Comprehensive Case Report: Relapsed Acute Basophilic Leukemia (Blast Crisis) Post-CML
Comprehensive Case Report: Relapsed Acute Basophilic Leukemia (Blast Crisis) Post-CML
Dr Lamiaa Abdel Khaleq Mohammed 1*, Dr Minerva Mohamed 2, Dr Amira Diaa Darwish 3
- Senior Resident of Hematology and BMT.
- Hematology and BMT Consultant/ Clinical Pathology Harmel Cancer Center.
- Hematooncology Consultant at NCI and Harmel Cancer Center.
*Correspondence to: Dr Lamiaa Abdel Khaleq Mohammed, Senior Resident, Hematology and BMT Harmel Cancer Center.
Copyright.
© 2025 Dr Lamiaa Abdel Khaleq Mohammed, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 27 October 2025
Published: 05 November 2025
Comprehensive Case Report: Relapsed Acute Basophilic Leukemia (Blast Crisis) Post-CML
Introduction
Chronic Myeloid Leukaemia (CML) is a myeloproliferative neoplasm defined by the Philadelphia chromosome, resulting from the t(9;22) (q34;q11) translocation, which creates the BCR::ABL1 fusion oncogene. Tyrosine kinase inhibitors (TKIs) like Imatinib are the standard first-line therapy, effectively controlling the chronic phase of the disease. However, a subset of patients may progress to a blast crisis (BC), an aggressive phase resembling acute leukaemia.
This report concerns a patient with known BCR::ABL1-positive CML, previously treated with Imatinib, who has now presented in a blast crisis. Laboratory and morphological analysis confirms a diagnosis of acute basophilic leukaemia.
- Rarity: Progression to a basophilic lineage blast crisis is an uncommon but well-documented event in CML. It represents a distinct and highly aggressive form of disease transformation.
- Pathophysiology: The blast crisis occurs due to the accumulation of additional genetic mutations beyond BCR::ABL1. Imatinib resistance can develop, often through mutations in the BCR::ABL1 kinase domain, allowing a clone of leukemic cells to proliferate uncontrollably and differentiate along the basophil lineage.
Diagnostic Features:
- Morphology: Blasts with basophilic differentiation, characterized by coarse, metachromatic staining granules on Wright-Giemsa stain.
- Immunophenotype (Flow Cytometry): Blasts are typically positive for CD34, HLA-DR, CD13, CD33, and may show expression of CD9 and CD25. A key feature is the presence of basophil-specific markers or the characteristic high side-scatter property of the granules.
- Clinical Presentation: Patients may present with symptoms related to hyperhistaminemia, such as pruritus, flushing, gastric hypersecretion, and hypotension, due to the release of histamine from basophil granules.
While Imatinib is highly effective in the chronic phase, its failure or the development of resistance is the primary risk factor for progression to blast crisis. This case underscores the critical need for continuous molecular monitoring in CML patients to detect resistance early and switch to a more potent TKI (e.g., 2nd/3rd generation) or consider allogeneic stem cell transplantation.
The development of acute basophilic leukaemia in a patient with CML treated with Imatinib represents a high-risk, treatment-resistant blast crisis. Management is challenging and requires rapid diagnosis, cessation of the failing TKI, initiation of a alternative TKI (if no T315I mutation is present), and aggressive acute leukaemia-style chemotherapy, with the goal of achieving remission and proceeding to allogeneic stem cell transplantation.
Case presentation
Name: Mr. B, N Armeia, 25-year Male, single, student Egyptian borne and lived in Egypt
Medical ID: 3300/2022
HOSPITAL: HARMEL CANCER CENTER (DIC)
Clinical Timeline and Summary
This case outlines the aggressive course of a 25-year-old male with a history of CML translocation (9;22) positive, who was initially diagnosed in late 2022 and controlled ans was in molecular remission on imatinib till November 2024 patient started to Complain from persistent fever for 14 days, attacks of intermittent fever associated with weight loss, night sweats, easy fatigability, and left hypochondrial pain and sever itching.
- Examination: Notable for splenomegaly.
- Key Initial Investigations:
CBC: Leukocytosis (WBC 24.3 x 10³/µL), anemia (Hb 8.4 g/dL), thrombocytopenia (Plt 59 x 10³/µL), and basophilia (7.3%). investigations
Virology: Negative
CT chest: Free
Echo: Normal
Crp: 40
Ultrasound: Marked splenomegaly.19cm
Bone Marrow (Nov 2024): Hypercellular with ~65% immature basophils.
