Microbial Contamination and Resistance Profile of Bacteria Isolated from Mobile Phones in a Health Care Setting in Dubai, UAE
Microbial Contamination and Resistance Profile of Bacteria Isolated from Mobile Phones in a Health Care Setting in Dubai, UAE
Madhulika Manoharan1*, Aaradhya Manoharan2
1. 10th grade student at GEMS Our Own English High School, Al Warqa, Dubai.
2. 5th grade student at GEMS Our Own English High School, Al Warqa, Dubai.
*Correspondence to: Madhulika Manoharan, 10th grade student at GEMS Our Own English High School, Al Warqa, Dubai.
Copyright.
© 2025 Madhulika Manoharan This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 13 Oct 2025
Published: 14 Oct 2025
Abstract
Objective: To assess the prevalence, microbial profile of organisms isolated from mobile phones used by health care personnel in Dubai, UAE.
Methodology: A total of 230 mobile phones from healthcare providers in various clinical settings were swabbed and cultured using standard microbiological techniques. The pathogens were identified and classified into Gram-positive cocci (GPC), Gram-negative bacilli (GNB), and others. Resistance screening was performed for Staphylococcus aureus isolates to detect methicillin resistance.
Results: Microbial growth was observed in 114 of the 230 samples (49.6% of the total samples). Among these, 98 samples (85.9%) were Gram Positive Cocci and 14 samples (12.3%) were Gran Negative Bacteriae. Gram Positive Isolates included 83 Micrococcus species. and 15 Staphylococcus aureus samples, of which 1 was an MRSA sample. GNB isolates comprised of 10 Klebsiella species. and 4 E. coli, with no resistance detected. One fungal spore and one Bacillus spore were also identified.
Conclusion: Mobile phones in clinical settings harbor significant microbial contamination,which poses a significant threat to the patients they treat. Although resistance was low, the presence of MRSA and spore-forming organisms underscores the need for routine disinfection protocols and awareness among healthcare personnel.
Keywords: Mobile phones, microbial contamination, MRSA, Gram-positive cocci, Gram-negative bacilli, Dubai.
Microbial Contamination and Resistance Profile of Bacteria Isolated from Mobile Phones in a Health Care Setting in Dubai, UAE
1. Introduction
Cell phones are essential in all aspects of daily life. They have especially played a critical role in enhancing services in the healthcare industry, making it easier for healthcare workers to communicate and go about their routine much more comfortably. Moreover, these improved facilities born as a result of the introduction of mobile phones in the aforementioned sector have resulted in the development of the healthcare system and the more efficient administration of treatment to patients. However, due to infrequent sanitization of cell phone surfaces and lack of surveillance of staff sanitary measures in hospitals and clinics alike, mobile phones have become germ reservoirs and have been found to serve as silent carriers of healthcare-associated diseases, putting the healthcare worker and their friends and family at risk. According to statistics regarding this concern, around 80,000 to 180,000 infections could be avoided annually (Gastmeier et al.,2010), specially via sufficient hand cleanliness and increased intensity of cellular phones cleaning (Tannhäuser et al.,2022).
The pathogenic composition and prevalence in the cell phones of various classes differ depending on a variety of external factors. For instance, in high-income countries, 75–96% of healthcare workers’ mobile phones were found to be infected, with Coagulase-negative staphylococci (CoNS) and Micrococcus being the most common strains. In low- and middle-income countries’ healthcare settings, the bacterial prevalence ranged from 42% to 100%., with the most prevalent bacteria being coagulase-negative staphylococci, E.coli, Acinetobacter species, Pseudomonas species, and MRSA bacteria.
The global burden of healthcare-associated infections (HAIs) is increasing, resulting in increased patient mortality and costs for maintenance and treatment. Contamination of the mobile phones used by healthcare workers has resulted in the discovery of the prevalence of drug-resistant organisms such as MRSA and vancomycin-resistant enterococci (VRE) on their surfaces, which could be fatal for the patients that healthcare workers are diagnosing and treating. This study aims to assess and investigate microbial carriage on mobile phones used by laboratory staff in Dubai, UAE, with emphasis on prevalence, organism distribution, and resistance profile.
2. Materials and Methods
2.1 Study Design
A cross-sectional study was conducted at QLABS Clinical Laboratory, Karama, Dubai. A total of 230 mobile phones were sampled over a 2-month period from various laboratories, clinics and other healthcare settings.
2.2 Study Period
This study was conducted from July 11th to September 11th 2025.
