“From Biomarkers to Risk Scores: Maternal First Trimester Dual Marker Screening, Risk Stratification for Trisomy 21/18/13”.
“From Biomarkers to Risk Scores: Maternal First Trimester Dual Marker Screening, Risk Stratification for Trisomy 21/18/13”.
Dr. Dheepa Manoharan *
*Correspondence to: Dr. Dheepa Manoharan, MBBS MD Medical Director, Specialist Microbiologist, QLABS Clinical Laboratory, Dubai.
Copyright.
© 2025 Dr. Dheepa Manoharan This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 09 Sep 2025
Published: 30 Sep 2025
“From Biomarkers to Risk Scores: Maternal First Trimester Dual Marker Screening, Risk Stratification for Trisomy 21/18/13”.
Background
Prenatal screening plays a vital role in identifying pregnancies at increased risk for chromosomal conditions such as Down’s syndrome (Trisomy 21), Edwards syndrome (Trisomy 18), and Patau syndrome (Trisomy 13). First trimester screening combines maternal serum biomarkers—free β-Human Chorionic Gonadotropin (β-HCG) and Pregnancy Associated Plasma Protein A (PAPP-A)—with ultrasound parameters like Nuchal Translucency (NT) and Crown Rump Length (CRL). These inputs, when analyzed using validated software such as SSDW, allow for early, non-invasive risk stratification. By integrating biochemical and sonographic data, this approach supports timely clinical decisions and minimizes the need for invasive diagnostic procedures.
Study Place: QLabs Clinical Laboratory, Dubai
Study Period: March–September 2025
Methods
- This retrospective study was conducted at QLabs over a 6-month period (March–September 2025), involving 189 pregnant women screened exclusively in the first trimester using SSDW software.
Figure 1
Cohort Overview
- Total Patients Screened: 189
- Study Duration: 6-month retrospective analysis
- Mean Age: 30.99 years (Reproductive age group)
- Mean Weight: 61.9 kg (Used for MoM normalization)
- Gestational Age at Screening: Median 88 days
- Trimester Distribution: 100% First Trimester
- Non-Caucasian patients: 1.6% (ethnic adjustment applied)
- No history of chromosomal anomalies, smoking, or diabetes
- Biochemical markers were expressed in Multiples of Median (MoM) and interpreted using SSDW algorithms. NT measurements were included in risk calculation. The SSDW cut-off for high risk was set at 1:250 for Trisomy 21, 18, and 13.
Figure 2
Clinical Background
- Previous Anomalies: None (No prior chromosomal abnormalities)
- Smokers / Diabetics: None (No confounding risk factors)
- Non-Caucasian Patients: 1.6% (Ethnic adjustment applied in SSDW)
Figure 3
Figure 4
Figure 5
Figure 6
Figure 7
Conclusion
Out of 189 patients screened, 3.72% were flagged as high-risk for Trisomy 21 based on SSDW analysis. Elevated β-HCG and reduced PAPP-A levels were consistent with known biochemical trends in affected pregnancies. NT measurements showed borderline elevation in flagged cases.
This study reinforces the value of first trimester double marker screening combined with ultrasound and SSDW analytics in identifying high-risk pregnancies early—supporting timely clinical decisions and reducing reliance on invasive testing.
Figure 8
Acknowledgments
Gratitude to our technical and entire QLABS CLINICAL LABORATORY team for their dedication and precision. This work reflects our shared commitment to excellence, innovation, and patient-centered care, and continuous improvement. Special appreciation to ROCHE DIGNOSTICS and data handling team of SSDW software.
“Empowering early detection through data-driven screening”.
Disclaimers
- This poster presents retrospective screening data analyzed using SSDW software and is intended for educational and audit purposes.
- Risk estimates are probabilistic and should be interpreted in conjunction with clinical findings, ultrasound parameters, and patient history.
- Biochemical values are expressed in Multiples of Median (MoM), normalized for maternal weight, gestational age, and ethnicity.
- Screening outcomes do not confirm diagnosis and should be followed by appropriate clinical evaluation and, where indicated, confirmatory testing.
Ethics Statement
This retrospective analysis was conducted using anonymized patient data in accordance with institutional protocols. No patient-identifiable information was used, and all procedures adhered to local regulation standards for ethical data reporting and laboratory practice.
References
1. Nicolaides KH. Screening for fetal chromosomal abnormalities. Best Pract Res Clin Obstet Gynaecol. 2012;26(1):3–13.
2. Spencer K et al. First trimester screening for trisomy 21: a review of biochemical and ultrasound markers. Prenat Diagn. 2000;20(6):523–529.
