Cerebral Venous Sinus Thrombosis during Chemotherapy with Capecitabine: Case Presentation and Strategy for Anticoagulation.
Cerebral Venous Sinus Thrombosis during Chemotherapy with Capecitabine: Case Presentation and Strategy for Anticoagulation.
Jorge Illarramendi1, Maialen Imizcoz2, Julia Coll3, Alvaro Gargallo2, Carmen Bacaicoa2, Jose Juan Illarramendi4*
1. Hematology and Cellular Therapy Department. Haut-Lévêque Hospital. Pessac, France.
2. Radiology Service. Hospital Universitario de Navarra. Pamplona, ??Spain.
3. Hematology Service. Hospital Universitario de Navarra. Pamplona, ??Spain.
4. Medical Oncology Service. Hospital Universitario de Navarra. Pamplona, ??Spain.
*Correspondence to: Jose Juan Illarramendi. Medical Oncology Service, Hospital Universitario de Navarra. Pamplona, Spain.
Copyright.
© 2025 Jose Juan Illarramendi., This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 29 Sep 2025
Published: 09 Oct 2025
DOI: https://doi.org/10.5281/zenodo.17404120
Abstract
Cerebral venous sinus thromboses (CVST) are rare but may be associated to life-threatening prognosis and prolonged disability. We report a case of CVST during chemotherapy with capecitabine in a patient with metastatic breast cancer without any additional risk factors. She presented with headache, depressed consciousness and sensorial aphasia. Several cerebral venous sinuses were involved, with associated deep venous extension, brain infarcts and secondary parenchymal bleeding. Evolution after anticoagulation with heparin during the acute phase and long-term rivaroxaban has been favorable, with full neurologic recovery and absence of thrombotic relapses and bleedings. Furthermore, chemotherapy with capecitabine was restarted and is maintained so far, with a durable response and no additional complications.
Keywords: Cerebral venous sinus thrombosis. Breast cancer. Chemotherapy. Capecitabine.
Cerebral Venous Sinus Thrombosis during Chemotherapy with Capecitabine: Case Presentation and Strategy for Anticoagulation.
Abbrevations:
BC: Breast cancer
CPB: Capecitabine
CT: Computed tomography
CVST: Cerebral venous sinus thrombosis
DOAC: Direct oral anticoagulant
MRI: Magnetic resonance imaging
VTD: Venous thromboembolic disease
Background
Cerebral venous sinus thrombosis (CVST) is an unusual site of venous thromboembolic disease (VTD) with a described incidence of 1.32 to 1.57 per 100,000 person-years, lower median age of appearance as compared to other sites of VTD, female predominance, and active cancer among the main risk factors (1). Several intracranial veins and sinuses may be affected, with some challenges for a rapid diagnosis, potential cerebral complications and recognized radiological signs for imaging evaluation (2). We present herein the case of a patient who developed CSVT during the treatment of metastatic breast cancer (BC) with capecitabine (CPB).
Case Presentation
A 62-year woman was admitted in May 2023 for loss of consciousness with sensory aphasia.
The patient was a non-smoker, without any risk factors for cardiovascular disease, no history of VTD, previous pregnancies and long-term use of estrogen-containing contraceptives without complications.
She had underwent a left breast segmentectomy plus axillary lymphadenectomy for a diagnosis of BC in September 2012. Histopathology disclosed a grade 3 invasive BC, pathologic stage pT1N2M0 and luminal phenotype, with positive hormone receptors and lack of expression of human epidermal receptor 2. Adjuvant chemotherapy with docetaxel, doxorubicin and cyclophosphamide was given afterwards, followed by irradiation and hormone therapy with letrozole for 5 years.
Follow-up was uneventful until May 2021, when a diagnosis of widespread bone and lung metastases was done. She started first-line therapy with palbociclib plus letrozole, that was changed to chemotherapy with CPB in June 2022, after findings of progressive metastatic disease. Initial tolerance to CPB was fair and there were early signs of tumor response.
While on treatment with CPB, she was admitted in May 2023 for loss of consciousness, and sensory aphasia. A severe headache, refractory to paracetamol, was noted by the patient during the previous week. Neurological exploration was relevant for somnolence, progressive aphasia and incomplete claudication of her right arm.