Flow Cytometry (Nov 2024): Immunophenotype consistent with Acute Basophilic Leukemia (CD33+, CD13+, CD117+, CD11b+, HLA-DR+).
Molecular Studies: BCR-ABL1 p210 positive at 35% IS.
Induction Therapy: The patient received the "3+7" regimen combined with Dasatinib.
Response Assessment: Day 14 BMA: Complete Morphological Remission. Day 32 BMA: Complete Remission with Incomplete Hematological Recovery (CRi).
The patient's course was severely complicated following induction:
- Severe Intramuscular Hemorrhage: The patient developed a significant intramuscular hematoma, likely secondary to Brachial artrey blood sample taking, Superficial ultrasound shows hematoma 5*2cm surgical consultation was done and due to prolonged thrombocytopenia there was no surgical role
- Compartment Syndrome: This was suspected as a consequence of the expanding hematoma. Skin Ulceration: A few days later, the patient developed skin ulceration on the left arm at the site of the complication. Management: The raw area was managed conservatively with daily dressings. No surgical intervention (e.g., fasciotomy) could be performed due to the patient's critical condition and incompatibility with major surgery, primarily persistent cytopenias and high infection risk. Patient developed Klebsiella MDR ttt acc to our antibiotic policy
- HLA typing was done at day 50 of induction due to delayed recovery and patient was haplo identical with his possible donor only sibling
-
Due to these severe complications and the patient's debilitated state, the planned consolidative stem cell transplant was no longer feasible neither consolidation with HDAC.
- Revised Consolidation: Given the contraindication for HDAC, consolidation was attempted with Dasatinib monotherapy100 mg per day This was the plane with co-management of surgery department till preparation for skin graft then proceeding to haplo BMT
- Bone Marrow Flow Cytometry (April 30, 2025): A follow-up immunophenotyping study revealed ~70% myeloblasts with an identical immunophenotype to the initial diagnosis (CD7 48%, CD33 87%, CD13 53%, CD117 71%).
- Conclusion: This confirmed morphological and immunophenotypic relapse of Acute Basophilic Leukemia approximately three months after achieving initial remission.
Comment
Immunophenotyping is performed on the provided BM sample with a reported history of CML. using CD45/SSC gating strategy.IT reveals about 70% myeloblast expressing the immunophenotype presented in the table above.
Treatment plan after relapse was 7+3 to start with cytosar and dose reduction of Adriamycin by 30%, pre chemotherapy ct-chest echo pan cultures was done n was normal at day three of Arac patient developed sinus tachycardia, no other lab abnormalities considering electrolytes cytosar was held, after short period late that night patient developed fits and sudden cardiac arrest. the patient was initiated on cytarabine-based salvage therapy. Shortly thereafter, he experienced an episode of sinus tachycardia, which rapidly progressed to fatal cardiac arrest. A recent echocardiogram had been normal. The etiology of the cardiac arrest is multifactorial, with the most likely causes being:
- Hyperhistaminemia from basophil degranulation secondary to cytarabine-induced blast lysis.
- Treatment-related mortality in a profoundly debilitated host, with possible contributions from sepsis or subclinical tumor lysis syndrome.
- Direct, albeit rare, cardiotoxicity of cytarabine.
Conclusion and Discussion
This case demonstrates the highly aggressive nature of CML blast crisis, particularly the rare basophilic lineage. Key learning points include:
Efficacy and Toxicity: While the "3+7 + TKI" regimen can induce remission, it carries a significant risk of severe toxicity, especially in patients with high tumor burden and pre-existing cytopenias.
Impact of Complications: The devastating complication of hemorrhage and compartment syndrome directly prevented the execution of the only potentially curative option—allogeneic stem cell transplant.
Limitations of TKI Monotherapy: For blast crisis, tyrosine kinase inhibitor monotherapy is insufficient as a long-term consolidative strategy, as evidenced by the rapid relapse once intensive therapy was discontinued.
Poor Prognosis: The relapse, compounded by the patient's compromised clinical state, signifies a very poor prognosis. Further management options are extremely limited and would focus on palliative care or experimental therapies.
The Challenge of Salvage Therapy: This case underscores the extreme difficulty of managing relapsed blast crisis. The severe treatment-related morbidity from the first induction (hemorrhage, compartment syndrome, infection) left the patient in a debilitated state, making him a poor candidate for further intensive chemotherapy. The fatal cardiac arrest following cytarabine administration highlights the exceedingly high risk of treatment-related mortality in this setting. When curative-allogeneic transplant is no longer feasible, the risk-benefit ratio of salvage chemotherapy must be carefully weighed against the option of best supportive care.
