(n=64) |
Total |
P value |
|
Post-operative nausea and vomiting |
1 (1.54%) |
0 (0%) |
1 (0.78%) |
1 |
|
Agitation |
1 (1.54%) |
2 (3.13%) |
3 (2.33%) |
0.619 |
|
Hallucination |
1 (1.54%) |
0 (0%) |
1 (0.78%) |
1 |
|
Respiratory depression |
0 (0%) |
0 (0%) |
0 (0%) |
NA |
|
Delayed extubation |
0 (0%) |
0 (0%) |
0 (0%) |
NA |
|
Hypotension/CVS depression |
0 (0%) |
2 (3.13%) |
2 (1.55%) |
0.244 |
|
Seizures |
0 (0%) |
0 (0%) |
0 (0%) |
NA |
|
LAST |
0 (0%) |
0 (0%) |
0 (0%) |
NA |
Fig 2. Comparison of catheter related bladder discomfort at 0, 1, 2, 6 and 24 hours between group A and B
Discussion
Patients undergoing intra operative urinary catheterisation often complains of CRBD. These symptoms are very similar to symptoms of overactive bladder which might be due to involuntary contractions of bladder wall smooth muscles mediated by muscarinic receptors.9
Durieux and Rainer suggested that clinically relevant concentrations of ketamine profoundly inhibit muscarinic receptor signaling.10, 11 They also showed ketamine’s effect as evidenced by reduced incidence and severity of CRBD. Several studies support the preemptive analgesic effects of ketamine while some show no benefit.12-16
Another mechanism underlying the development of CRBD is local inflammation with release of prostaglandins due to mucosal damage during urinary catheterisation.17 Lidocaine due to its action on sodium channel, muscarinic and NMDA receptors and nociceptive transmission pathways has significant antimuscarinic and antiinflammatory properties. 18
As evidenced by the results of our study lidocaine reduced the incidence of moderate to severe CRBD to the similar extent as ketamine. Also, there was no evidence of lidocaine related side effects. Previous studies have shown that lidocaine has a significant inhibitory effect on muscarinic receptors, along with suppression of immune cell–mediated inflammatory reactions.19, 20 In addition, lidocaine has shown antihyperalgesic effects in patiens undergoing laparoscopic nephrectomy.21, 22
In this study it is possible that reduced severity of CRBD and pain in lignocaine group were due to the reduction of urinary catheter related inflammation, muscarinic and hyperalgesic responses. Intravenous lignocaine can cause neuro and cardiac adverse effects when plasma concentration exceeds more than 5mcg/ml. However, intravenous lidocaine when administered at the standard doses (1–2 mg/kg as an initial bolus, followed by a continuous infusion of 0.5–3 mg/kg/h) generally results in plasma concentrations that remains below 5 mcg/ml.23, 24 In this study there was no significant side effects seen in lidocaine group suggesting that lidocaine was well tolerated by patients undergoing TURBT.
Male gender and large urinary catheters are known risk factors for the development of CRBD.1, 8
TURBT destroys the normal barrier mechanism of the urinary bladder wall, and continuous irrigation of the urinary bladder triggers urinary bladder spasms. Also, patients undergoing TURBT requires large diameter urinary catheters resulting in a higher degree of CRBD severity in comparison with those undergoing other types of urological and nonurological surgery.8 In this study the incidence of moderate to severe CRBD in lidocaine group is 24.6% which is lower than that reported in previous studies.
There are some limitations to the current study. First, plasma lidocaine concentrations were not measured in this study but taking into consideration that there was no significant side effects reported in any group and there are many studies which had already demonstrated the safety profile of intravenous ketamine and lidocaine use, 25-28 we suggest that the doses used in current study are safe and appropriate for the management of CRBD. Second, no standardized tool was used for the assessment of postoperative delirium hence it was difficult to distinguish between severe CRBD with behavioral responses and postoperative delirium. In future studies, a verified tool must be required to accurately assess postoperative delirium and distinguish it from severe CRBD.
In conclusion, intravenous lidocaine and ketamine both were seen to effectively reduce the incidence of moderate-to-severe CRBD in male patients undergoing TURBT and there were no evidence of significant side effects. These data suggest that both the drugs may be an effective option to decrease the incidence of moderate-to-severe CRBD in male patients undergoing TURBT who require a large diameter urinary catheter.