Blood parameters including coagulation, chest X-ray and electrocardiogram were normal. An initial brain CT scan revealed a right occipital hematoma with subdural involvement.
The patient was translated to the intensive care unit, as there was deterioration of her neurologic and clinical status. A second brain CT scan gave similar results, but a CT imaging revaluation disclosed bilateral thromboses in the transverse and sigmoid sinuses, as well as in the straight sinus and a small right occipital parenchymal hematoma. Sclerotic calvarial bone metastases were also present. Anticoagulant therapy with sodium heparin was initially started, and continued with therapeutic doses of enoxaparin during the in-hospital period.
A brain MRI study displayed an extensive CVST, with main involvement of the right transverse and sigmoid sinuses (Figure 1), and extension to the straight sinus and vein of Galen. There were infarction signs in the thalamus (Figure 2) and in the right occipital region, with associated bleeding in this zone (Figure 3).
Evolution of the patient was favorable, and she was discharged after 15 days of hospitalization. Anticoagulation was changed to tinzaparin in June 2023 and to rivaroxaban (20 mg/day) in September 2023.
Outpatient chemotherapy with CPB was resumed with good tolerance and persisting signs of tumor response. She continues on rivaroxaban and started the 55th cycle of CPB in September 2025. Neurologic recovery has been complete and there has been no new episodes of VTD or bleedings.
Fig. 1 MRI T1-weighted images with contrast. Filling defects in the transverse and sigmoid venous sinuses (green arrows, axial view). Lineal filling defect in the straight venous sinus (red arrows, sagittal view).
Fig. 2 MRI FLAIR sequence showing bilateral thalamic hyperintensity related to parenchymal infarcts (yellow arrows, coronal view).
Fig. 3 MRI T1-weighted image showing signal disturbance in the right occipital lobe with hyperintense peripheral halo, corresponding to hemorrhagic infarct (blue asterisk, axial view).
Discussion
Active cancer is among the risk factor for CVST. Data from a case-control study suggested that a history of cancer increased the risk of CVT approximately 5-fold and that this association was strongest with hematological neoplasms and in the first year after cancer diagnosis (3). Large studies, as the ACTION-VT cohort, suggest than CSVT in cancer patients is significantly related to older age, absence of headache and a higher risk of recurrent VTD or major hemorrhage, as compared to patients without cancer (4). A recent systematic review and meta-analysis has also confirmed a poorer functional outcome for cancer patients (5).
CVST is uncommon in patients with BC. In a large study from the US National Inpatient Sample (2016-2019), only 40/6,140 patients with CVST had a diagnosis of BC (6). There is one published case of CVST during the treatment with CPB for metastatic BC (7). The patient had multiple bone lesions as the only sign of metastatic disease, was in the second month of therapy with CPB and was also on treatment with denosumab. She had CVST of the left sigmoid sinus, with associated thrombosis of the left upper internal jugular vein, and received therapy with low-molecular-weight heparin followed by warfarin. Posterior reversible encephalopathy syndrome (PRES) may have a similar or overlapping symptomatology to CVST, and has been associated to the use of chemotherapy with CPB in some instances (8).
Anticoagulation is the main therapy for CSVT. Classical guidelines recommended the use of low- molecular-weight heparins during the acute phase (9), and recent guides suggest the use of direct oral anticoagulants (DOAC) instead of vitamin K antagonists after the acute phase (10). Cancer patients were not included in the experimental arm of pivotal randomized trials on the use of DOAC in CVST, like the SECRET (11) and RE-SPECT CVT (12) studies. The ACTION-CVT trial randomized patients to DOAC versus warfarin in this setting but excluded the participation of cancer patients on the basis that DOAC were already considered as better options for those patients (13). In contrast, oncologic patients comprised one third of those treated with DOAC for CVST in a large real-world study based on the TriNetX database (14) with better results than a matched warfarin arm, especially for patients treated with rivaroxaban.
Some authors suggest that patients with CVST related to cancer have a narrow therapeutic index of anticoagulation, and that careful monitoring of anticoagulation effect and bleeding complications are of utmost clinical relevance in cancer patients (15). Pharmacokinetic interactions between capecitabine and warfarin may be problematic in this context (16). In contrast, DOAC are devoid of such interactions and considered safer for this reason in these patients (17).
