Impact of p95HER2 Expression on Clinical Outcomes in HER2-Positive Breast Cancers: A Literature Review
Impact of p95HER2 Expression on Clinical Outcomes in HER2-Positive Breast Cancers: A Literature Review
Dr. Lesiba Terrance Khalo *
*Correspondence to: Dr. Lesiba Terrance Khalo, Military Hospital, Pretoria, Gauteng Province, South Africa.
Copyright.
© 2025 Dr. Lesiba Terrance Khalo This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 08 Oct 2025
Published: 17 Oct 2025
Impact of p95HER2 Expression on Clinical Outcomes in HER2-Positive Breast Cancers: A Literature Review
Introduction
Human epidermal growth factor receptor 2 (HER2), is one of a family of 4 membrane tyrosine kinases (TKs), which was first discovered 25 years ago, and shown to be overexpressed in a human breast cancer cell (5) with its prevalence shown to be important in pathogenesis and disease progression in breast cancer patients (6).
The HER2 pathway was described in systems biology terms as a complex biological network composed of 3 layers: 1) An input layer of membrane receptors, alongside their ligands, whose function is to activate signal extracellular cascades, 2) A core system processing layer of protein kinases (PKs) who’s roles are signal transmission to the nucleus, followed by 3) An output layer of transcription factors, which function by regulating genes that effect various cellular functions, such as proliferation, migration, apoptosis as well as angiogenesis (1).
The input layer is composed of 4 membrane receptors/kinases (HER1–4) and their many ligands, including endothelin growth factor (EGF), transforming growth factor alpha (TGF-α), and heregulin. However, in breast cancer, HER2 is the dominant kinase receptor, overexpressed in 20% of cases (6). Upon ligand binding, the HER receptors undergo conformational changes that enable homo- or heterodimerization, followed by transphosphorylation of the receptors. HER2 does not have ligand- binding capabilities, but its open conformation allows for dimerization. When overexpressed, the activated form heterodimerizes with other HER members that have ligand-binding capabilities or undergoes homo-dimerization (1). This dimerization leads to the activation of the PI3K/AKT anti- apoptosis pathway (Figure 1), thereby stimulating the activation of transcription factors and exhibiting a proliferative, migratory, angiogenic, and differentiation cancer phenotypes. The estrogen receptor (ER) and the HER2 signaling pathways are the dominant drivers of cell proliferation and survival in most (85%) breast cancers (1).
Figure 1: HER-2 signaling pathway (24).
Some HER2 positive breast cancers have been shown to express the truncated form of HER2, known as p95HER2, which lacks the extracellular domain. This lack of the extracellular domain has been attributed to resistance to drugs such as trastuzumab, as it works through domain-binding mechanisms (7, 8).
This study focuses on the expression of p95HER2, a truncated form of HER2, and its potential impact on breast cancer patients receiving trastuzumab. The objective is to determine whether high levels of p95HER2 in these patients are associated with a worse disease prognosis compared to patients without these mutations. The writer will conduct a comprehensive literature review to explore this issue in the context of South Africa.
Hypothesis
Elevated expression of P95HER2 in HER2-positive breast cancers is associated with poorer clinical outcomes, including progression-free survival (PFS) and overall survival (OS), due to its role in mediating resistance to trastuzumab therapy.
Project objectives
A comprehensive literature review delineating the impact of p95HER2 expression on clinical outcomes in HER2-positive breast cancers has was undertaken, to assess the correlation between p95HER2 expression levels and clinical outcomes such as overall survival (OS), progression-free survival (PFS), recurrence-free survival (RFS), pathologic complete response (pCR) as well as examining the therapeutic implications of p95HER2 expression on the efficacy of HER2 targeted therapies in trastuzumab.
Methodology
We conducted comprehensive literature review; 1) To gain a better understanding on the topic, by identifying recent literature reviews on the aforementioned, followed by selection of relevant original papers, 2) The search strategy and selection criteria to narrow down the search, 3) Appropriate methods for selecting relevant and up-to-date papers were undertaken; such include, but are not limited to the search language, prominent authors, papers with more citations, as well as credible peer-reviewed journals with higher impact factors, 4) The search was not limited to peer-reviewed papers and books, but extended to published conference proceedings and thesis. Below, in Figure 2 is a graphical depiction of the review strategy and workflow.