References
- Wang, S. A., Hasserjian, R. P., & Loew, J. M. (2019). Acute Basophilic Leukemia. In WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues (Revised 4th ed., pp. 96-98). IARC.
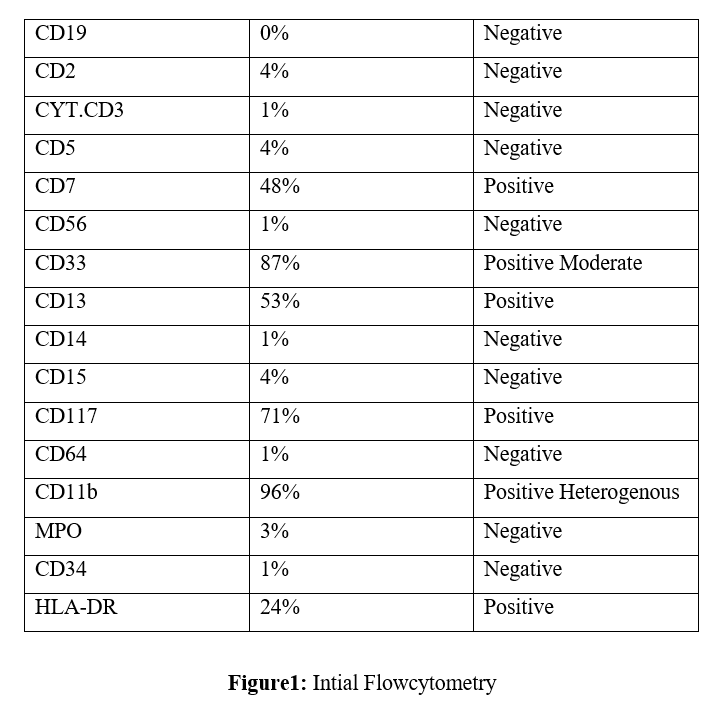
Figure 1
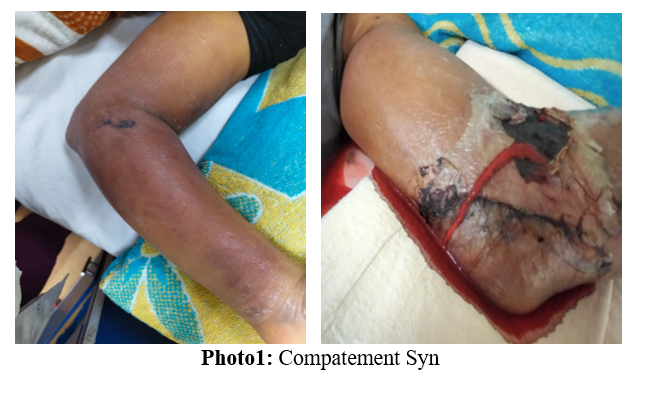
Figure 2
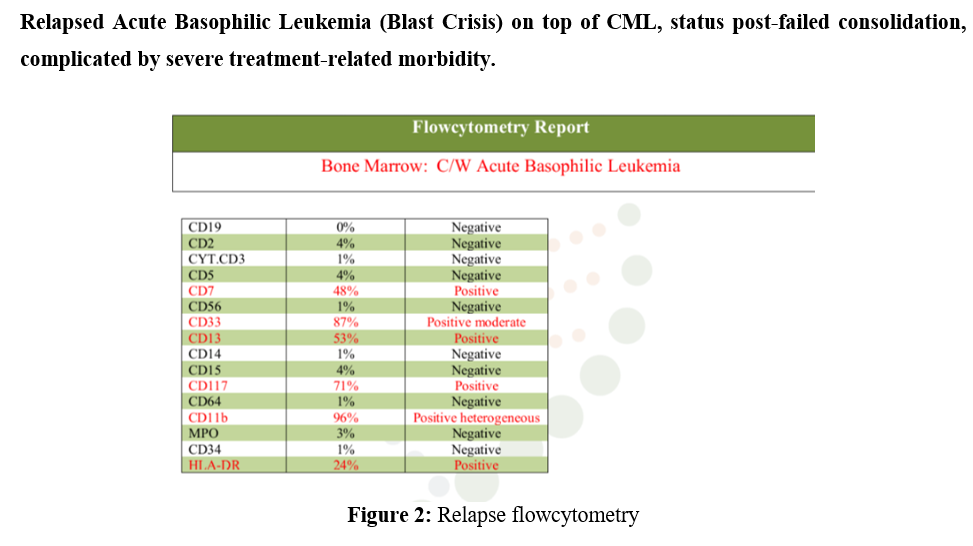
Figure 3