3. SSDW Software User Manual, Versions 5 & 6 – Risk Calculation Algorithms and MoM Normalization Protocols.
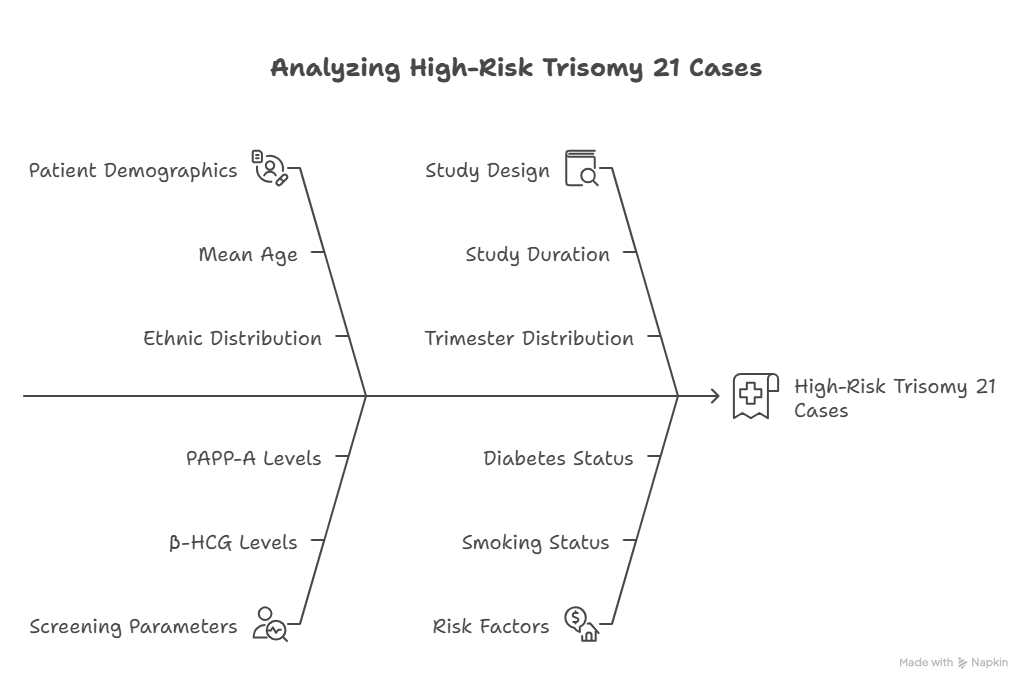
Figure 1
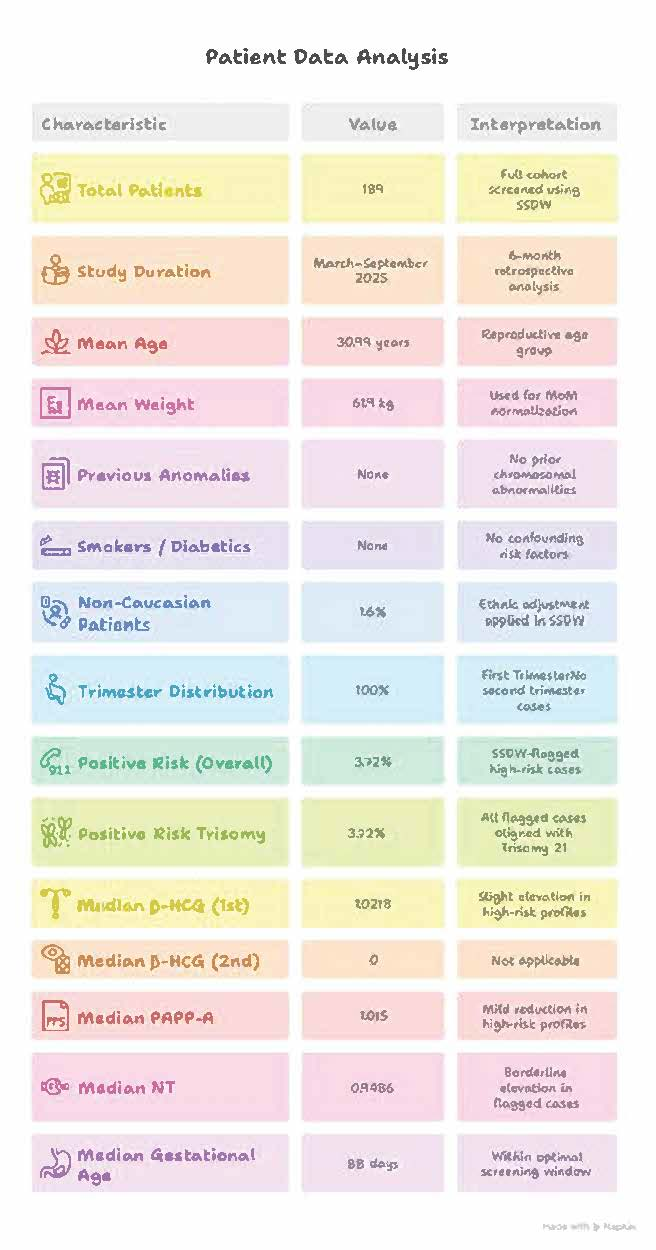