Figure 2: The graphical depiction of the methodology workflow. A selection of 16 peer reviewed articles specifically looking at clinical data depicting the impact of p95HER2 expression, and its association with disease severity were carefully selected for this review.
Scholarly databases and search engines, such as PubMed, Scopus, Google Scholar, Elsevier, and ScienceDirect, were used to develop a comprehensive search strategy. The researcher used the following keywords in the search: p95HER2, HER2-positive breast cancer, clinical outcomes, trastuzumab, treatment response, and resistance. The Medical Subject Headings (MeSH) keywords and Boolean operators were used to narrow the search. Studies published 10-15 years ago, to date, were included. Both clinical studies and original research articles were included in the criteria. Studies that included the use of the following scientific techniques —immunohistochemistry, FISH, and sequencing of HER2- positive tissue/cells — were included. Preference was given to authors whose research was primarily conducted within the South African context, particularly in clinical studies. Pre-clinical (animal) studies were excluded from the search, alongside articles that were older than 15 years
Results
Following a comprehensive literature search using the above databases (PubMed, Scopus, Google Scholar, Elsevier, ScienceDirect), a vast body of literature was retrieved. Followed by a manual pick of original articles, thus excluding literature reviews. The focus was on clinical studies, whether retrospective or Phase 1, 2, or 3 clinical trials. Here, we picked very few articles that specifically touched on p95HER2-positive breast cancer patients on trastuzumab treatment. Below is an account of the selected articles that depict the correlation between p95HER2 expression in breast cancer patients and clinical outcomes, specifically PFS and OS (Table 1). Other endpoint clinical outcomes in this literature, such as recurrence-free survival (RFS), disease-free survival (DFS), and pathological complete response (pCR), were also included.
Table 1: Studies that depict the correlation between p95HER2 expression in breast cancer patients and clinical outcomes, specifically the PFS and OS, as well as second-line treatment studies.
|
Author |
Study type (clinical) |
Patient sample type (Number of participants) |
Major findings |
Reference |
|
Goh et al., 2024 |
Retrospective |
FFPE (59) |
Prevalence of p95HER2 in premenopausal women, with worsening DFS. |
14 |
|
Rigakos et al., 2021 |
Retrospective |
FFPE (114) |
An aggressive cancer phenotype was associated with high expression of p95HER2, as per high CNVs, as well as expression of Ki67. |
15 |
|
Lipton et al., 2013 |
Retrospective |
FFPE (89) |
Both HER3 and p95HER2 expressing patients exhibited poor clinical outcomes. |
16 |
|
Chumsri et al., 2018 |
Retrospective |
FFPE (91) |
The ratio of p95HER2/HER2 was significantly associated with worsening PFS. |
17 |
|
Duchnowska et al., 2017 |
Retrospective |
FFPE (189) |
Second-line Lapatinib treated patients with high p95HER2 expressions may have the best clinical outcomes while on treatment. |
20 |
|
Han et al., 2012 |
Retrospective |
FFPE (52) |
With combination therapy of Lapatinib plus Capecitabine, there was no significant association between p95 isoform and PFS/OS. |
21 |
|
Cao et al., 2023 |
Single-arm, multicentre phase 2 trial |
Clinical trial (100 patients) |
With a combination of pyrotinib and capecitabine, the median PFS increased from 8 to 11 months post treatment. |
22 |
Several studies have shown that the increased expression of a truncated version of HER2, p95HER2, is a critical determinant of poor prognosis and therapeutic response in HER2-positive breast cancer. It has also been shown that the median survival rate of these patients can be as low as 8 months. To better understand these underlying resistance mechanisms, conventionally, 2D tumor cell culture models have been used. However, these models, due to their monolayer nature, cannot fully recapitulate the tumor micro-environment, thereby warranting the development of more biologically complex, yet clinically relevant models of disease. Scaffold (matrix)-free self-assembled aggregates of cancer cells (spheroids), particularly known as organoids, are suitable in vitro models of disease (9-12). They have therefore been extensively used in various settings to study tumor progression and mechanisms of resistance (12).