References
1. Binhas M, Motamed C, Hawajri N, Yiou R, Marty J. Predictors of bladder related discomfort in the post-anesthesia care unit. Ann Fr Anesth Reanim. 2011;30:122–125.
2. Park JY, Hong JH, Yu J, et al. Effect of ketorolac on the prevention of postoperative catheter-related bladder discomfort in patients undergoing robot-assisted laparoscopic radical prostatectomy: a randomized, double-blinded, placebo-controlled Study. J Clin Med. 2019;8:E759.
3. Agarwal A, Gupta D, Kumar M, Dhiraaj S, Tandon M, Singh PK. Ketamine for treatment of catheter related bladder discomfort: a prospective, randomized, placebo controlled and double blind study. Br J Anaesth. 2006;96:587–589
4. Li S, Song L, Ma Y, Lin X. Tramadol for the treatment of catheter-related bladder discomfort: a randomized controlled trial. BMC Anesthesiol. 2018;18:194.
5. Yun YZ, Mei XJ, Rong Z, Ping DR, Lei L. Dexmedetomidine reduces catheter-related bladder discomfort: a prospective, randomized, placebo-controlled, double-blind study. P R Health Sci J. 2016;35:191–196.
6. Kim HC, Lee YH, Jeon YT, et al. The effect of intraoperative dexmedetomidine on postoperative catheter-related bladder discomfort in patients undergoing transurethral bladder tumour resection: a double-blind randomised study. Eur J Anaesthesiol. 2015;32:596–601.
7. Zhang Z, Cao Z, Xu C, et al. Solifenacin is able to improve the irritative symptoms after transurethral resection of bladder tumors. Urology. 2014;84:117–121.
8. Bai Y, Wang X, Li X, et al. Management of catheter-related bladder discomfort in patients who underwent elective surgery. J Endourol. 2015;29:640–649
9. Agarwal A, Raza M, Singhal V, et al. The efficacy of tolterodine for prevention of catheter-related bladder discomfort: a prospective, randomized, placebo-controlled, doubleblind study. Anesth Analg. 2005;101:1065–1067.
10. Dawson R, von Fintel N, Nairn S. Sedation assessment using the Ramsay scale. Emergency Nurse. 2010 Jun 9;18(3).
11. Durieux ME. Inhibition by ketamine of muscarinic acetylcholine receptor function. Anesth Analg. 1995;81:57–62.
12. Kohrs R, Durieux ME. Teaching an old drug new tricks. Anesth Analg. 1998;87:1186–1193
13. . Launo C, Bassi C, Spagnolo L, et al. Preemptive ketamine during general anesthesia for postoperative analgesia in patients undergoing laparoscopic cholecystectomy. Minerva Anestesiol 2004; 70: 727–734; 734–738. 13.
14. Aida S, Yamakura T, Baba H, Taga K, Fukuda S, Shimoji K. Preemptive analgesia by intravenous low-dose ketamine and epidural morphine in gastrectomy: a randomized, double-blind study. Anesth Analg. 2000;92:1624–1630. 14.
15. Fu Es, Miguel R, Scharf JE. Preemptive Ketamine decreases postoperative narcotic requirement in patients undergoing abdominal surgery. Anesth Analg. 1997;84: 1086–1090.
16. Becke K, Albrecht S, Schmitz B, et al. Intraoperative low dose S – Ketamine Has no preventive effects on postoperative pain and morphine consumption after major urological surgery in children. Peadiatr Anesth. 2005;15:484–490.
17. Juksch W, Lang S, Reichhalter R, Raab G. Dannk, fitzal S. Perioperative small dose S (+) ketamine has no incremental beneficial effects on postoperative pain when standard practice opioid infusion are used. Anesth Analg. 2002;94:981–986
18. Ergenoglu P, Akin S, Yalcin Cok O, et al. Effect of intraoperative paracetamol on catheter-related bladder discomfort: a prospective, randomized, double-blind study. Curr Ther Res Clin Exp. 2012;73:186–194
19. . Lauretti GR. Mechanisms of analgesia of intravenous lidocaine. Rev Bras Anestesiol. 2008
20. Hollmann MW, Ritter CH, Henle P, de Klaver M, Kamatchi GL, Durieux ME. Inhibition of m3 muscarinic acetylcholine receptors by local anaesthetics. Br J Pharmacol. 2001;133:207–216.