2.3 Sample Collection
Sample Collection and Microbiological Analysis
Mobile phone surfaces were swabbed using saline-moistened sterile swab sticks, following strict aseptic protocols. Particular attention was given to the keypad and rear surfaces. Swabs were inoculated onto blood agar (BA) plates, MACConkey medium, Sabouraud dextrose agar and incubated aerobically at 37°C for 24–48 hours.
Identification of Bacterial Isolates
Bacterial colonies were identified using standard microbiological techniques, including Gram staining, selective media culture, biochemical profiling (TSI, Citrate, Indole, MR, VP, Urease, Coagulase, Catalase and Oxidase) and confirmed through API and bioMérieux software and latex agglutination.
Antibiogram Profiling
Antibiotic susceptibility was assessed using the Kirby-Bauer disk diffusion method. Test organisms (adjusted to 0.5 McFarland standard) were inoculated onto Mueller-Hinton agar (MHA) plates. Antibiotic discs —(Gram positive: Penicillin, Oxacillin, Cefoxitin, Ciprofloxacin and Erythromycin) Gram Negative (Ampicillin, 1st 2nd and 3rd generation Cephalosporin, Carbapenem, Beta lactam lactamase inhibitor)—were applied using sterile forceps. Plates were incubated at 37°C for 24 hours under aerobic conditions.
2.4 Identification and Classification
Isolates were identified using Gram staining and standard biochemical tests. ATCC strains, specifically ATCC staph aureus 25923 and ATCC E.coli 25922 were used for quality control. Organisms were classified as GPC (Gram Positive Bacteria), GNB (Gram Negative Bacteria), fungal spores, or Bacillus spp.
2.5 Resistance Screening
Staphylococcus aureus isolates were screened for methicillin resistance using cefoxitin disc diffusion. Gram negative organisms were screened for ESBL and other resistance pattern.
3. Results
3.1 Overview
A total of 230 samples were assessed for bacterial growth, out of which 114 (49.6%) isolates showed growth of GPCs and GNB’s alike. Out of the 114 samples, 98 (85.9%) were Gram Positive Bacterial Isolates, 14 (12.3%) were Gram Negative Bacterial Isolates, and the remaining two samples consisted of 1 fungal spore (0.9%) and 1 bacillus isolate (0.9%). For the GPC isolate distribution, 83 (84.7%) of the 98 samples were Micrococci isolates, 15 (15.3%) were Staphylococcus isolates, and within those 15 samples, 1 (6.7% of S. aureus isolates) was identified as an MRSA isolate. Continuing with the GNB distribution, 10 out of the 14 samples were Klebsiella Species (71.4%) and the remaining 4 were E. coli isolates.
3.2 Distribution of Isolates
Table 1: Distribution of Isolates |
|---|
3.3 Breakdown of GPC
|
Group |
Count |
% of Positives |
|
Micrococcus spp. |
83 |
84.7% |
|
Staphylococcus aureus |
15 |
15.3% |
|
MRSA |
1 from S. aureus |
6.7% of S. aureus isolates |
Table 2: Breakdown of GPC
3.4 Breakdown of GNB
|
Group |
Count |
% of Positives |
|
Klebsiella spp |
10 |
71.4% |
|
E. coli |
4 |
28.6% |
Table 3: Breakdown of GNB
Figure 1: Brief Culture Growth Distribution Chart
Figure 2: Distribution of Microbial Isolates
Figure 3: Detailed Distribution of Isolates
Figure 4: Distribution of Gram-Positive Bacterial Isolates
Figure 5: Distribution of Gram-Negative Bacterial
Figure 6: Sample Collection and Processing Results
4. Discussion
The contamination rate of 49.6% aligns with global studies reporting 40–70% prevalence on mobile phones in healthcare settings. GPCs, particularly Micrococcus spp., dominated the isolates, further highlighting the significance of skin flora transfer in hospital environments. The detection of MRSA, though low, is clinically significant due to its role in hospital-acquired infections. Absence of resistance in GNB isolates may reflect effective antimicrobial stewardship or limited exposure to resistant strains in this cohort. However, the presence of fungal and bacillus spores is concerning and despite strict adherence to sanitation measures in healthcare settings, proves that pathogenic contamination cannot be completely avoided. HCWs need to take more caution in this aspect to avoid such dangerous circumstances.