This literature review aims to assess the correlation between p95HER2 expression levels and clinical outcomes, including progression-free survival (PFS) and overall survival (OS). In addition, it examined the therapeutic implications of p95HER2 expression on the efficacy of HER2-targeted therapies in trastuzumab. Trastuzumab (Herceptin) and pertuzumab are two of the HER-2 receptor-binding humanized monoclonal antibodies that have been approved by the FDA as therapeutic agents for HER2-positive breast cancers (13). However, some breast cancers have been shown to express an aberrant form of HER2, missing the extracellular domain. This truncated isoform of HER2 is commonly referred to as p95HER2, which has been attributed to resistance to drugs such as trastuzumab. Below is an account of this resistance mechanism, alongside the resulting clinical outcomes in patients harboring such mutations.
A recent study by Goh et al. (2024) showed a higher recurrence and metastasis rate in patients with p95HER2-positive tumors compared to those with no p95HER2 expression. This was a retrospective study wherein formalin-fixed paraffin-embedded (FFPE) tissue sections were stained with the p95HER2 antibody to evaluate expression. This study was designed to include samples from 30 non-relapsed breast cancer patients, alongside 29 primary trastuzumab-resistant patient samples. In their study, the group demonstrated that p95HER2 positivity was associated with worse disease-free survival (DFS) in patients undergoing trastuzumab treatment (14). Similarly, a study by Rigakos et al. (2021) showed an association between the P95-isoform and resistance to trastuzumab. Here, the authors found that high p95-isoform expressing patients had a greater risk of death, and that this isoform was correlated to increased expression levels of proliferation marker (ki-67) as well as gain of HER2 copy number variations (CNVs), thus indicative of a more aggressive cancer phenotype (15).
In their quest to further elucidate the “trastuzumab escape” biomarkers, a study by Lipton et al. (2013 designed a dual antibody proximity-based assay for the precise quantitation of HER3 total protein (H3T). Here, 89 FFPE sections of metastatic breast patients previously on trastuzumab treatment were assessed for clinical outcomes such as OS and PFS using a VeraTag H3T assay. The data was analyzed via Kaplan–Meier and decision tree analysis. This study also showed that patients on the treatment exhibited poor clinical outcomes (16). Here, patients with high expression levels of H3T showed a lower PFS compared to those with low expression levels. However, there were no significant differences in OS between the high- and low-expressing samples. Furthermore, to validate the results, IHC, ELISA, and FACS were also performed, and the results correlated (16).
Due to the varying data regarding the correlation between p95HER2 expression and poor clinical outcomes, Chumsri et al. (2018 sought to evaluate the ratio of p95HER2/HER2 in breast cancer patients. Here, two Phase II clinical trials were conducted to evaluate the correlation between quantitative levels of HER2, p95HER2, and p95HER2/HER2, and clinical outcomes following trastuzumab-based chemotherapy in metastatic HER2-positive breast cancer patients (17). The study concluded that worsening PFS and OS are significantly correlated with p95HER2/HER2 ratio (17).
Furthermore, the studies described mechanisms underlying the correlation between the p96-isoform and poor clinical outcomes were limited in the clinical setting. Most similar studies are in vitro (9-12). Recent clinical studies have indicated that there may be specific co-expressed biomarkers correlating to the pathological and clinical parameters of poor prognosis in HER2-positive breast cancer (18), as well as specific mutations (19). A study by Hergueta-Redondo et al. (2016 showed that the HER2 co- amplified and co-expressed Gasdermin (encoded by the gene GSDMB is a potential critical determinant of poor prognosis and therapeutic response in HER2-positive breast cancer (23).
To elucidate the genome alterations in this cancer phenotype, a recent study by Ma et al. (2024 performed a gene-targeted genome sequencing in HER2+ breast cancer patients. In this study, approximately 650 mutations were reported. Two mutations of note were those in the tumor suppressor genes NF1 and ATM, which were associated with trastuzumab resistance (19). Specifically, the NFI mutation was associated with a higher risk of early patient relapses (19). This is the first study to identify the unique cancer-related gene mutation profile, thus laying a foundation for prospective studies aimed at further elucidating resistance mechanisms to trastuzumab.