21. Hollmann MW, Herroeder S, Kurz KS, et al. Time-dependent inhibition of G protein-coupled receptor signaling by local anesthetics. Anesthesiology. 2004;100:852–860.
22. Tauzin-Fin P, Bernard O, Sesay M, et al. Benefits of intravenous lidocaine on post-operative pain and acute rehabilitation after laparoscopic nephrectomy. J Anaesthesiol Clin Pharmacol. 2014;30:366–372.
23. Koppert W, Ostermeier N, Sittl R, Weidner C, Schmelz M. Low-dose lidocaine reduces secondary hyperalgesia by a central mode of action. Pain. 2000;85:217–224
24. Eipe N, Gupta S, Penning JJBE. Intravenous lidocaine for acute pain: an evidence-based clinical update. BJA Educ. 2016;16:292–298.
25. McCarthy GC, Megalla SA, Habib AS. Impact of intravenous lidocaine infusion on postoperative analgesia and recovery from surgery: a systematic review of randomized controlled trials. Drugs. 2010;70:1149–1163.
26. Grigoras A, Lee P, Sattar F, Shorten G. Perioperative intravenous lidocaine decreases the incidence of persistent pain after breast surgery. Clin J Pain. 2012;28:567–572
27. Baral BK, Bhattarai BK, Rahman TR, Singh SN, Regmi R. Perioperative intravenous lidocaine infusion on postoperative pain relief in patients undergoing upper abdominal surgery. Nepal Med Coll J. 2010;12:215–220
28. Kaba A, Laurent SR, Detroz BJ, et al. Intravenous lidocaine infusion facilitates acute rehabilitation after laparoscopic colectomy. Anesthesiology. 2007;106:11–18.
29. Wu CT, Borel CO, Lee MS, et al. The interaction effect of perioperative cotreatment with dextromethorphan and intravenous lidocaine on pain relief and recovery of bowel function after laparoscopic cholecystectomy. Anesth Analg. 2005;100:448–453.
Figure 1
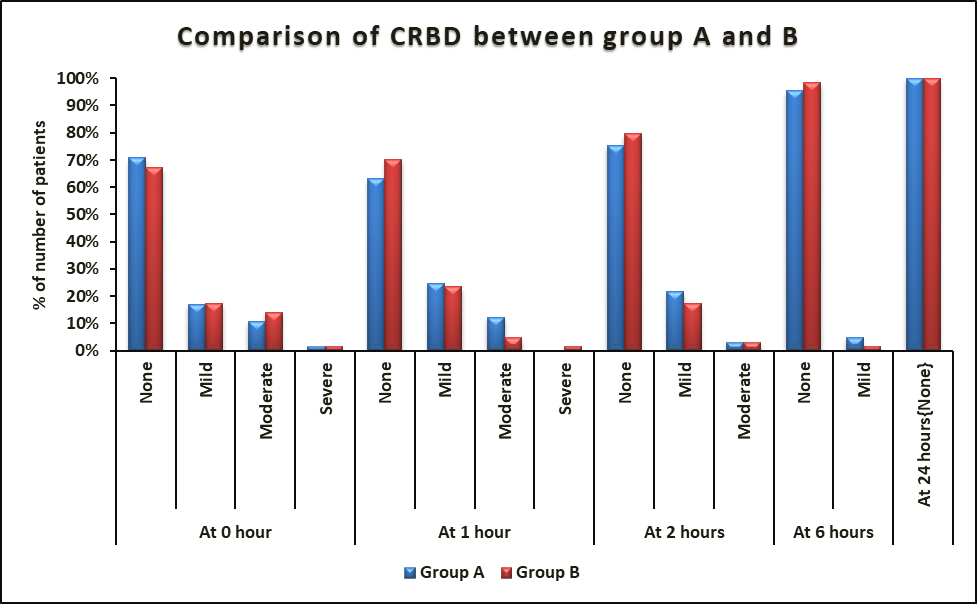
Figure 2