Figure 2
Figure 3
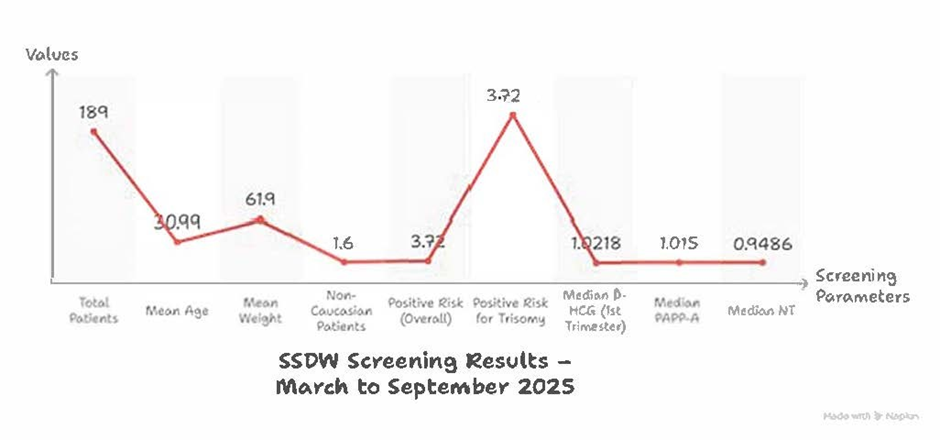
Figure 4
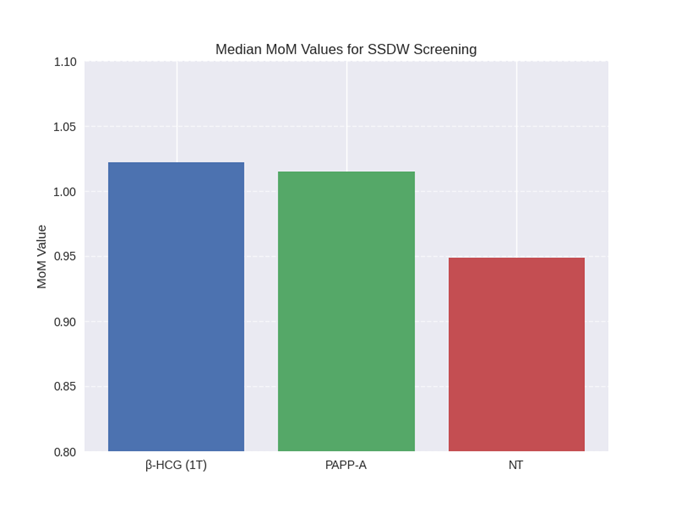
Figure 5
Figure 6
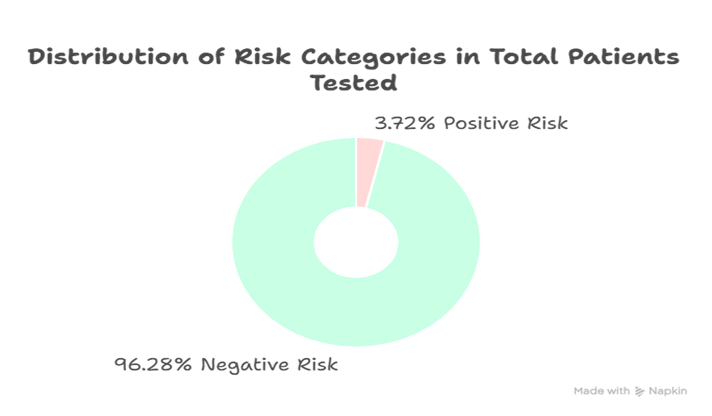
Figure 7

Figure 8