Due to the nature of resistance to treatment in patients with positive p95HER2 expression, second-line treatment options have been made available to these patients to increase their PFS and OS rates. In their retrospective study, Duchnowska et al. (2017) quantified the expression levels of HER2, HER3, and p95HER2 proteins, thus allowing for a more in-depth analysis of their potential predictive relevance in determining patients’ PFS and OS. This study showed that patients treated with Lapatinib (a HER1 and HER2 tyrosine kinase inhibitor (TKI)) who have high p95HER2 expression may have the best clinical outcomes while on treatment (20).
The Lapatinib Expanded Access Program (a combinatory treatment regimen of Lapatinib and capecitabine) utilized VeraTag assays to quantify the expression levels of HER2, HER3, and p95HER2 proteins from FFPE tissue samples from breast cancer patients who showed resistance to trastuzumab. Here, there was no significant correlation between clinical outcomes and p95 expression levels in their cohort of HER2-positive, trastuzumab-refractory metastatic breast cancer patients (21). On the contrary, a recent investigator-initiated, single-arm, phase 2 trial conducted in China at 16 sites revealed that both combinations of pyrotinib and capecitabine can be a treatment option for HER2-positive advanced breast cancer patients exhibiting trastuzumab resistance (22). In this study, the primary endpoint was the investigator-initiated PFS. This study showed that the median PFS increased from 8 to 11 months post-treatment (22).
Discussion
This literature review aimed to assess the correlation between p95HER2 expression levels and clinical outcomes, including progression-free survival (PFS) and overall survival (OS). In addition, we examined the therapeutic implications of p95HER2 expression on the efficacy of HER2-targeted therapies in trastuzumab. Several studies have shown that the increased expression of p95HER2 is a critical determinant of poor prognosis and therapeutic response in HER2-positive breast cancer, with the median survival rate of these patients as low as 8 months.
Studies have shown a higher recurrence and metastasis rate in patients with p95HER2-positive tumors. The research discovered that these patients had a greater risk of death. The p95HER2 expression correlated with increased expression levels of proliferation markers, such as Ki-67, as well as the gain of HER2 copy number variations (CNVs), thus indicating a more aggressive cancer phenotype, which led to lower PFS as well as OS rates in patients with high expression levels of p95HER2 in a clinical setting.
While our understanding of the mechanisms underlying the correlation between the p95-isoform and poor clinical outcomes is growing, there is still much to be done. Most research in this area has been conducted in vitro, and the clinical setting presents its own limitations. However, recent clinical studies have indicated that there may be specific co-expressed biomarkers and gene mutations that correlate with the pathological and clinical parameters of poor prognosis in HER2-positive breast cancer. For instance, a study by Hergueta-Redondo et al. (2016) identified GSDMB as a co-expressed biomarker. This gene product is a member of a family of gasdermins that regulate apoptosis in epithelial cells and contribute to disease progression. Through Rab7 activation, GSDMB is involved in autophagy and plays a pivotal role in modulating autophagosome maturation. The process, in turn, facilitates cancer cell resistance through the mechanism of autophagy. Ma et al. (2024) also identified two mutations in the tumor suppressor genes NF1 and ATM, showing that these were associated with trastuzumab resistance. Both studies highlight the need for further research to fully understand the mechanisms underlying the correlation between p95HER2 expression levels and poor clinical outcomes. This work is urgent and of utmost importance in the fight against p95HER2-positive breast cancer.