Comparative studies from India and the UAE have shown similar trends, with CoNS and S. aureus being the most frequently isolated organisms. The low resistance profile in this study contrasts with findings from tertiary care centres where multidrug-resistant strains are more prevalent. This may reflect differences in patient load, disinfection practices, and environmental controls. However, the presence of low resistance bacteria doesn’t completely eliminate the dangers associated with this type of contamination. If the same unsanitary measures continue in healthcare settings, the prevalence and intensity of pathogens and their impact on patients and healthcare workers in the near future will certainly rise to a concerning extent.
5. Conclusion
The present study demonstrates that mobile phones used by healthcare personnel (HCPs) can serve as reservoirs for Spread of infection.
Although the majority of isolates identified were opportunistic in nature, their potential to cause nosocomial infections in vulnerable patient populations underscores the importance of routine device hygiene. Based on our findings, we recommend that HCPs adopt regular disinfection practices, such as cleaning mobile phones with 70% isopropyl alcohol. Such measures are simple yet effective in reducing the risk of healthcare-associated infections (HAIs), especially during the presence of mutant strains, when stringent infection control protocols are imperative.
Mobile phones used in clinical laboratories are frequently contaminated with bacteria, primarily Gram-positive organisms. While antibiotic resistance was minimal, the detection of MRSA and spores warrants regular disinfection and staff education. Implementation of phone hygiene protocols may reduce the risk of cross-contamination and nosocomial transmission.
Acknowledgements
The author thanks the CEO of QLabs Clinical Laboratory, Mr. Pankaj Sohaney for their approval to conduct this project in their laboratory, and the proactive microbiology and technical staff department for their support during sample collection and analysis. The author thanks Dr. Dheepa Manoharan, Medical Director and Specialist Microbiologist of QLabs Clinical Laboratory for her valuable insights, guidance and assistance throughout the conduction of this study. The author also thanks this study’s participants for their cooperation in conducting this research.
Conflicts of Interest
None declared.
References
1. Ulger F et al. Ann Clin Microbiol Antimicrob. 2009;8:7.