Conclusion
CVST is an unusual but dreaded complication during antineoplastic chemotherapy. There are some findings that are more common in cancer patients and must be considered for the strategy of evaluation and care in this context. Anticoagulation with low-molecular-weight heparin during the acute phase and long-term maintenance with direct oral anticoagulants is the preferred therapy. Good functional outcomes and absence of thrombotic relapses and major bleeding may be accomplished even in patients with negative prognostic factors, as is illustrated by our case report.
Acknowledgements: To all the health worker implicated in this case and to the support of her family.
Conflicts of interest: None declared.
References
1. Zuo W, Chen S, Lin J, Wan Y, Cao F, Hu B. Cerebral Venous Thrombosis: Current Status and Challenges. Brain Behav. 2025 Sep;15(9):e70844. doi: 10.1002/brb3.70844.
2. Carletti F, Vilela P, Jäger HR. Imaging Approach to Venous Sinus Thrombosis. Radiol Clin North Am. 2023; 61(3): 501-519. doi: 10.1016/j.rcl.2023.01.011.
3. Silvis SM, Hiltunen S, Lindgren E, Jood K, Zuurbier SM, Middeldorp S. Cancer and risk of cerebral venous thrombosis: a case-control study. J Thromb Haemost. 2018 ;16(1): 90-95. doi: 10.1111/jth.13903.
4. Vedovati MC, Shu L, Henninger N, Zubair AS, Heldner MR, Al Kasab S, et al. Cerebral venous sinus thrombosis associated with cancer: analysis of the ACTION-CVT study. J Thromb Thrombolysis. 2024; 57(6):1008-1017. doi: 10.1007/s11239-024-02997-w.
5. Lin L, Liu S, Wang W, He XK, Romli MH, Rajen Durai R. Key prognostic risk factors linked to poor functional outcomes in cerebral venous sinus thrombosis: a systematic review and meta-analysis. BMC Neurol. 2025 Feb 6;25(1):52. doi: 10.1186/s12883-025-04059-x.
6. Vazqez S, Das A, Spirollari E, Brabant P, Nolan B, Clare K, et al. Inpatient Outcomes of Cerebral Venous Thrombosis in Patients With Malignancy Throughout the United States. J Stroke. 2024 ;26(3): 425-433. doi: 10.5853/jos.2023.04098.
7. Singhal A, Singh G, Thakur B, Srivastava A. Cerebral Venous Thrombosis in Patient with Metastatic
Carcinoma Breast: Mimicking Brain Metastasis-a Case Report and Review of Literature. Indian J Surg Oncol. 2020;11(Suppl 2):244-246. doi:10.1007/s13193-020-01101-x
8. Dirani K, Marusca G, Wang J, Juzych D. Capecitabine-induced posterior reversible encephalopathy syndrome (PRES) in a patient with gastric adenocarcinoma. BMJ Case Rep. 2023 Nov 30;16(11):e252059. doi: 10.1136/bcr-2022-252059.
9. Ferro JM, Bousser MG, Canhão P, Coutinho JM, Crassard I, Dentali F, et al. European Stroke Organization guideline for the diagnosis and treatment of cerebral venous thrombosis - Endorsed by the European Academy of Neurology. Eur Stroke J. 2017; 2(3):195-221. doi: 10.1177/2396987317719364.
10.Weimar C, Beyer-Westendorf J, Bohmann FO, Hahn G, Halimeh S, Holzhauer S, et al. New recommendations on cerebral venous and dural sinus thrombosis from the German consensus-based (S2k) guideline. Neurol Res Pract. 2024 Apr 19;6(1):23. doi: 10.1186/s42466-024-00320-9.
11. Field TS, Dizonno V, Almekhlafi MA, Bala F, Alhabli I, Wong H, et al. Study of Rivaroxaban for Cerebral Venous Thrombosis: A Randomized Controlled Feasibility Trial Comparing Anticoagulation With Rivaroxaban to Standard-of-Care in Symptomatic Cerebral Venous Thrombosis. Stroke. 2023 ;54(11):2724-2736. doi: 10.1161/STROKEAHA.123.044113.