Due to the nature of resistance to treatment in patients with p95HER2 expression, second-line treatment regimens, as well as combination chemotherapies, have been made available to these patients to increase their PFS and OS rates. Drugs that target other HER isoforms, in place of HER2, have been used in the clinical setting—for instance, Lapatinib (a HER1 and HER3 inhibitor was used as a second- line drug. Furthermore, in a study by Han et al. (2012), Lapatinib, a small-molecule dual tyrosine-kinase inhibitor was used in combination therapies, where it was paired with capecitabine, which is activated to 5'-fluorouracil (5FU) and inhibits thymidine synthesis. However, there have been contradictory clinical outcomes, with variations in patient PFS. Some studies have shown an increase in PFS following their second-line combination chemotherapies (22), while others have shown no significant increase (21). These variations were attributed to the use of different combinations of drugs. For instance, the Lapatinib Expanded Access Program study by Han et al. (2012), which used a combinatory treatment regimen of Lapatinib and capecitabine, showed no significant increase in patient PFS. On the contrary, a PICTURE study by Cao et al. (2023 used pyrotinib plus capecitabine and showed an increase in PFS. Pyrotinib is a small-molecule tyrosine-kinase inhibitor that prevents homo-/homo-/heterodimerization of HER2. The inconsistency in the data suggests that further work is needed to standardize second-line combination therapies. This standardization is crucial for guiding future research and clinical decisions, as it will help identify the most effective treatment regimens for patients with p95HER2-positive breast cancer.
Conclusions and Further Recommendations
This literature review highlights some of the significant studies that delineate the effects of p95HER2 expression on clinical outcomes of breast cancer patients. It also provides an overview of the recent significant advances in therapeutic interventions for these patients, which aim to improve patient prognosis. Given the inconsistency in data, future work needs to be done to standardize the second- line combination therapies. Furthermore, more studies are needed to identify the unique cancer-related gene mutation profiles, thus laying a foundation for prospective studies aimed at further elucidating resistance mechanisms to trastuzumab. Lastly, we emphasize the need for urgent and important research to elucidate further the mechanisms underlying the correlation between p95HER2 expression levels and poor clinical outcomes.
Acknowledgement: European School of Oncology(ESO) , Centre for Biosciences(CEB), Madrid, all colleaques from 1 Military Hospital Surgical oncology department and my family support.
References
1. Gutierrez C and Schiff R. HER2: Biology, Detection, and Clinical Implications. Arch Pathol Lab Med. January 2011; 135(1): 55–62. doi:10.1043/2010-0454-RAR.1.
2. Goh CW, Yang B, Chi Y, and Wu J. p95HER2 expression in HER2-positive breast cancer exhibiting primary resistance. Precision Medical Sciences. 2024; 13:99–106. DOI: 10.1002/prm2.12128.
3. Kallergi G., Agelaki S., Papadaki M.A., Nasias D., Matikas A., Mavroudis D., and Georgoulias
V. Expression of truncated human epidermal growth factor receptor 2 on circulating tumor cells of breast cancer patients. Breast Cancer Research (2015) 17:113 DOI 10.1186/s13058-015- 0624-x.
4. Chumsri S, Sperinde J, Liu H, Gligorov J, Spano JP, Antoine M, Aspitia AM, Tan W, Winslow J, Petropoulos CJ, Chenna A, Bates M, Weidler JM, Huang W, Dueck A, and Perez EA. High p95HER2/HER2 Ratio Associated with Poor Outcome in Trastuzumab-Treated HER2-Positive Metastatic Breast Cancer NCCTG N0337 and NCCTG 98-32-52 (Alliance). 2018 American Association for Cancer Research. doi: 10.1158/1078-0432.CCR-17-1864.
5. King CR, Kraus MH, Aaronson SA. Amplification of a novel v-erbB-related gene in a human mammary carcinoma. Science. 1985;229(4717):974–976.
6. Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235(4785):177–182.
7. Molina MA, Saez R, Ramsey EE, et al. NH(2)-terminal truncated HER-2 protein but not full- length receptor is associated with nodal metastasis in human breast cancer. Clin Cancer Res. 2002;8(2):347–353.
8. Scaltriti M, Rojo F, Ocana A, et al. Expression of p95HER2, a truncated form of the HER2 receptor, and response to anti-HER2 therapies in breast cancer. J Natl Cancer Inst. 2007;99(8):628–638.