2. Banawas S et al. Can J Infect Dis Med Microbiol. 2018.
3. Shah PD et al. Indian J Public Health. 2019;63:147–50.
4. Shivani S et al. Pharm Biosci J. 2020;8(6):18–23.
5. Saroj AC et al. Int J Curr Pharm Res. 2021;13(1):54–56.
6. Zenbaba, D., Sahiledengle, B., Beressa, G., Desta, F., Teferu, Z., Nugusu, F., Atlaw, D., Shiferaw, Z., Gezahegn, B., Mamo, A., Desalegn, T., Negash, W., Negash, G., Mama, M., Nigussie, E., & Chattu, V. K. (2023). Bacterial contamination of healthcare workers’ mobile phones in Africa: A systematic review and meta-analysis. *Tropical Medicine and Health, 51*(1), Article 55. https://doi.org/10.1186/s41182-023-00547-3
7. Algmati, A. A. M., Ali, A. A., Mohammed, S. S., Iqdoura, S. J., Ahmoda, S. S., Abualgasam, S. A., Ali, S. K., Younis, S. M., Abulobaida, M. A., & Salem, Z. R. (2024). Prevalence and risks of mobile phone microbial contamination among medical, dental, and health sciences students in the healthcare setting. International Journal of Biomedical and Clinical Research, 1(4), 1–8. https://doi.org/10.59657/2997-6103.brs.24.024
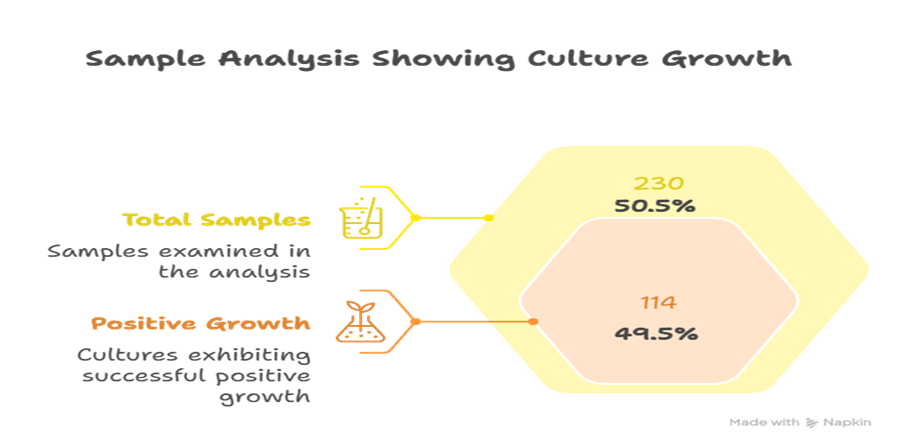
Figure 1
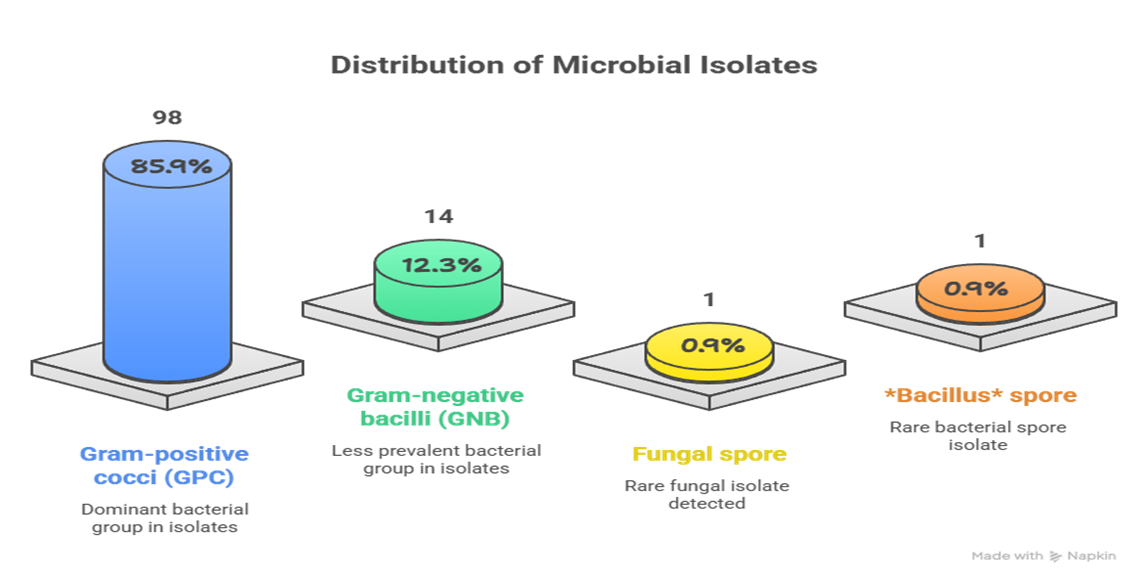
Figure 2
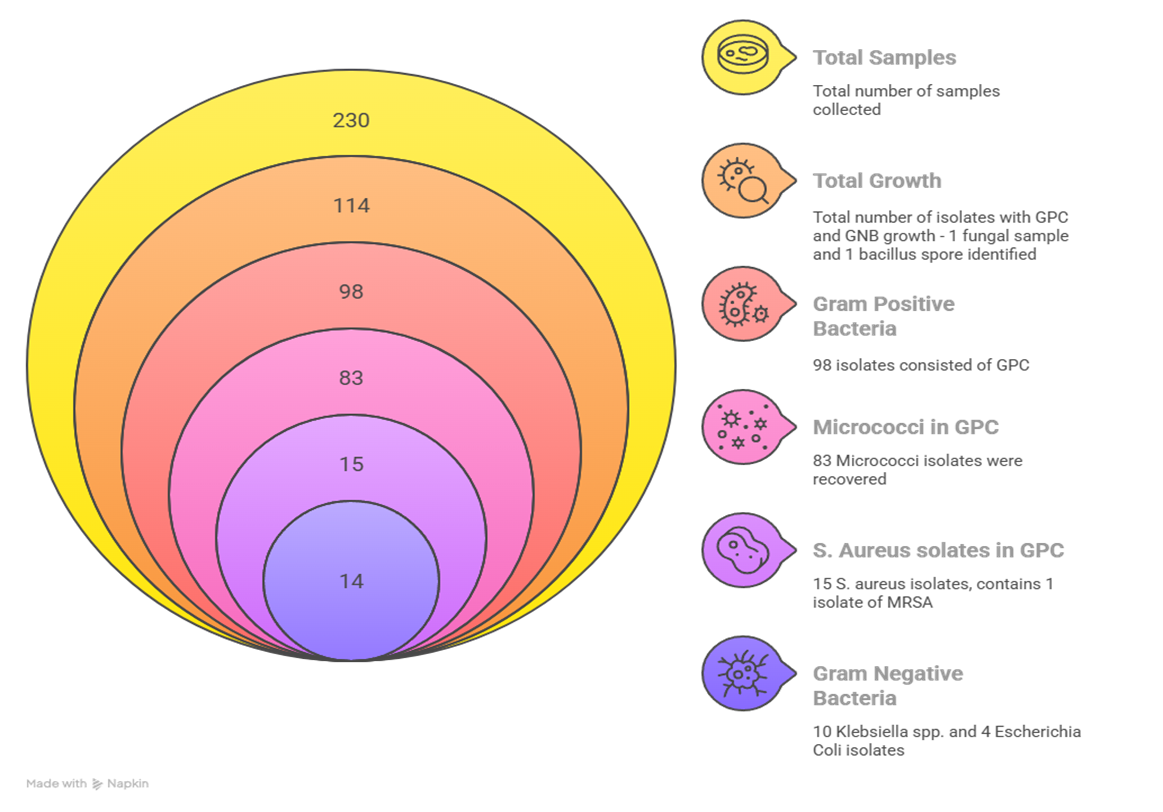
Figure 3
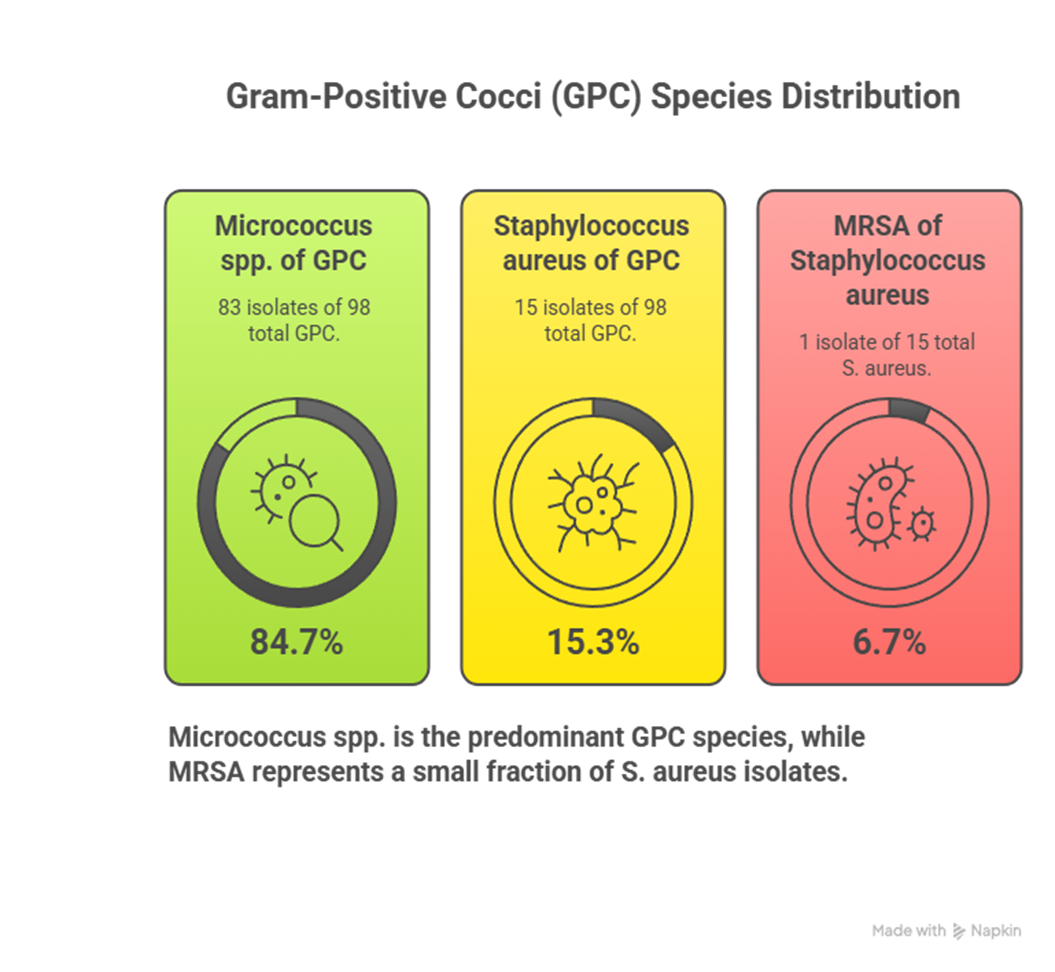
Figure 4

Figure 5

Figure 6