12. Ferro JM, Coutinho JM, Dentali F, et al. Safety and Efficacy of Dabigatran Etexilate vs Dose-Adjusted Warfarin in Patients With Cerebral Venous Thrombosis: A Randomized Clinical Trial. JAMA Neurol. 2019;76(12):1457-1465. doi:10.1001/jamaneurol.2019.2764
13.Yaghi S, Shu L, Bakradze E, Salehi Omran S, Giles JA, Amar JY, et al. Direct Oral Anticoagulants Versus Warfarin in the Treatment of Cerebral Venous Thrombosis (ACTION-CVT): A Multicenter International Study. Stroke. 2022 ;53 (3):728-738. doi: 10.1161/STROKEAHA.121.037541.
14.Wu JY, Lai CC, Lin HJ, Lu KH, Hsu WH, Chu TY. Comparing the effectiveness and safety of direct oral anticoagulants and warfarin in patients with cerebral venous thrombosis: A real-world study. J Stroke Cerebrovasc Dis. 2025 Jun;34(6):108290. doi: 10.1016/j.jstrokecerebrovasdis.2025.108290.
15. Abelhad NI, Qiao W, Garg N, Rojas-Hernandez CM. Thrombosis and bleeding outcomes in the treatment of cerebral venous thrombosis in cancer. Thromb J. 2021 Jun 1;19(1):37. doi: 10.1186/s12959-021-00292-9.
16. Althiab K, Aljohani M, Alraddadi S, Algarni M. Capecitabine and Warfarin Interaction: A Case Report
With Review of Literature and Management Options. Front Cardiovasc Med. 2022 Jan 31;8:707361. doi:
10.3389/fcvm.2021.707361.
17. Nowinski K, Chaireti R. Discrepancies in Recommendations on Pharmacokinetic Drug Interactions for Anticancer Medications and Direct Oral Anticoagulants (DOAC): A Comparative Analysis of Different Clinical Decision Support Systems and Sources. Pharmaceuticals (Basel). 2025 Jul 16;18(7):1044. doi:
10.3390/ph18071044.
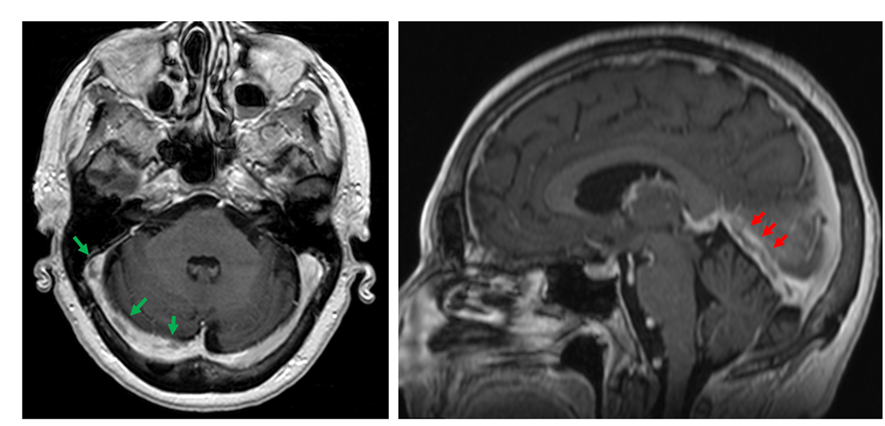
Figure 1
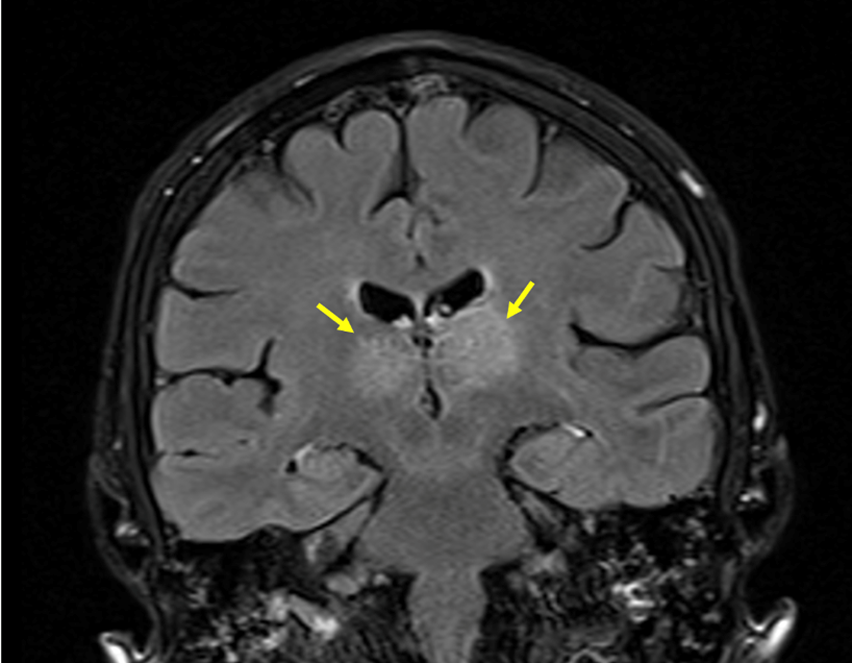
Figure 2
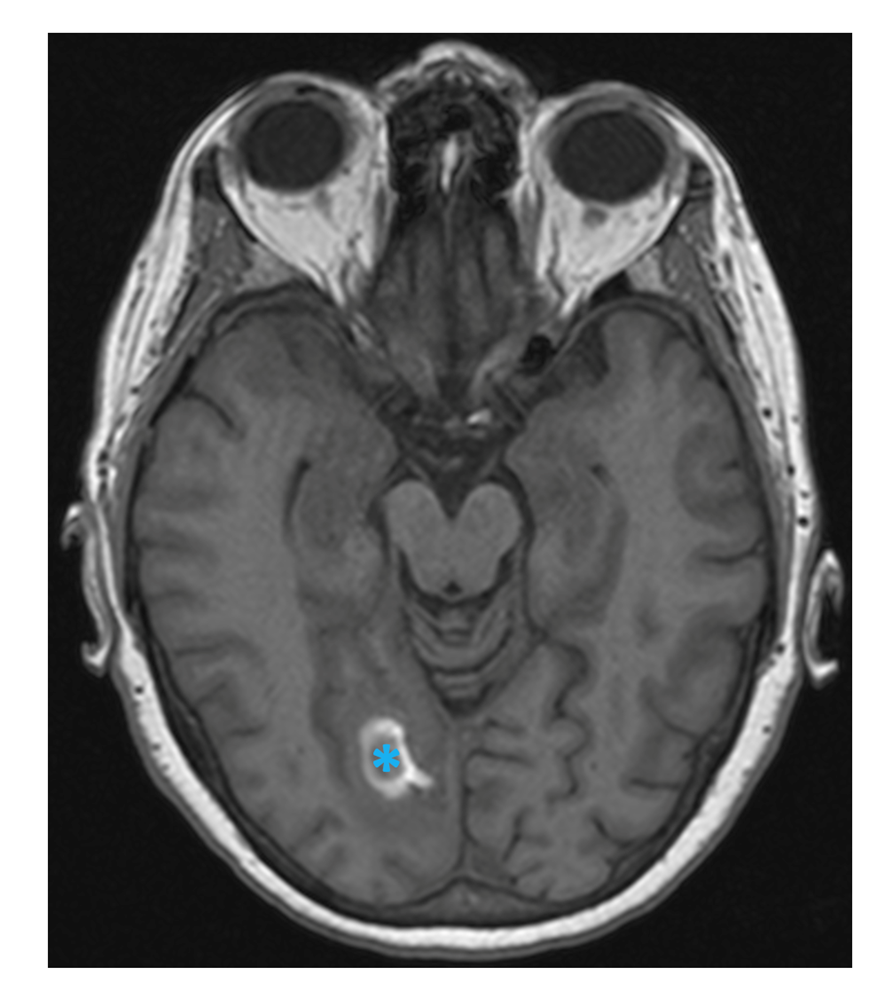
Figure 3