9. Muñoz L, Espinosa M, Quintanar-Jurado V, Hidalgo A, Melendez-Zajgla J, Maldonado V. Paradoxial changes in the expression of estrogen receptor alpha in breast cancer multicellular spheroids. Tissue and Cell. 2010 Oct;42(5):334–7.
10. Rodríguez CE, Reidel SI, Bal ED, Jasnis MA, Fiszman GL. Autophagy Protects from Trastuzumab-Induced Cytotoxicity in HER2 Overexpressing Breast Tumor Spheroids. PloS one. 2015 Sep 11;10(9):e0137920–0.
11. Han SJ, Kwon S, Kim KS. Challenges of applying multicellular tumor spheroids in preclinical phase. Cancer Cell International. 2021 Mar 4;21(1).
12. Tapia IJ, Perico D, Wolos VJ, Villaverde MS, Abrigo M, Di Silvestre D, et al. Proteomic Characterization of a 3D HER2+ Breast Cancer Model Reveals the Role of Mitochondrial Complex I in Acquired Resistance to Trastuzumab. International journal of molecular sciences [Internet]. 2024 May;25(13):7397. Available from: https://pubmed.ncbi.nlm.nih.gov/39000504/
13. Gemmete JJ, Mukherji SK. Trastuzumab (Herceptin). American Journal of Neuroradiology. 2011 Aug 4;32(8):1373–4.
14. Goh CW, Yang B, Chi Y, Wu J. p95HER2 expression in HER2?positive breast cancer with primary resistance. Precision Medical Sciences. 2024 Apr 4;13(2):99–106.
15. Georgios Rigakos, Evangelia Razis, Georgia-Angeliki Koliou, Georgios Oikonomopoulos, Eleftheria Tsolaki, Sperinde J, et al. Evaluation of the Role of p95 HER2 Isoform in Trastuzumab Efficacy in Metastatic Breast Cancer. Anticancer Research. 2021 Apr 1;41(4):1793–802.
16. Lipton A, Goodman L, Leitzel K, Cook J, Sperinde J, Haddad M, et al. HER3, p95HER2, and HER2 protein expression levels define multiple subtypes of HER2-positive metastatic breast cancer. Breast Cancer Research and Treatment. 2013 Aug;141(1):43–53.
17. Chumsri S, Sperinde J, Liu H, Gligorov J, Spano JP, Antoine M, et al. High p95HER2/HER2 Ratio Associated With Poor Outcome in Trastuzumab-Treated HER2-Positive Metastatic Breast Cancer NCCTG N0337 and NCCTG 98-32-52 (Alliance). Clinical Cancer Research [Internet]. 2018 Jul 1;24(13):3053–8. Available from: https://clincancerres.aacrjournals.org/content/24/13/3053.
18. Hergueta-Redondo M, Sarrio D, Molina-Crespo Á, Vicario R, Bernadó-Morales C, Martínez L, et al. Gasdermin B expression predicts poor clinical outcome in HER2-positive breast cancer. Oncotarget. 2016 Jul 22;7(35).
19. Ma G, Huo B, Shen Y, Zhu X, Cheng C, Li W, et al. Genomic Alterations Correlated to Trastuzumab Resistance and Clinical Outcomes in HER2+/HR- Breast Cancers of Patients Living in Northwestern China. Journal of Cancer [Internet]. 2024;15(14):4467–76. Available from: https://pubmed.ncbi.nlm.nih.gov/39006074/
20. Duchnowska R, Sperinde J, Czartoryska-Ar?ukowicz B, My?liwiec P, Winslow J, Radecka B, et al. Predictive value of quantitative HER2, HER3 and p95HER2 levels in HER2-positive advanced breast cancer patients treated with lapatinib following progression on trastuzumab. Oncotarget. 2017 Oct 24;8(61):104149–59.
21. Han SW, Cha Y, Paquet A, Huang W, Weidler J, Lie Y, et al. Correlation of HER2, p95HER2 and HER3 Expression and Treatment Outcome of Lapatinib plus Capecitabine in her2-Positive Metastatic Breast Cancer. Tan M, editor. PLoS ONE. 2012 Jul 27;7(7):e39943.
22. Cao J, Teng Y, Li H, Zhang L, Ouyang Q, Xie W, et al. Pyrotinib plus capecitabine for trastuzumab-resistant, HER2-positive advanced breast cancer (PICTURE): a single-arm, multicenter phase 2 trial. BMC Medicine. 2023 Aug 9;21(1).
23. Gámez-Chiachio M, Ángela Molina-Crespo, Ramos-Nebot C, Martinez-Val J, Martinez L, Gassner K, et al. Gasdermin B over-expression modulates HER2-targeted therapy resistance by inducing protective autophagy through Rab7 activation. Journal of Experimental & Clinical Cancer Research. 2022 Sep 26;41(1).
24. HER2 Signaling Antibodies | Rockland [Internet]. www.rockland.com. Available from: https://www.rockland.com/resources/her2-signaling-antibodies/.
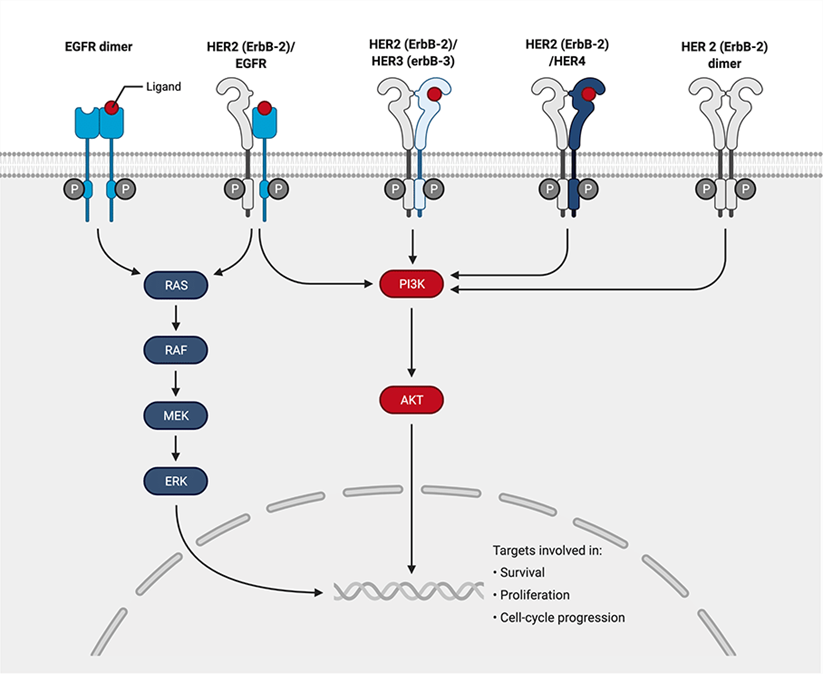
Figure 1
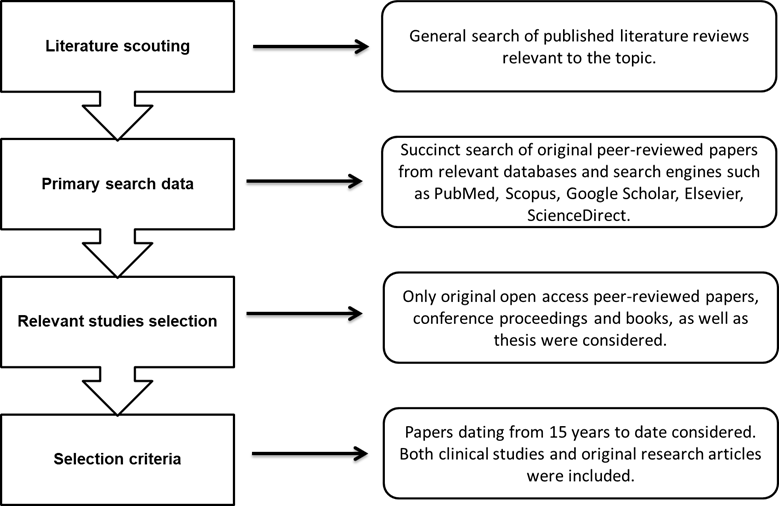
Figure 2
